1.1: The structure of crystals
- Page ID
- 352420
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)In the context of this chapter, you will also be invited to visit these sections...
- The crystalline state
- Interatomic forces in crystals
- Early historical notes about crystals and crystallography
We all have heard about natural minerals and crystals. We find them daily without entering a museum. A rock and a mountain are made up of minerals, as crystalline as a lump of sugar, a bit of porcelain or a gold ring. However, only occasionally is the size of a crystal large enough to draw our attention, as is the case of these beautiful mineral examples of: Diamond (pure carbon) - Quartz (silicon dioxide) - Scapolite (aluminum silicate) - Pyrite (iron sulfide)

Several of these images are property of Amethyst Galleries, Inc.
Other excellent images of minerals can be found through this link.
Although you can continue reading these pages without any special difficulty, probably you would like to know some aspects about the historical development of our understanding of the crystals. For these readers we offer some further notes that can be found through this link.
The ancient Greeks identified quartz with the word crystal (κρύσταλλος, crustallos, or phonetically kroos'-tal-los = cold + drop), ie, very cold icicles of extraordinary hardness. But the formation of crystals is not a unique property of minerals; they are also found (but not necessarily in a natural manner) in the so-called organic compounds, and even in nucleic acids, in proteins and in viruses...
A crystal is a material whose constituents, such as atoms, molecules or ions, are arranged in a highly ordered microscopic structure. These constituents are held together by interatomic forces (chemical bonds) such as metallic bonds, ionic bonds, covalent bonds, van der Waals bonds, and others.
The crystalline state of matter is the state with the highest order, ie, with very high internal correlations and at the greatest distance range. This is reflected in their properties: anisotropic and discontinuous. Crystals usually appear as unadulterated, homogenous and with well-defined geometric shapes (habits) when they are well-formed. However, as we say in Spanish, "the habit does not make the monk" (clothes do not make the man) and their external morphology is not sufficient to evaluate the crystallinity of a material.
The movie below shows the process of crystal growth of lysozyme (a very stable enzyme) from an aqueous medium. The duration of the real process, that takes a few seconds on your screen, corresponds approximately to 30 minutes.

The original movie was found on an old website offered by George M. Sheldrik.
The figure on the left shows a representation of the faces of a given crystal. If your browser allows the Java Runtime, clicking on the image will open a new window and you will be able to turn this object. If you do not have this application, you can still observe the model rotation in continuous mode from this link.
Other Java pop-ups of faces and forms (habits) for ideal crystals can be obtained through this link.
So, we ask ourselves, what is unique about crystals which distinguishes them from other types of materials? The so-called microscopic crystal structure is characterized by groups of ions, atoms or molecules arranged in terms of some periodic repetition model, and this concept (periodicity) is easy to understand if we look at the drawings in an carpet, in a mosaic, or a military parade...

Repeated motifs in a carpet

Repeated motifs in a mosaic

Repeated motifs in a military parade
If we look carefully at these drawings, we will discover that there is always a fraction of them that is repeated. In crystals, the atoms, ions or molecules are packed in such a way that they give rise to "motifs" (a given set or unit) that are repeated every 5 Angstrom, up to the hundreds of Angstrom (1 Angstrom = 10-8 cm), and this repetition, in three dimensions, is known as the crystal lattice. The motif or unit that is repeated, by orderly shifts in three dimensions, produces the network (the whole crystal) and we call it the elementary cell or unit cell. The content of the unit being repeated (atoms, molecules, ions) can also be drawn as a point (the reticular point) that represents every constituent of the motif. For example, each soldier in the figure above could be a reticular point.
But there are occasions where the repetition is broken, or it is not exact, and this feature is precisely what distinguishes a crystal from glass, or in general, from materials called amorphous (disordered or poorly ordered)...
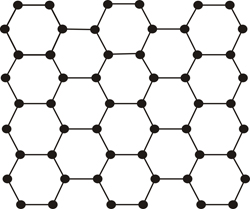
Planar atomic model of an ordered material (crystal)
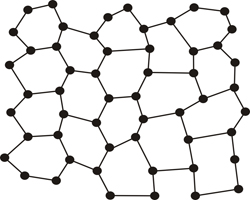
Planar atomic model of glass (an amorphous material)
However, matter is not entirely ordered or disordered (crystalline or non-crystalline) and so we can find a continuous degradation of the order (crystallinity degree) in materials, which goes from the perfectly ordered (crystalline) to the completely disordered (amorphous). This gradual loss of order which is present in materials is equivalent to what we see in the small details of the following photograph of gymnastic training, which is somewhat ordered, but there are some people wearing pants, other wearing skirts, some in different positions or slightly out of line...

In the crystal structure (ordered) of inorganic materials, the repetitive units (or motifs) are atoms or ions, which are linked together in such a way that we normally do not distinguish isolated units and hence their stability and hardness (ionic crystals, mainly)...
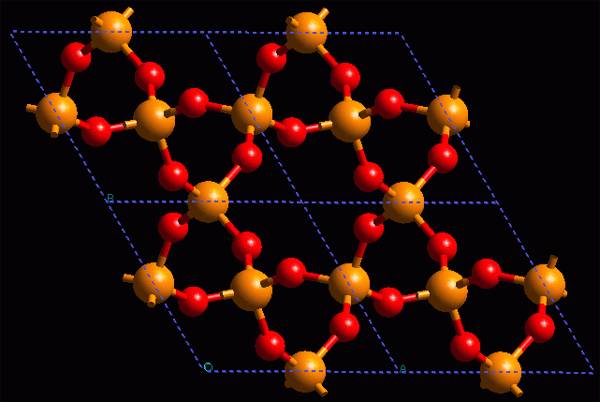
Crystal structure of an inorganic material: α-quartz
Where we clearly distinguish isolated units is in the case of the so-called organic materials, where the concept of the isolated entity (molecule) appears. Molecules are made up of atoms linked together. However, the links between the molecules within the crystal are very weak (molecular crystals). Thus, they are generally softer and more unstable materials than the inorganic ones.
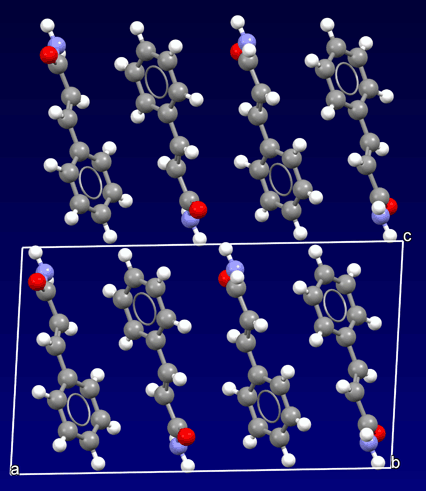
Crystal structure of an organic material: Cinnamamide
Protein crystals also contain molecular units (molecules), as in the organic materials, but much larger. The type of forces that bind these molecules are also similar, but their packing in the crystals leaves many holes that are filled with water molecules (not necessarily ordered) and hence their extreme instability...

Crystal structure of a protein: AtHal3.
The molecular packing produces very large holes
The different packing modes in crystals lead to the so-called polymorphic phases (allotropic phases of the elements) which confer different properties to these crystals (to these materials). For example, we all know the different appearances and properties of the chemical element carbon, which is present in Nature in two different crystalline forms, diamond and graphite:


Right: Graphite (pure carbon)
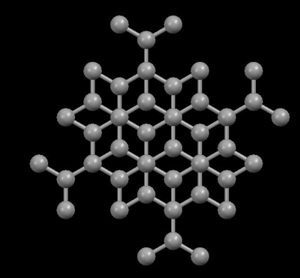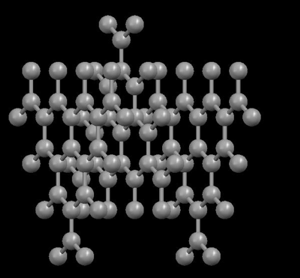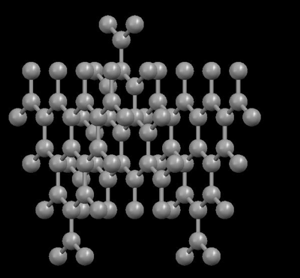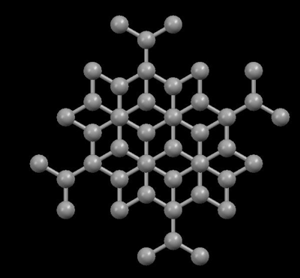
Graphite is black, soft and an excellent lubricant, suggesting that its atoms must be distributed (packed) in such a way as to explain these properties. However, diamonds are transparent and very hard, so that we can expect their atoms very firmly linked. Indeed, their sub-microscopic structures (at atomic level) show us their differences ...
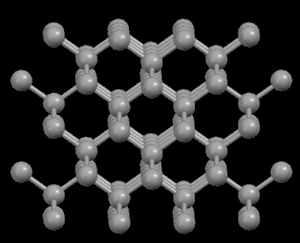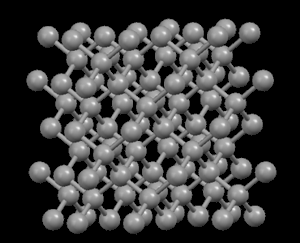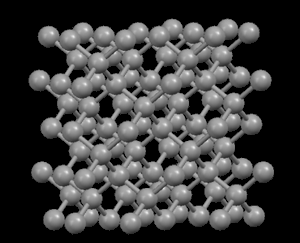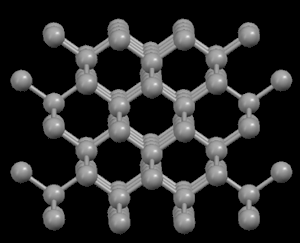
Right: Graphite, showing its layered crystal structure
In the diamond structure, each carbon atom is linked to four other ones in the form of a very compact three-dimensional network (covalent crystals), hence its extreme hardness and its property as an electric insulator. However, in the graphite structure, the carbon atoms are arranged in parallel layers much more separated than the atoms in a single layer. Due to these weak links between the atomic layers of graphite, the layers can slide, without much effort, and hence graphite's suitability as a lubricant, its use for pens and as an electrical conductor.
And speaking about conductors... The metal atoms in the metallic crystals are structured in such a way that some delocalized electrons give cohesion to the crystals and are responsible for their electrical properties.
Before ending this chapter let us introduce a few words about the so-called quasicrystals...
A quasicrystal is an "ordered" structure, but not perfectly periodic as the crystals are. The repeating patterns (sets of atoms, etc.) of the quasicrystalline materials can fill all available space continuously, but they do not display an exact repetition by translation. And, as far as symmetry is concerned, while crystals (according to the laws of classical crystallography) can display axes of rotation of order 2, 3, 4 and 6 only, the quasicrystals show other rotational symmetry axes, as for example of order 10
In this website we will not pay attention to the case of quasicrystals. Therefore, if you are interested on it, please go to this link, where Steffen Weber, in a relatively simple way, describes these types of materials from the theoretical point of view, and where some additional sources of information can also be found..Advanced readers should also consult the site offered by Paul J, Steinhardt at the University of Princeton.
The Nobel Prize in Chemistry 2011 was awarded to Daniel Shechtman by the discovery of quasicrystals in 1984..
There are obviously many questions that the reader will ask, having come this far, and one of the most obvious ones is: how do we know the structure of crystals? This question, and others, will be answered in following chapters and therefore we encourage you to consult them...