8.6: Measuring the Specific Surface Area of Nanoparticle Suspensions using NMR
- Page ID
- 55922
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Surface area is a property of immense importance in the nano-world, especially in the area of heterogeneous catalysis. A solid catalyst works with its active sites binding to the reactants, and hence for a given active site reactivity, the higher the number of active sites available, the faster the reaction will occur. In heterogeneous catalysis, if the catalyst is in the form of spherical nanoparticles, most of the active sites are believed to be present on the outer surface. Thus it is very important to know the catalyst surface area in order to get a measure of the reaction time. One expresses this in terms of volume specific surface area, i.e., surface area/volume although in industry it is quite common to express it as surface area per unit mass of catalyst, e.g., m2/g.
Overview of NMR
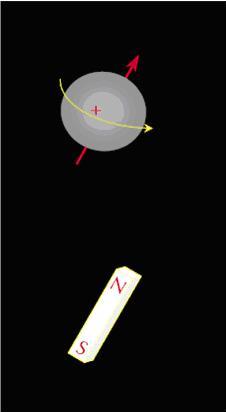
Nuclear magnetic resonance (NMR) is the study of the nuclei of the response of an atom to an external magnetic field. Many nuclei have a net magnetic moment with I ≠ 0, along with an angular momentum in one direction where I is the spin quantum number of the nucleus. In the presence of an external magnetic field, a nucleus would precess around the field. With all the nuclei precessing around the external magnetic field, a measurable signal is produced. NMR can be used on any nuclei with an odd number of protons or neutrons or both, like the nuclei of hydrogen (1H), carbon (13C), phosphorous (31P), etc. Hydrogen has a relatively large magnetic moment (μ = 14.1 x 10-27 J/T) and hence it is used in NMR logging and NMR rock studies. The hydrogen nucleus composes of a single positively charged proton that can be seen as a loop of current generating a magnetic field. It is may be considered as a tiny bar magnet with the magnetic axis along the spin axis itself as shown in Figure \(\PageIndex{1}\). In the
absence of any external forces, a sample with hydrogen alone will have the individual magnetic moments
randomly aligned as shown in Figure \(\PageIndex{2}\).
Nuclear magnetic resonance (NMR) is the study of the nuclei of the response of an atom to an external magnetic field. Many nuclei have a net magnetic moment with I≠0, along with an angular momentum in one direction where I is the spin quantum number of the nucleus. In the presence of an external magnetic field, a nucleus would precess around the field. With all the nuclei precessing around the external magnetic field, a measurable signal is produced.
NMR can be used on any nuclei with an odd number of protons or neutrons or both, like the nuclei of hydrogen (1H), carbon (13C), phosphorous (31P), etc. Hydrogen has a relatively large magnetic moment (μ = 14.1 x 10-27 J/T) and hence it is used in NMR logging and NMR rock studies. The hydrogen nucleus composes of a single positively charged proton that can be seen as a loop of current generating a magnetic field. It is may be considered as a tiny bar magnet with the magnetic axis along the spin axis itself as shown in Figure. In the absence of any external forces, a sample with hydrogen alone will have the individual magnetic moments randomly aligned as shown in Figure \(\PageIndex{2}\).

Advantages of NMR over BET Technique
BET measurements follow the BET (Brunner-Emmet-Teller) adsorption isotherm of a gas on a solid surface. Adsorption experiments of a gas of known composition can help determine the specific surface area of the solid particle. This technique has been the main source of surface area analysis used industrially for a long time. However BET techniques take a lot of time for the gas-adsorption step to be complete while one shall see in the course of this module that NMR can give you results in times averaging around 30 minutes depending on the sample. BET also requires careful sample preparation with the sample being in dry powder form, whereas NMR can accept samples in the liquid state as well.
NMR Relaxation Mechanism in Solid Suspensions
Calculations
From an atomic stand point, T1 relaxation occurs when a precessing proton transfers energy with its surroundings as the proton relaxes back from higher energy state to its lower energy state. With T2 relaxation, apart from this energy transfer there is also dephasing and hence T2 is less than T1 in general. For solid suspensions, there are three independent relaxation mechanisms involved:-
- Bulk fluid relaxation which affects both T1 and T2 relaxation.
- Surface relaxation, which affects both T1 and T2 relaxation.
- Diffusion in the presence of the magnetic field gradients, which affects only T2 relaxation
These mechanisms act in parallel so that the net effects are given by \ref{1} and \ref{2}.
\[ \frac{1}{T_{2}}=\frac{1}{T_{2, bulk}}\ +\ \frac{1}{T_{2,surface}}+\frac{1}{T_{2,diffusion}} \label{1} \]
\[ \frac{1}{T_{1}} = \frac{1}{T_{1, bulk}}\ +\ \frac{1}{T_{1,surface}} \label{2} \]
The relative importance of each of these terms depend on the specific scenario. For the case of most solid suspensions in liquid, the diffusion term can be ignored by having a relatively uniform external magnetic field that eliminates magnetic gradients. Theoretical analysis has shown that the surface relaxation terms can be written as
\ref{3} and \ref{4}.
\[ \frac{1}{T_{1,surface}} = \rho _{1} (\frac{S}{V})_{particle} \label{3} \]
\[ \frac{1}{T_{2,surface}} = \rho_{2} (\frac{S}{V})_{particle} \label{4} \]
Thus one can use T1 or T2 relaxation experiment to determine the specific surface area. We shall explain the case of the T2 technique further as \ref{5}.
\[ \frac{1}{T_{2}} = \frac{1}{T_{2,bulk}}+ \rho_{2}(\frac{S}{V})_{particle} \label{5} \]
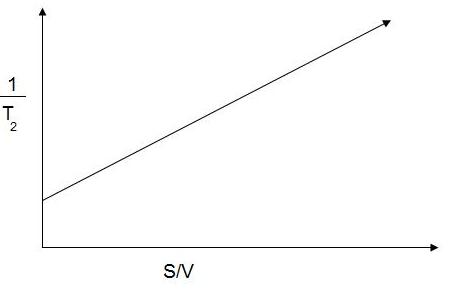
One can determine T2 by spin-echo measurements for a series of samples of known S/V values and prepare a calibration chart as shown in Figure \(\PageIndex{3}\), with the intercept as 1/T2,bulk and the slope as ρ2, one can thus find the specific surface area of an unknown sample of the same material.
Sample Preparation and Experimental Setup
The sample must be soluble in the solvent. For proton NMR, about 0.25-1.00 mg/mL are needed depending on the sensitivity of the instrument.
The solvent properties will have an impact of some or all of the spectrum. Solvent viscosity affects obtainable resolution, while other solvents like water or ethanol have exchangeable protons that will prevent the observation of such exchangeable protons present in the solute itself. Solvents must be chosen such that the temperature dependence of solute solubility is low in the operation temperature range. Solvents containing aromatic groups like benzene can cause shifts in the observed spectrum compared to non-aromatic solvents.
NMR tubes are available in a wide range of specifications depending on specific scenarios. The tube specifications need to be extremely narrow while operating with high strength magnetic fields. The tube needs to be kept extremely clean and free from dust and scratches to obtain good results, irrespective of the quality of the tube. Tubes can cleaned without scratching by rinsing out the contents and soaking them in a degreasing solution, and by avoiding regular glassware cleaning brushes. After soaking for a while, rinse with distilled water and acetone and dry the tube by blowing filterened nitrogen gas through a pipette or by using a swob of cotton wool.
Filter the sample solution by using a Pasteur pipette stuffed with a piece of cotton wool at the neck. Any suspended material like dust can cause changes in the spectrum. When working with dilute aqueous solutions, sweat itself can have a major effect and so gloves are recommended at all times.
Sweat contains mainly water, minerals (sodium 0.9 g/L, potassium 0.2 g/L, calcium 0.015 g/L, magnesium 0.0013 g/L and other trace elements like iron, nickel, zinc, copper, lead and chromium), as well as lactate and urea. In presence of a dilute solution of the sample, the proton-containing substances in sweat (e.g., lactate and urea) can result in a large signal that can mask the signal of the sample.
The NMR probe is the most critical piece of equipment as it contains the apparatus that must detect the small NMR signals from the sample without adding a lot of noise. The size of the probe is given by the diameter of the NMR tube it can accommodate with common sizes 5, 10 and 15 mm. A larger size probe can be used in the case of less sensitive samples in order to get as much solute into the active zone as possible. When the sample is available in less quantity, use a smaller size tube to get an intrinsically higher sensitivity.
NMR Analysis
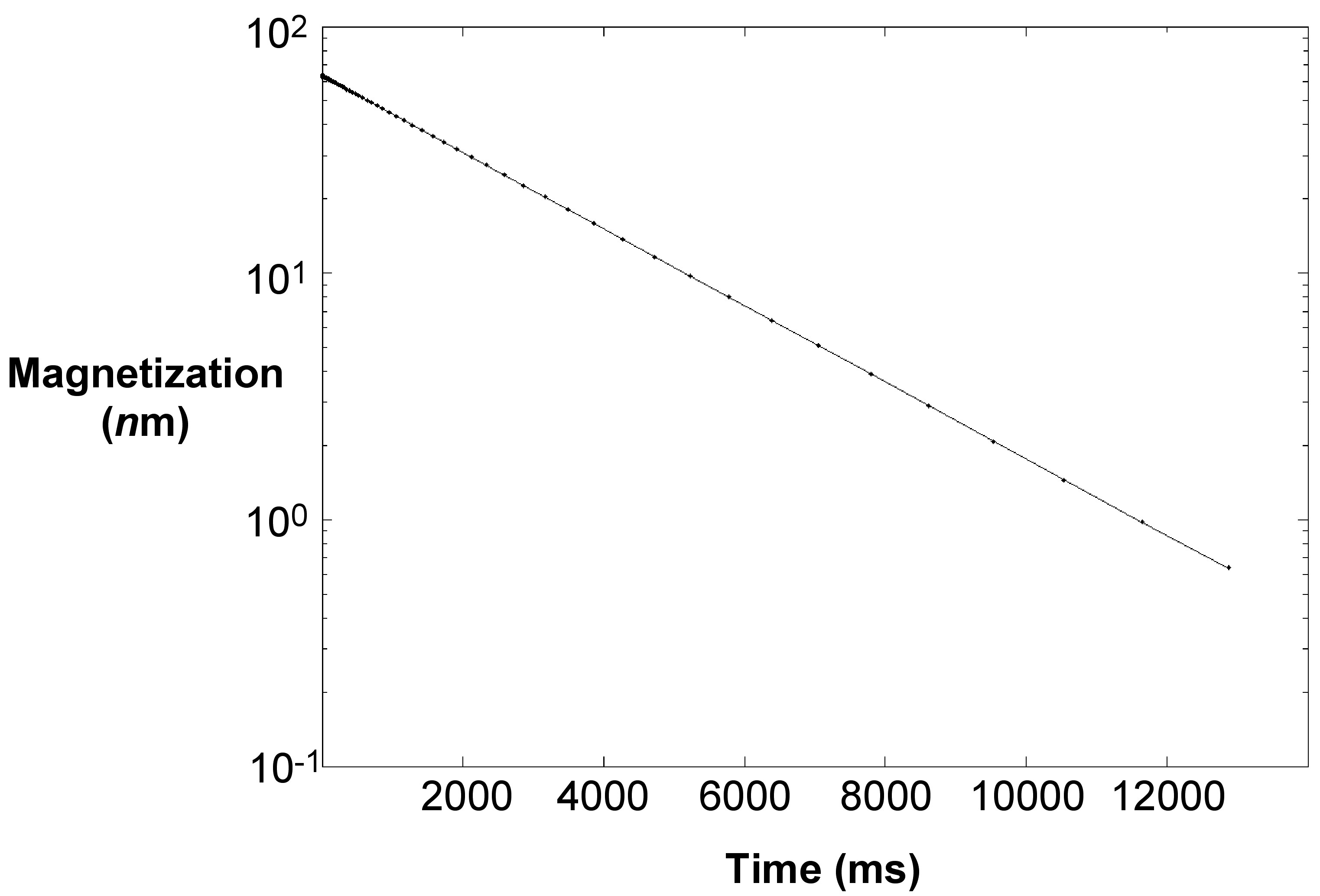
A result sheet of T2 relaxation has the plot of magnetization versus time, which will be linear in a semi-log plot as shown in Figure \(\PageIndex{4}\). Fitting it to the equation, we can find T2 and thus one can prepare a calibration plot of 1/T2 versus S/V of known samples.
Limitations of the T2 Technique
The following are a few of the limitations of the T2 technique:
- One can’t always guarantee no magnetic field gradients, in which case the T1 relaxation technique is to be used. However this takes much longer to perform than the T2 relaxation.
- There is the requirement of the odd number of nucleons in the sample or solvent.
- The solid suspension should not have any para- or ferromagnetic substance (for instance, organics like hexane tend to have dissolved O2 which is paramagnetic).
- The need to prepare a calibration chart of the material with known specific surface area.
Example of Usage
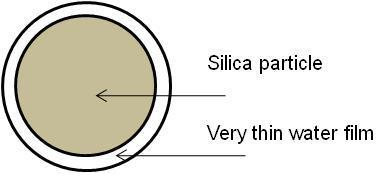
A study of colloidal silica dispersed in water provides a useful example. Figure \(\PageIndex{5}\) shows a representation of an individual silica particle.
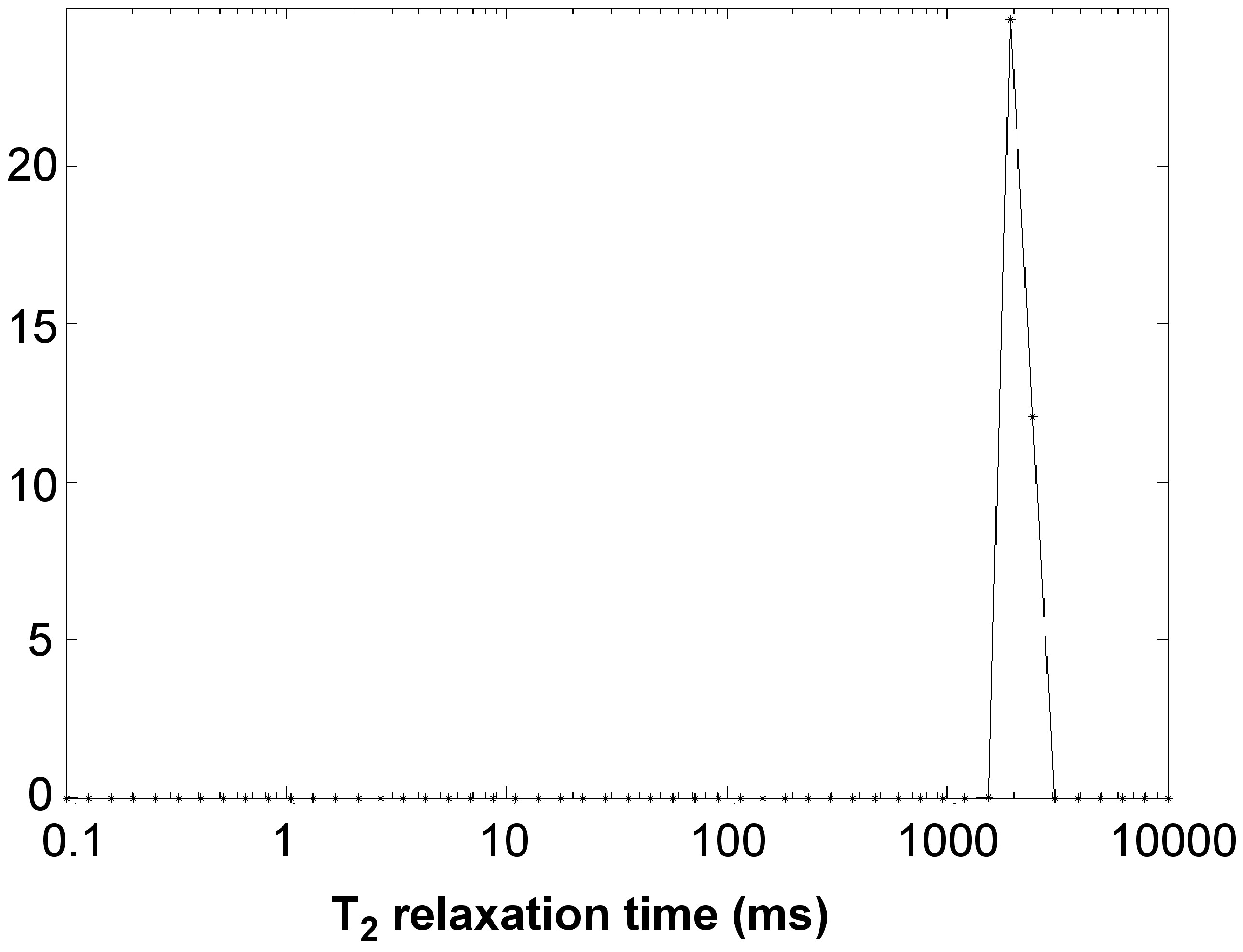
A series of dispersion in DI water at different concentrations was made and surface area calculated. The T2 relaxation technique was performed on all of them with a typical T2 plot shown in Figure \(\PageIndex{6}\) and T2 was recorded at 2117 milliseconds for this sample.
A calibration plot was prepared with 1/T2 – 1/T2,bulk as ordinate (the y-axis coordinate) and S/V as abscissa (the x-axis coordinate). This is called the surface relaxivity plot and is illustrated in Figure \(\PageIndex{7}\).
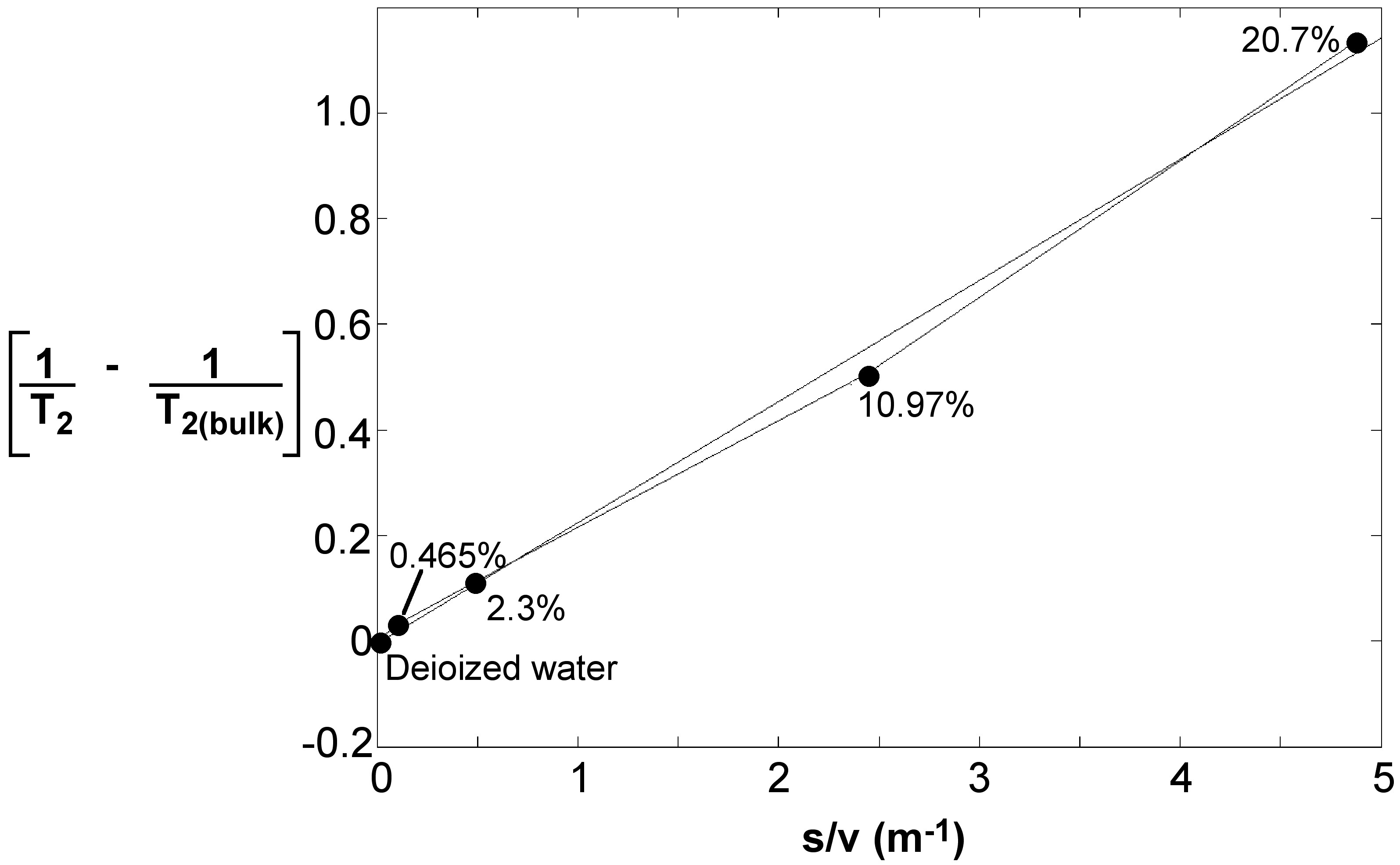
Accordingly for the colloidal dispersion of silica in DI water, the best fit resulted in \ref{6}, from which one can see that the value of surface relaxivity, 2.3 x 10-8, is in close accordance with values reported in literature.
\[ \frac{1}{T_{2}}\ -\ \frac{1}{T_{2,bulk}}\ =\ 2.3 \times 10^{-8} (\frac{S}{V})\ -\ 0.0051 \label{6} \]
The T2 technique has been used to find the pore-size distribution of water-wet rocks. Information of the pore size distribution helps petroleum engineers model the permeability of rocks from the same area and hence determine the extractable content of fluid within the rocks.
Usage of NMR for surface area determination has begun to take shape with a company, Xigo nanotools, having developed an instrument called the Acorn AreaTM to get surface area of a suspension of aluminum oxide. The results obtained from the instrument match closely with results reported by other techniques in literature. Thus the T2 NMR technique has been presented as a strong case to obtain specific surface areas of nanoparticle suspensions.


