Luminol
- Page ID
- 3122
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)The combination of a luminescing solution with an oxidizing solution will generate light instead of heat through a chemical reaction. This is different from fluorescence and phosphorescence in that the energy in the emitted photons originates from chemistry, not from a previous photoexcitation event.
Materials
- 1 Erlenmeyer flask to combine both solutions in
- LUMINESCING Solution
- 4.0 g sodium carbonate
- 0.2 g luminol
- 24.0 g sodium bicarbonate (baking soda)
- 0.5 g ammonium carbonate
- 0.4 g copper (II) sulfate pentahydrate
- 1 liter of distilled water
- OXIDIZING Solution
- 50 mL of 3% hydrogen peroxide diluted to 1 liter
Alternative Materials
- Chemiluminescent powder: 4 g potassium ferrocyanide (K4Fe(CN)6), 0.2 g luminol, 64 g Chlorox 2 powder - Mix the powders thoroughly but do not grind. Just add water. You could try various amounts of the powder with various amounts of water. (From Rhonda Reist, CHEM 13 NEWS, April 2002, p. 6.)
Directions
- Carefully measure out all the substances for the luminescing solution.
- Put the substances in the 1 liter bottle
- Make the oxidizing solution and put the solution in a 1 liter bottle.
- If possible, make the room as dark as possible.
- Pour equal amounts of each solution into the Erlenmeyer flask
- Observe the eerie blue light.
Explanation
The chemical reaction called chemiluminescence is one of the very few reactions where the energy produced through the reaction is given off as light instead of heat. This same reaction occurs in lightening bugs when the chemical luciferase, that is in the insect's lower abdomen, reacts with the oxygen in the air. This reaction is happens when I mixed the luminescing solution with the oxidizing solution. This reaction is also a lot like how the glowing in the glow sticks occurs.
Chemiluminescence
The oxidizing solution, water and 3% hydrogen peroxide, is mixed with the Cu+2 ion catalyst and then the blue light is produced because of this reaction with luminol. The luminol is oxidized by the hyrogen peroxide in a basic solution to an excited state. The excited state of the luminol than changes back to the stable state with the emission of light.
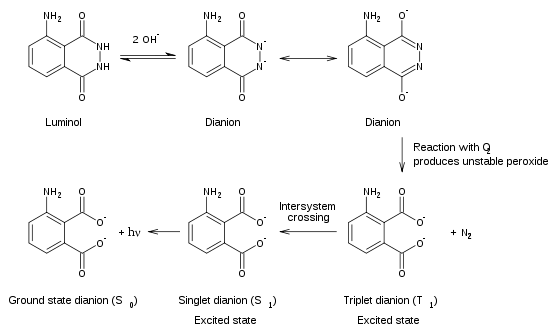
To exhibit its luminescence, the luminol must first be activated with an oxidant. Usually, a solution of hydrogen peroxide (H2O2) and a hydroxide salt in water is used as the activator. In the presence of a catalyst such as an iron compound, the hydrogen peroxide is decomposed to form oxygen and water:
2 H2O2 → O2 + 2 H2O
In a laboratory setting, the catalyst used is often potassium ferricyanide. In the forensic detection of blood, the catalyst is the iron present in hemoglobin. Enzymes in a variety of biological systems may also catalyze the decomposition of hydrogen peroxide.
When luminol reacts with the hydroxide salt, a dianion is formed. The oxygen produced from the hydrogen peroxide then reacts with the luminol dianion. The product of this reaction, an organic peroxide, is very unstable and immediately decomposes with the loss of nitrogen to produce 3-aminophthalic acid with electrons in an excited state. As the excited state relaxes to the ground state, the excess energy is liberated as a photon, visible as blue light.
Safety
- Ammonium carbonate has a very strong smell - carefully open in the hood
Waste Disposal
- All liquid materials may be put down the drain.
References
- Shakhashiri, Bassam, Chemical Demonstrations, Vol. 1, U. Wisconsin Press, 1983, p. 156.
- Summerlin, L, Ealy, J, Chemical Demonstrations, American Chemical Society, 1985, p. 138.
Contributors
- Charles Ophardt, Professor Emeritus, Elmhurst College Virtual Chembook
- http://en.Wikipedia.org/wiki/Luminol

