6.2D: Inductively Coupled Plasma
- Page ID
- 111877
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)A plasma is a gaseous mixture in which a significant proportion of the gas-phase species are ionized. An illustration of an inductively coupled plasma (ICP) is shown in Figure \(\PageIndex{5}\). The device consists of a quartz tube (about ¾ inch in diameter), the end of which is wrapped in a high power radiofrequency (RF) induction coil. Argon gas flows down the quartz tube at a high rate (about 15 liters/minute). A current of electricity is run through the RF coil, which produces a magnetic field inside the end of the quartz tube. Sparking the argon creates some Ar+ ions, which are now paramagnetic and absorb energy from the magnetic field. The argon ions absorb enough energy that a plasma is created in the area of the tube covered by the RF induction coil. The nature of the magnetic field causes the plasma to flow in a closed annular path (basically a donut shape). What is especially impressive is that enough energy is absorbed from the magnetic field to heat the plasma up to a temperature of about 6,000 K. As a comparison, this temperature is about the same as the temperature of the surface of the sun. The hot temperature means that new argon flowing into the plasma is ionized, which maintains the plasma. The plasma is kept from melting the walls of the quartz tube by an additional tangential flow of argon along the walls of the tube. Finally, the sample is nebulized and sprayed as an aerosol mist into the center of the plasma.
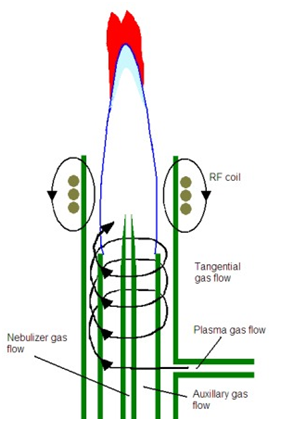
An ICP offers several advantages over flame and furnace atomization sources. One is that it is so hot that it leads to a more complete atomization and leads to the formation of many excited state atoms. Because sufficient numbers of atoms are excited, they can be detected by emission instead of absorbance. The illustration in Figure \(\PageIndex{5}\) shows the plume that forms in an ICP above the RF coil. Above the plasma is a zone in which argon regeneration occurs. A continuum background emission is given off in this zone. Above this zone in the plume, there are excited atoms that emit the characteristic lines of each particular element in the sample. In our discussion of fluorescence spectroscopy, we learned that emission methods have an inherent sensitivity advantage over absorbance methods. This occurs because emission entails measuring a small signal over no background and absorbance entails measuring a small difference between two large signals. This same sensitivity advantage exists for measurements of atomic emission over atomic absorbance. Light emitted by atoms in the plume can be measured either radially (off to the side of the plume) or axially (looking down into the plume. Axial measurements are often more sensitive because of the increase in path length. However, in some cases, depending on the element profile in the plasma, radial measurements may be preferable. Instruments today often allow for either axial or radial measurements.
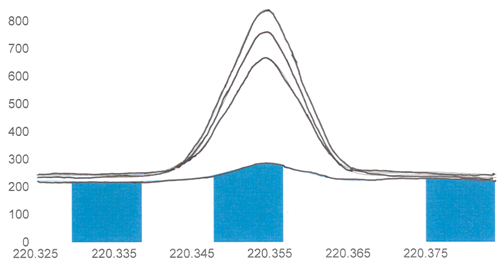
A second advantage of an ICP is that all of the elements can be measured simultaneously. All metals in the sample will be atomized at the same time and all are emitting light. Some instruments measure elements in a sequential arrangement. In this case, the operator programs in the elements to be measured, and the monochromator moves one-by-one through the specific wavelengths necessary for the measurement of each element. Other instruments use an array detector with photoactive pixels that can measure all of the elements at once. Array instruments are preferable as the analysis will be faster and less sample is consumed. Figure \(\PageIndex{6}\) shows the printout of the pixels on a array detector that include and surround the lead emission that occurs at 220.353 nm. The peak due to the lead emission from the four different samples is apparent. Also note that there is a background emission on the neighboring pixels and the intensity of this background emission must be subtracted from the overall emission occurring at the lead wavelength.
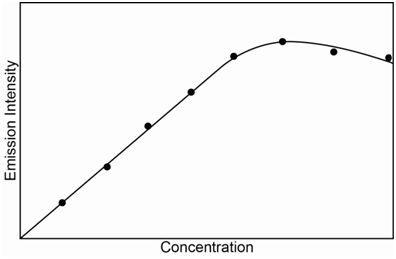
An observation with emission spectroscopy to be aware of is the possibility of self-absorption. We already discussed this in the unit on fluorescence spectroscopy. Self-absorption refers to the situation in which an excited state atom emits a photon that is then absorbed by another atom in the ground state. If the photon was headed toward the detector, then it will not be detected. Self-absorption becomes more of a problem at higher concentrations as the emitted photons are more likely to encounter a ground state atom. The presence of self-absorption can lead to a diminishment of the response in a calibration curve at high concentrations as shown in Figure \(\PageIndex{7}\). Atomic emission transitions always correspond with absorption transitions for the element being analyzed so the likelihood of observing self-absorption is higher in atomic emission spectroscopy than in fluorescence spectroscopy. For a set of samples with unknown concentrations of analyte, it may be desirable to test one or two after dilution to insure that the concentration decreases by a proportional factor and that the samples are not so high in concentration to be out in the self-absorption portion of the standard curve.
Another advantage is that the high number of Ar+ ions and free electrons suppress the ionization of other elements being measure, thereby increasing the number of neutral atoms whose emission is being measured. The argon used to generate the plasma is chemically inert compared to the chemical species that make up a flame, which increases the atomization efficiency. The inductively coupled plasma tends to be quite stable and reproducible. The combination of high temperature with chemically inert environmental reduces matrix effects in the plasma relative to other atomization sources, but it does not eliminate them and matrix effects must always be considered. Some elements (e.g., mercury, arsenic, phosphorus) that are impractical to analyze on a flame or furnace instrument without specialized atomization techniques can often be measured on an ICP.
A final advantage of the plasma is that there are now methods to introduce the atoms into a mass spectrometer (MS). The use of the mass spectrometer may further reduce certain matrix effects. Also, mass spectrometry usually provides more sensitive detection than emission spectroscopy.


