1.1: Introduction to Molecular Spectroscopy
- Page ID
- 111313
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Molecular spectroscopy relates to the interactions that occur between molecules and electromagnetic radiation. Electromagnetic radiation is a form of radiation in which the electric and magnetic fields simultaneously vary. One well known example of electromagnetic radiation is visible light. Electromagnetic radiation can be characterized by its energy, intensity, frequency and wavelength.
What is the relationship between the energy (E) and frequency (\(\nu\)) of electromagnetic radiation?
The fundamental discoveries of Max Planck, who explained the emission of light by a blackbody radiator, and Albert Einstein, who explained the observations in the photoelectric effect, led to the realization that the energy of electromagnetic radiation is proportional to its frequency. The proportionality expression can be converted to an equality through the use of Planck’s constant.
\[\mathrm{E = h\nu} \nonumber \]
What is the relationship between the energy and wavelength (\(\lambda\)) of electromagnetic radiation?
Using the knowledge that the speed of electromagnetic radiation (c) is the frequency times the wavelength (\(\mathrm{c = \lambda\nu}\)), we can solve for the frequency and substitute in to the expression above to get the following.
\[\mathrm{E = \dfrac{hc}{\lambda}} \nonumber \]
Therefore the energy of electromagnetic radiation is inversely proportional to the wavelength. Long wavelength electromagnetic radiation will have low energy. Short wavelength electromagnetic radiation will have high energy.
Write the types of radiation observed in the electromagnetic spectrum going from high to low energy. Also include what types of processes occur in atoms or molecules for each type of radiation.
| High E, high \(\nu\), short \(\lambda\): | \(\gamma\)-rays – Nuclear energy transitions |
| X-rays – Inner-shell electron transitions | |
| Ultraviolet – Valence electron transitions | |
| Visible – Valence electron transitions | |
| Infrared – Molecular vibrations | |
| Microwaves – Molecular rotations, Electron spin transitions | |
| Low E, low \(\nu\), long \(\lambda\): | Radiofrequency – Nuclear spin transitions |
Atoms and molecules have the ability to absorb or emit electromagnetic radiation. A species absorbing radiation undergoes a transition from the ground to some higher energy excited state. A species emitting radiation undergoes a transition from a higher energy excited state to a lower energy state. Spectroscopy in analytical chemistry is used in two primary manners: (1) to identify a species and (2) to quantify a species.
Identification of a species involves recording the absorption or emission of a species as a function of the frequency or wavelength to obtain a spectrum (the spectrum is a plot of the absorbance or emission intensity as a function of wavelength). The features in the spectrum provide a signature for a molecule that may be used for purposes of identification. The more unique the spectrum for a species, the more useful it is for compound identification. Some spectroscopic methods (e.g., NMR spectroscopy) are especially useful for compound identification, whereas others provide spectra that are all rather similar and therefore not as useful. Among methods that provide highly unique spectra, there are some that are readily open to interpretation and structure assignment (e.g., NMR spectra), whereas others (e.g., infrared spectroscopy) are less open to interpretation and structure assignment. Since molecules do exhibit unique infrared spectra, an alternative means of compound identification is to use a computer to compare the spectrum of the unknown compound to a library of spectra of known compounds and identify the best match. In this case, identification is only possible if the spectrum of the unknown compound is in the library.
Quantification of a species using a spectroscopic method involves measuring the magnitude of the absorbance or intensity of the emission and relating that to the concentration. At this point, we will focus on the use of absorbance measurements for quantification.
Consider a sample through which you will send radiation of a particular wavelength as shown in Figure \(\PageIndex{1}\). You measure the power from the radiation source (Po) using a blank solution (a blank is a sample that does not have any of the absorbing species you wish to measure). You then measure the power of radiation that makes it through the sample (P).
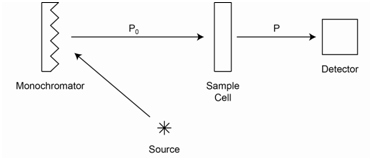
The ratio P/Po is a measure of how much radiation passed through the sample and is defined as the transmittance (T).
\[\mathrm{T = \dfrac{P}{P_o} \hspace{20px} and \hspace{20px} \%T = \left(\dfrac{P}{P_o}\right)\times 100} \nonumber \]
The higher the transmittance, the more similar P is to Po. The absorbance (A) is defined as:
\[\mathrm{A = -\log T \textrm{ or } \log\left(\dfrac{P_o}{P}\right).} \nonumber \]
The higher the absorbance, the lower the value of P, and the less light that makes it through the sample and to the detector.


