The Manufacture of Alcohols
- Page ID
- 3900
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)This page looks at the manufacture of alcohols by the direct hydration of alkenes, concentrating mainly on the hydration of ethene to make ethanol. It then compares that method with making ethanol by fermentation.
Manufacturing alcohols from alkenes
Ethanol is manufactured by reacting ethene with steam. The catalyst used is solid silicon dioxide coated with phosphoric(V) acid. The reaction is reversible.

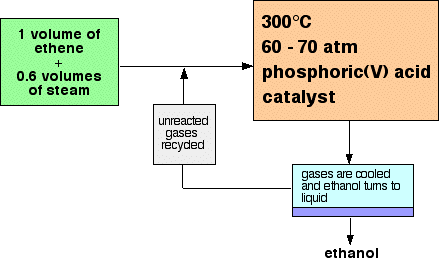
Only 5% of the ethene is converted into ethanol at each pass through the reactor. By removing the ethanol from the equilibrium mixture and recycling the ethene, it is possible to achieve an overall 95% conversion. A flow scheme for the reaction looks like this:
The manufacture of other alcohols from alkenes
Some - but not all - other alcohols can be made by similar reactions. The catalyst used and the reaction conditions will vary from alcohol to alcohol. The reason that there is a problem with some alcohols is well illustrated with trying to make an alcohol from propene, CH3CH=CH2. In principle, there are two different alcohols which might be formed:
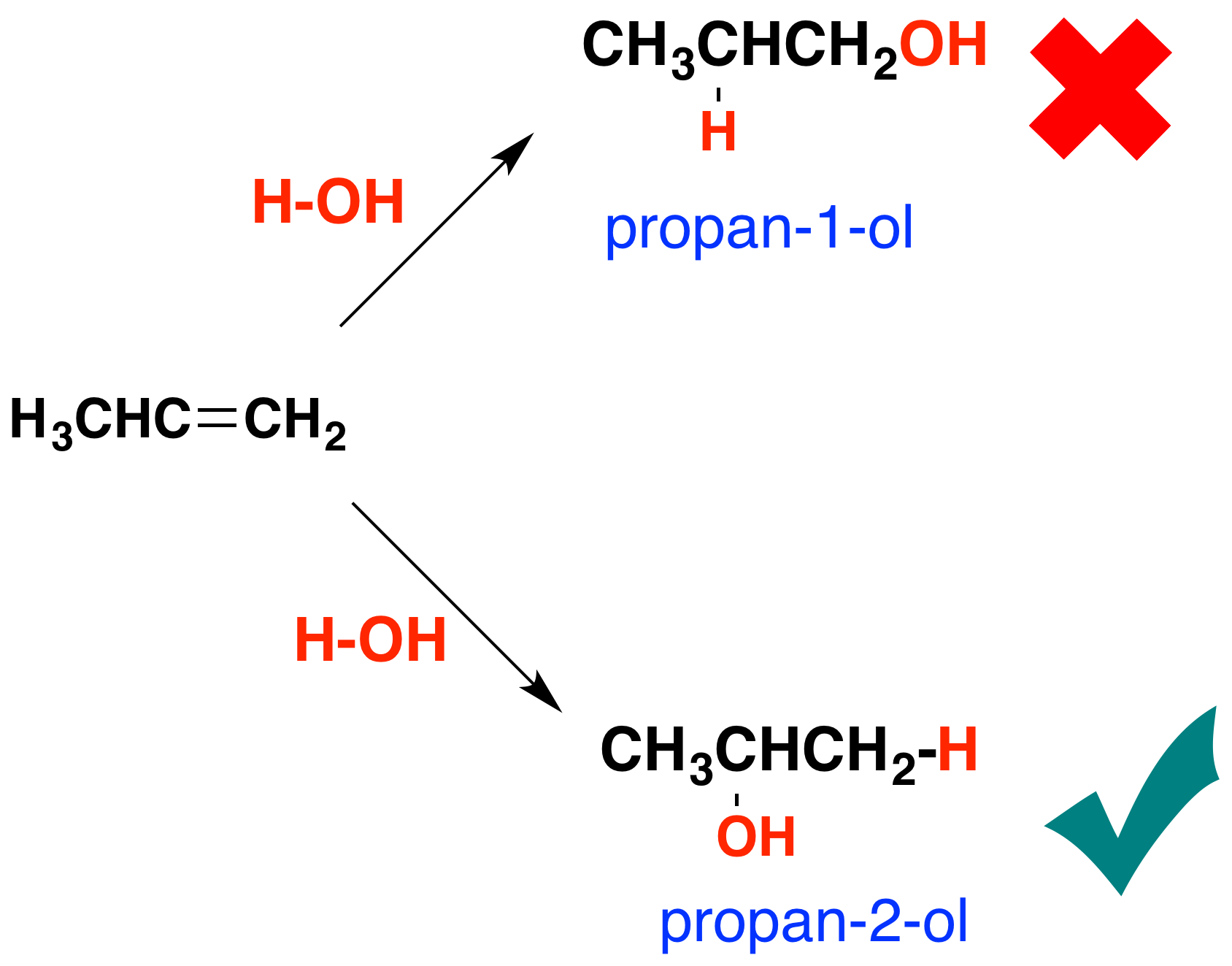
You might expect to get either propan-1-ol or propan-2-ol depending on which way around the water adds to the double bond. In practice what you get is propan-2-ol. If you add a molecule H-X across a carbon-carbon double bond, the hydrogen nearly always gets attached to the carbon with the most hydrogens on it already - in this case the CH2 rather than the CH. The effect of this is that there are bound to be some alcohols which it is impossible to make by reacting alkenes with steam because the addition would be the wrong way around.
Making ethanol by fermentation
This method only applies to ethanol and you cannot make any other alcohol this way. The starting material for the process varies widely, but will normally be some form of starchy plant material such as maize (US: corn), wheat, barley or potatoes. Starch is a complex carbohydrate, and other carbohydrates can also be used - for example, in the lab sucrose (sugar) is normally used to produce ethanol. Industrially, this wouldn't make sense. It would be silly to refine sugar if all you were going to use it for was fermentation. There is no reason why you should not start from the original sugar cane, though.
The first step is to break complex carbohydrates into simpler ones. For example, if you were starting from starch in grains like wheat or barley, the grain is heated with hot water to extract the starch and then warmed with malt. Malt is germinated barley which contains enzymes which break the starch into a simpler carbohydrate called maltose, \(C_{12}H_{22}O_{11}\). Maltose has the same molecular formula as sucrose but contains two glucose units joined together, whereas sucrose contains one glucose and one fructose unit.
Yeast is then added and the mixture is kept warm (say 35°C) for perhaps several days until fermentation is complete. Air is kept out of the mixture to prevent oxidation of the ethanol produced to ethanoic acid (vinegar). Enzymes in the yeast first convert carbohydrates like maltose or sucrose into even simpler ones like glucose and fructose, both \(C_6H_{12}O_6\), and then convert these in turn into ethanol and carbon dioxide. You can show these changes as simple chemical equations, but the biochemistry of the reactions is much, much more complicated than this suggests.
\[ C_{12}H_{22}O_{11} + H_2O \longrightarrow 2C_6H_{12}O_6 \]
\[ C_6H_{12}O_6 \longrightarrow 2CH_3CH_2OH + 2CO2\]
Yeast is killed by ethanol concentrations in excess of about 15%, and that limits the purity of the ethanol that can be produced. The ethanol is separated from the mixture by fractional distillation to give 96% pure ethanol. For theoretical reasons (minimum boiling point azeotrope), it is impossible to remove the last 4% of water by fractional distillation.
| Fermentation | Hydration of ethene | |
|---|---|---|
| Type of process | A batch process. Everything is put into a container and then left until fermentation is complete. That batch is then cleared out and a new reaction set up. This is inefficient. | A continuous flow process. A stream of reactants is passed continuously over a catalyst. This is a more efficient way of doing things. |
| Rate of reaction | Very slow. | Very rapid. |
| Quality of product | Produces very impure ethanol which needs further processing | Produces much purer ethanol. |
| Reaction conditions | Uses gentle temperatures and atmospheric pressure. | Uses high temperatures and pressures, needing lots of energy input. |
| Use of resources | Uses renewable resources based on plant material. | Uses finite resources based on crude oil. |
Contributors
Jim Clark (Chemguide.co.uk)


