Chapter 16 Solutions
- Page ID
- 1114
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)In-chapter exercises
E16.1: An aldol condensation is not a redox reaction. There is no transfer of electrons from one species to another, either in the form of a hydrogenation/dehydrogenation step or in terms of an insertion of an oxygen atom (or other electronegative atom).
E16.2: Epoxidation reactions are redox transformations: both carbon atoms lose a bond to carbon and gain a bond to oxygen. The cyclase reaction, however, is not redox.
E16.3: This is a transformation that is thermodynamically favorable, but the kinetics are very slow. The activation energy, in other words, is too high for the transformation to occur uncatalyzed at room temperature at a rate that is noticeable.
E16.4: The anomeric carbon (in other words, the carbon that is the center for the hemiacetal) has two bonds to oxygen – thus it is in the carbonyl oxidation state. This becomes clear when we look at the straight-chain form of glucose, in which this carbon is an aldehyde.
E16.5: Saturated fat is in a more highly reduced state, and therefore contains more electrons to transfer to oxygen and more energy to release when oxidized to CO2 during respiration.
E16.6: In grape fermentation, glucose is converted to two equivalents of ethanol and one of carbon dioxide (this is the bubbling that is observed). When wine turns to vinegar, the ethanol is being oxidized to acetic acid. Grape juice (full of sugar) contains the most calories, and vinegar the least.
E16.7:
a) For every molecule of substrate that is oxidized, one molecule of NADP+ is reduced to NADPH. We can determine the concentration of NADPH formed by using its extinction coefficient, e = 6290 M-1 cm-1.
So the amount of substrate oxidized is (1.53 x 10-5 M)(1 x 10-3 L) = 1.53 x 10-8 moles.
b) (100 x 10-6 M) – (1.53 x 10-5 M) = 8.47 x 10-5 M = 84.7 mM.
c)
E16.8:
E16.9:
a)
b)
End-of-chapter problems
P16.1:
b) The hydride adds to the front of pyruvate as drawn in the figure. This is the re face.
P16.2:
a)
b)
P16.3:
P16.4:
a)
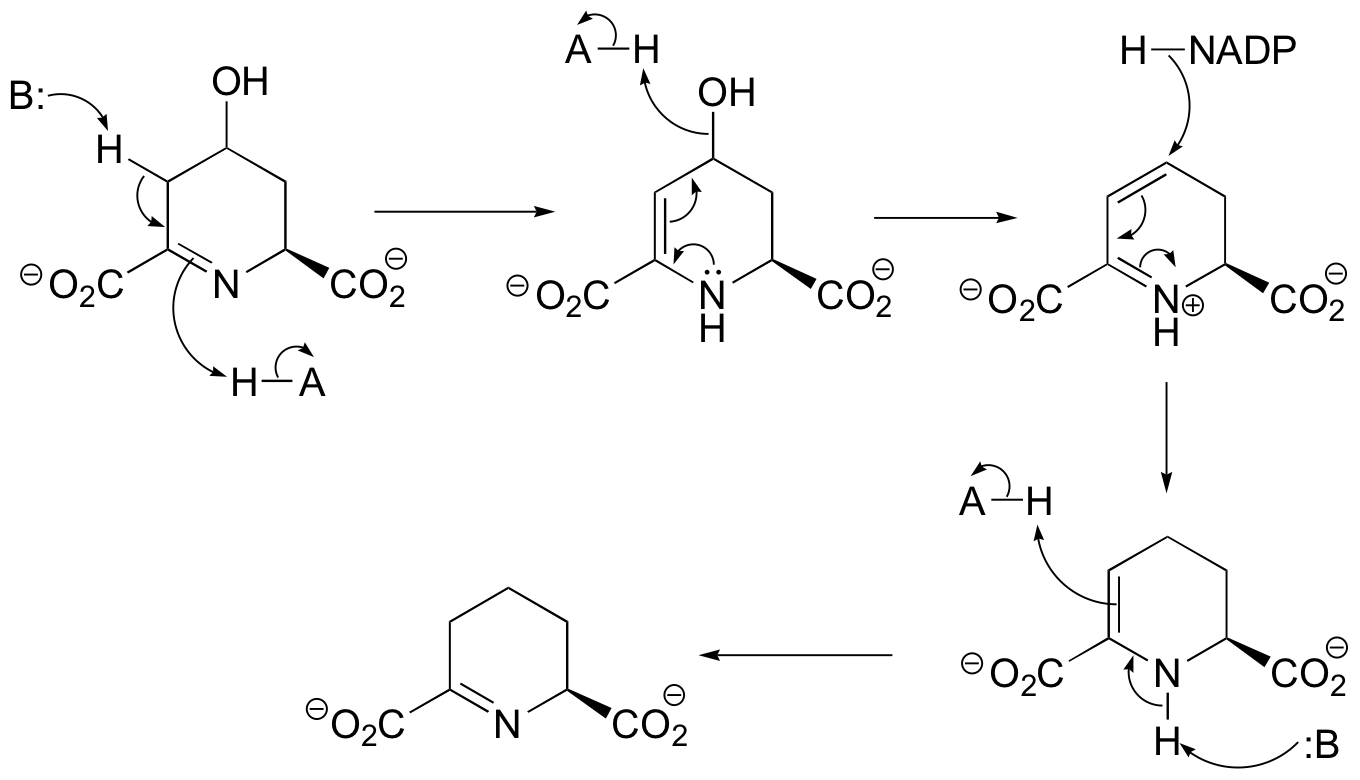
b)
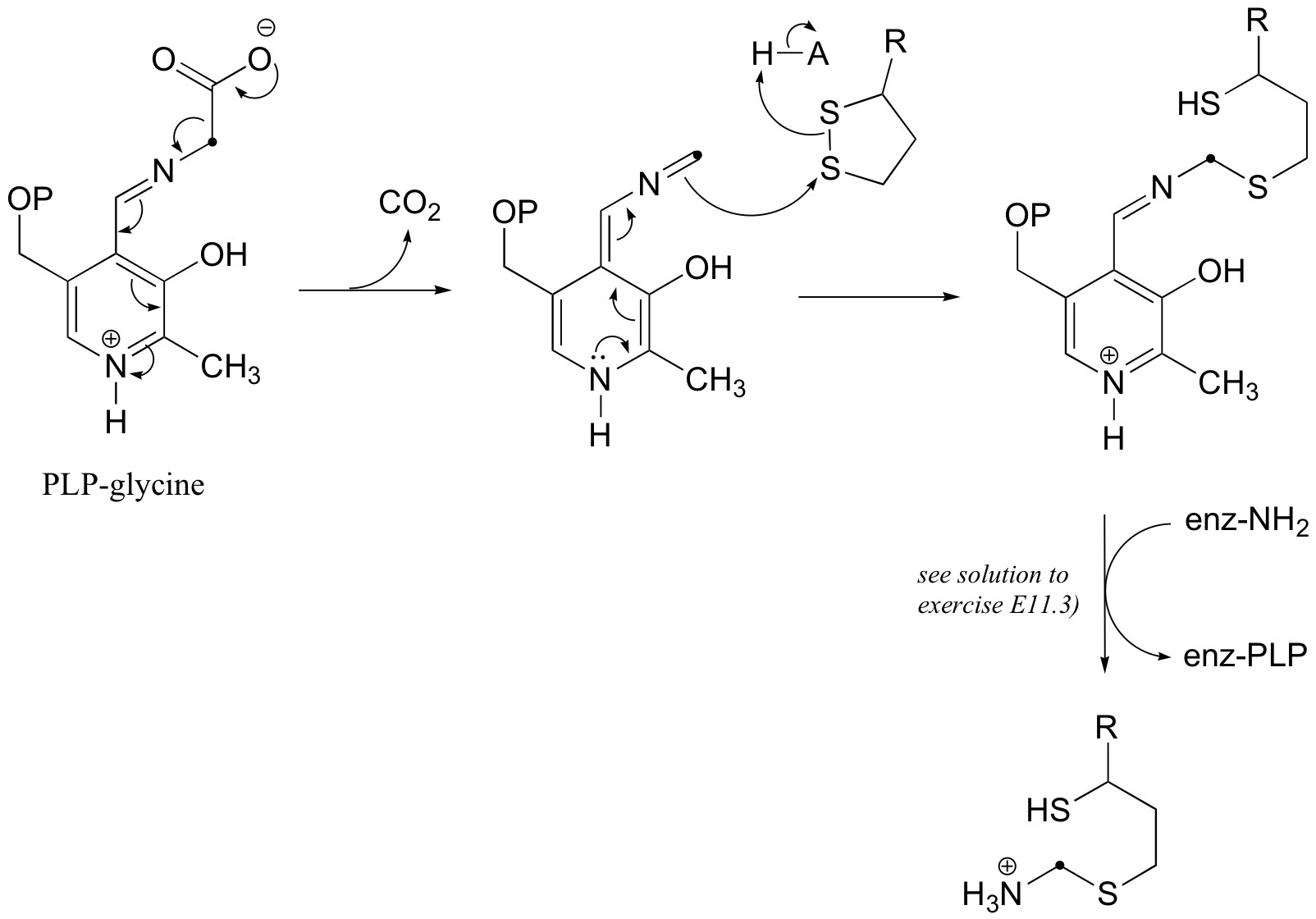
(The last step above is simply a Schiff base transfer to the lysine nirtrogen of the enzyme - an example of this mechanism is drawn out in the solution to exercise E11.3. You were asked to show the mechanism for the release of free ammonia and transfer of the alpha-carbon of glycine to tetrahydrofolate in challenge problem C11.5, and the solution is shown in J. Biol. Chem. 2010, 285, 18684, fig 5. Regeneration of lipoamide from dihydrolipoamide is addressed in section 16.12.)
P16.5:
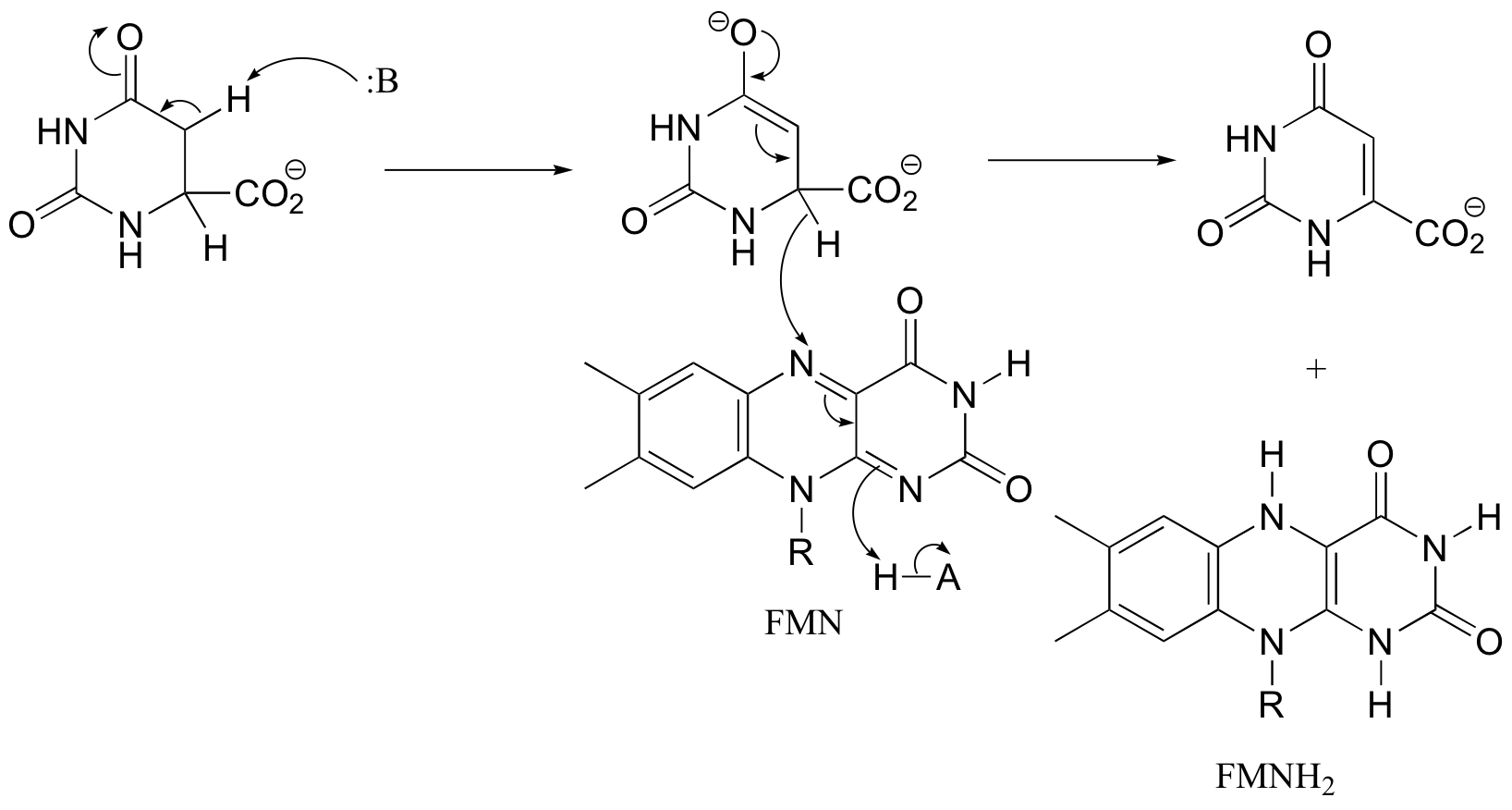
P16.6:
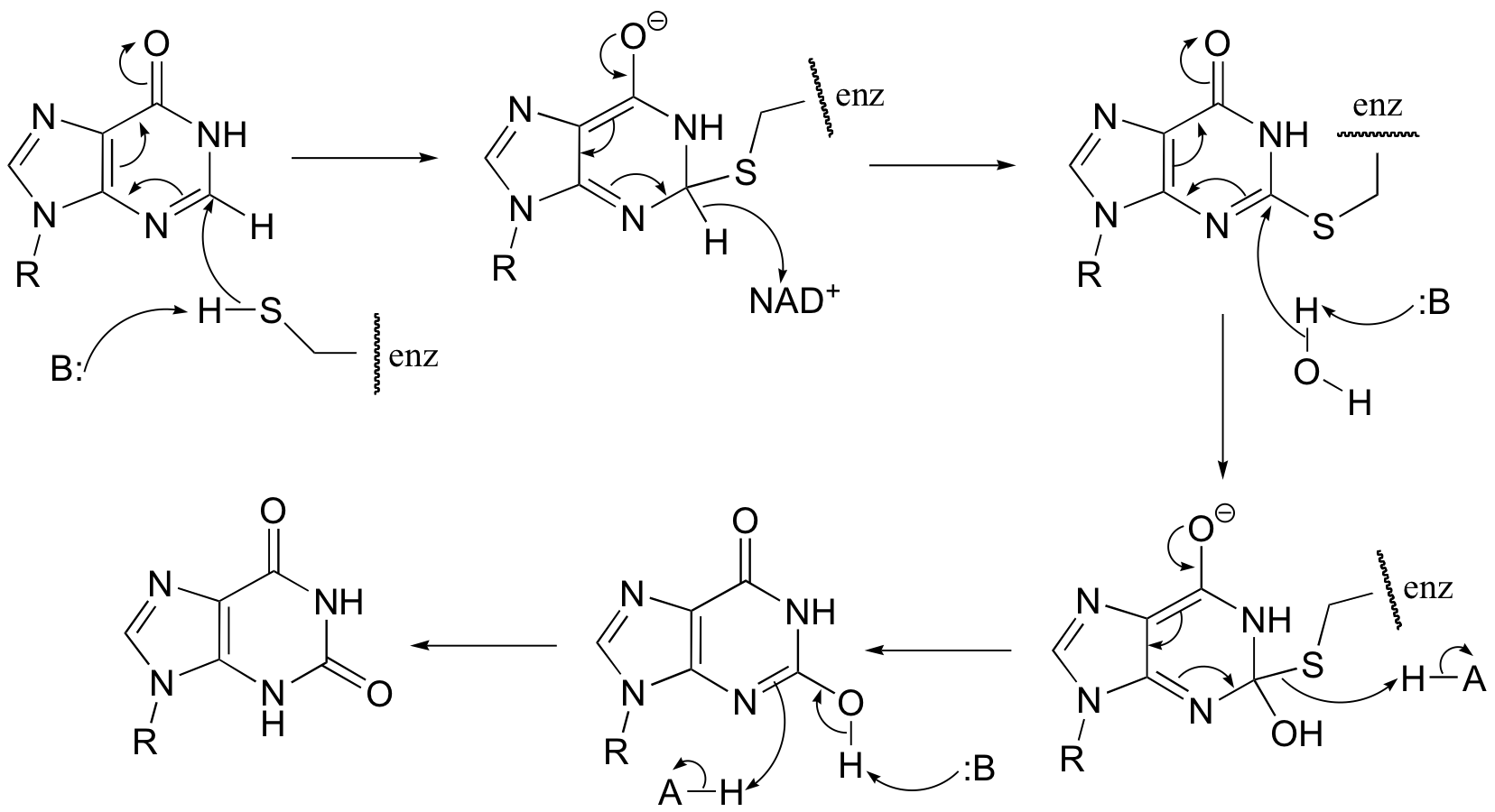
P16.7:
a) First an imine (Schiff base) linkage forms, then the imine is reduced to an amine by NADPH.
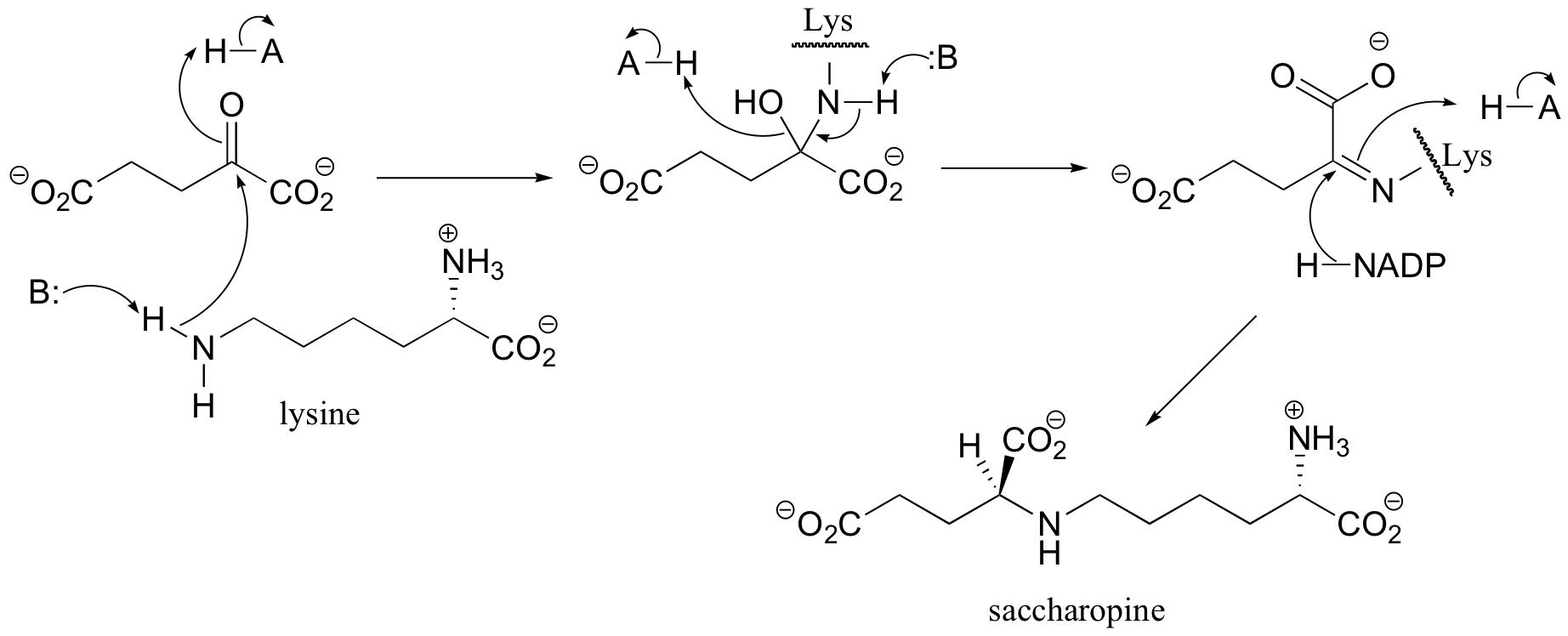
b) The amine formed in the previous reaction (part a) is oxidized to an imine (not the reverse of the reduction step in part a – it occurs on the other side of the molecule!). Hydrolysis of the imine results in the products.
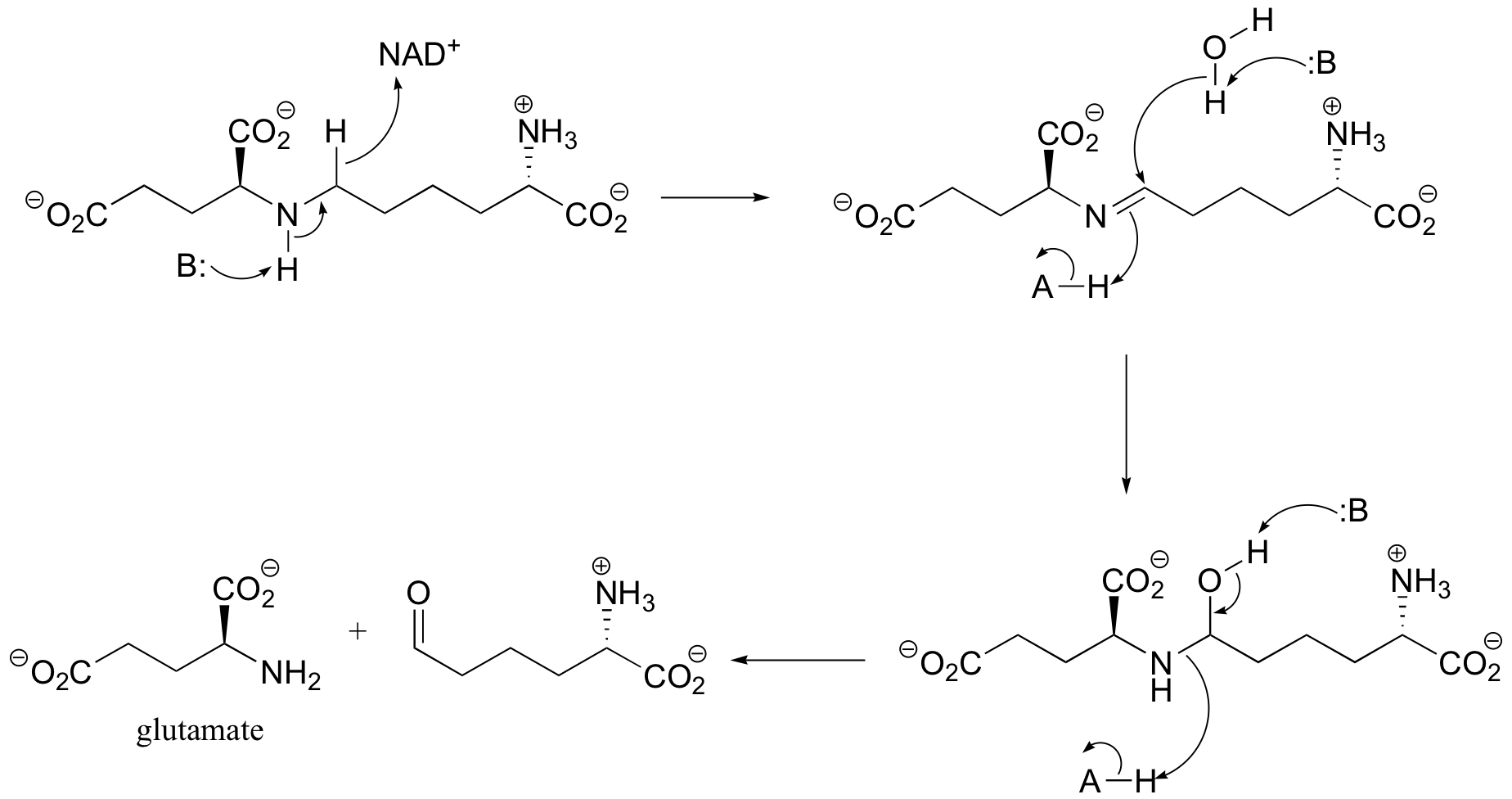
P16.8: This reaction is analogous to that catalyzed by the pyruvate dehydrogenase complex (section 16.12B). The additional cofactors are thiamine diphosphate, lipoamide, and FAD.
P16.9:
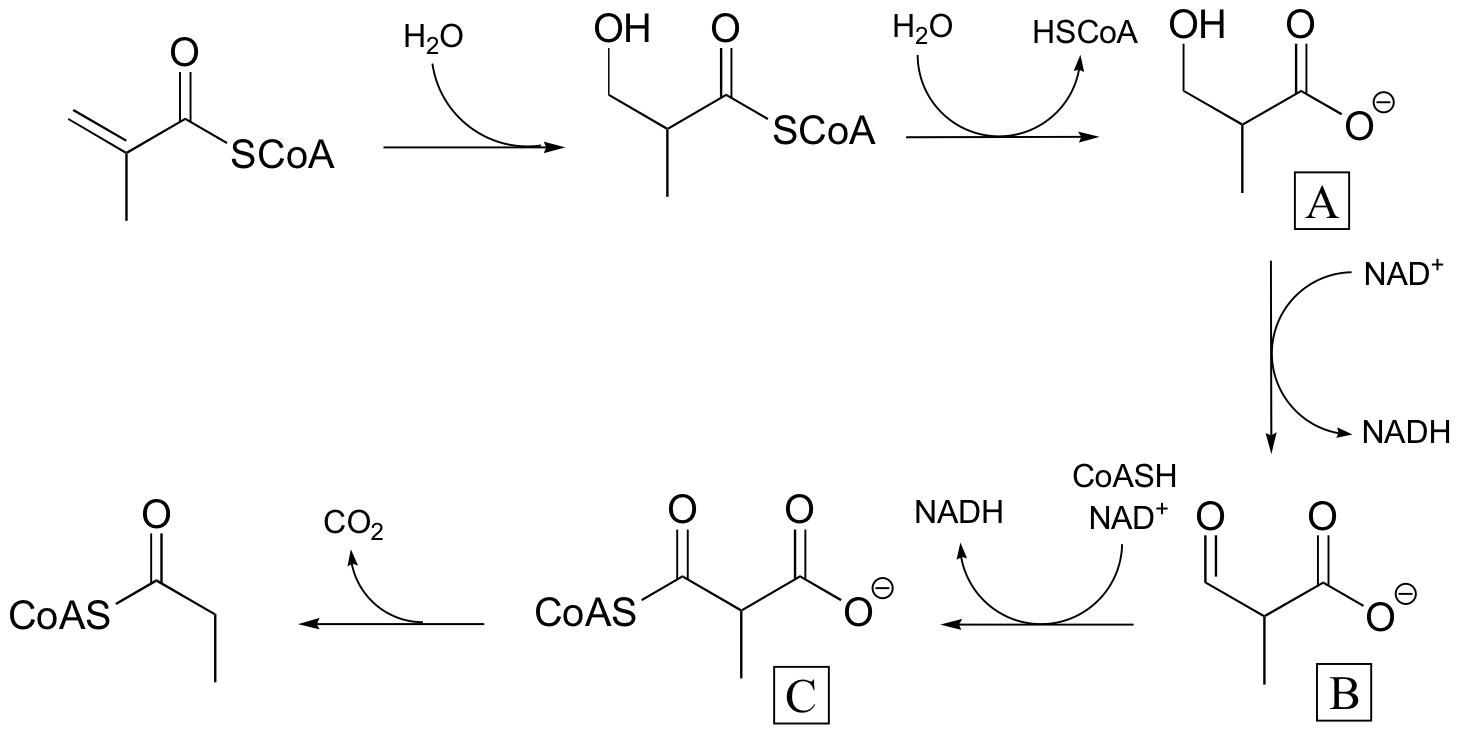
P16.10: See Biochemistry 39, 6732 for the original report on this experiment.
a)
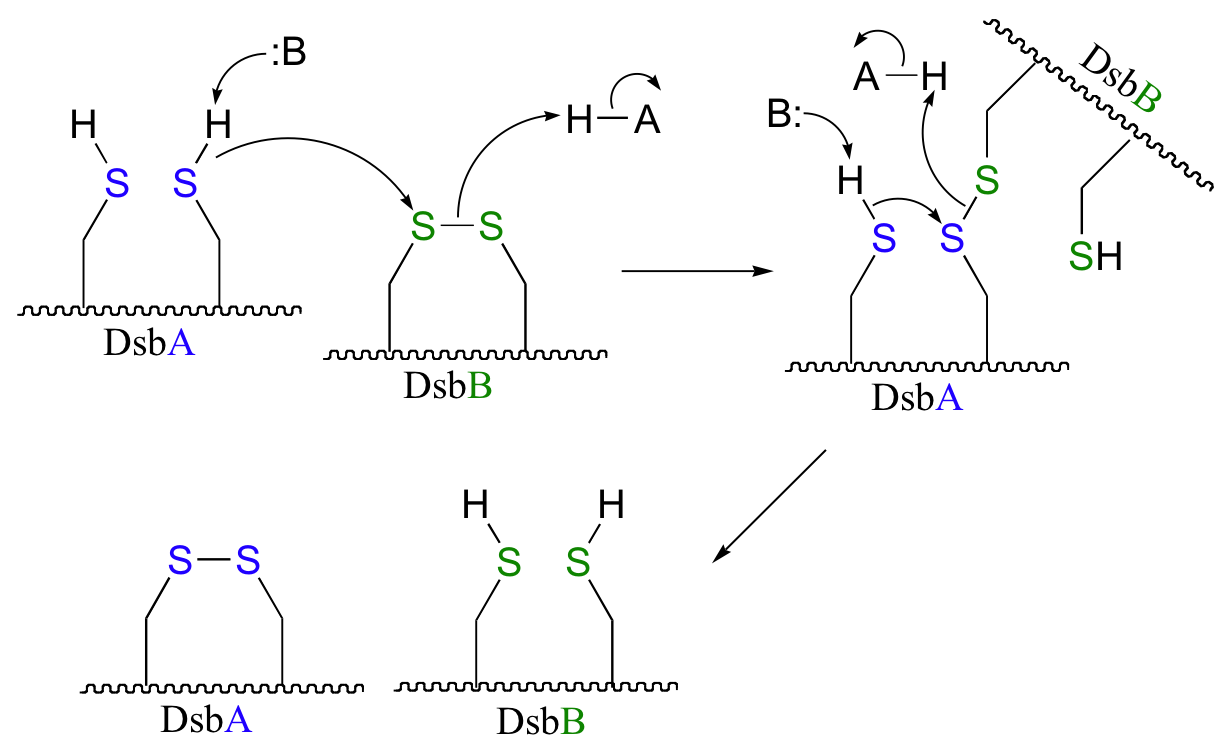
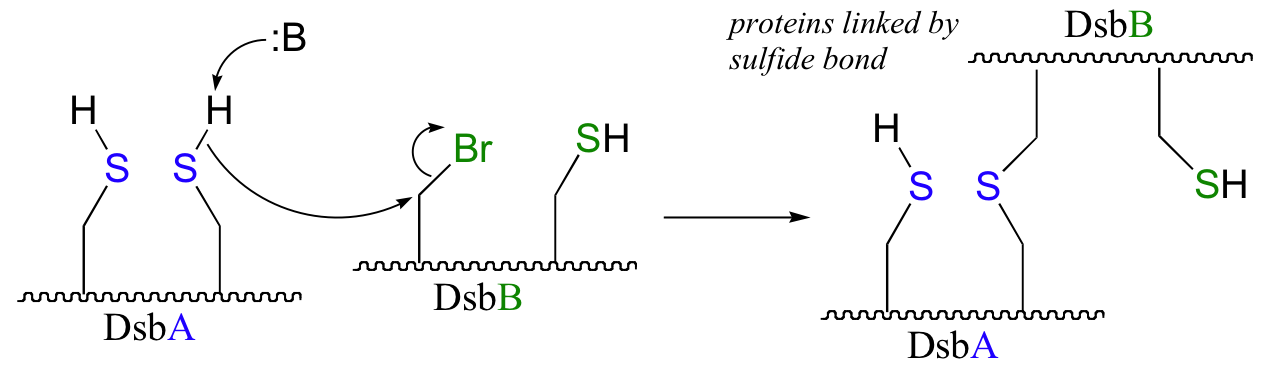
b) The bromoalanine side chain prevents formation of the key disulfide bond in DsbB, and provides an alternative carbon electrophile for the cysteine in DsbA to attack. The result of this SN2 displacement is a stable sulfide linkage beetween the two proteins, and isolation of this species provides evidence for the existance of the unstable disulfide-linked DsbA-DsbB intermediate in the normal reaction.
P16.11This reaction is an electrophilic aromatic substitution, with the flavin peroxide oxygen as the electrophile. The substitution takes place ortho to the electron-donating, ortho/para-directing exocyclic amine, and meta to the electron-withdrawing, meta-directing carbonyl.
P16.12: (See also J. Am. Chem. Soc. 2004, 126, 15060; J. Nat. Prod. 2001, 64, 597).
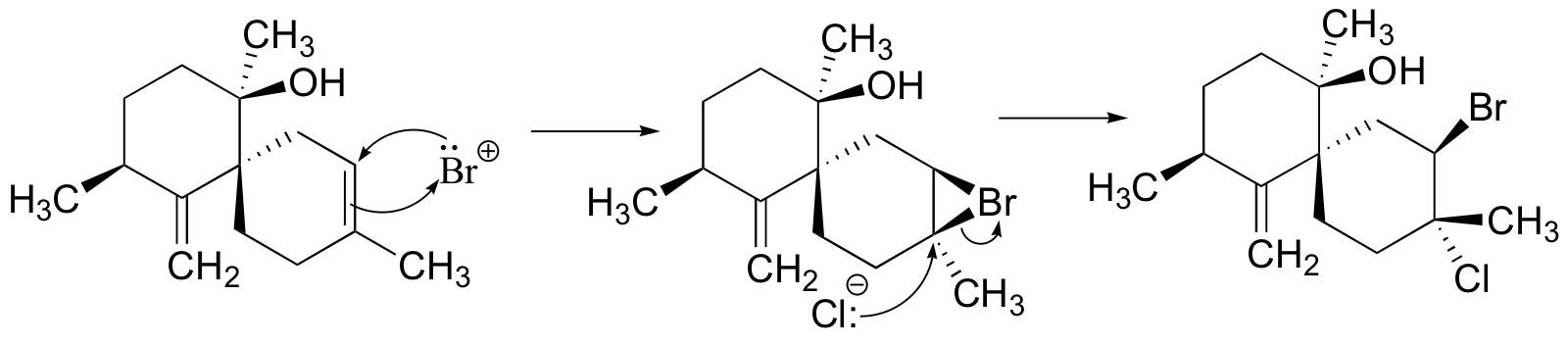
P16.13:
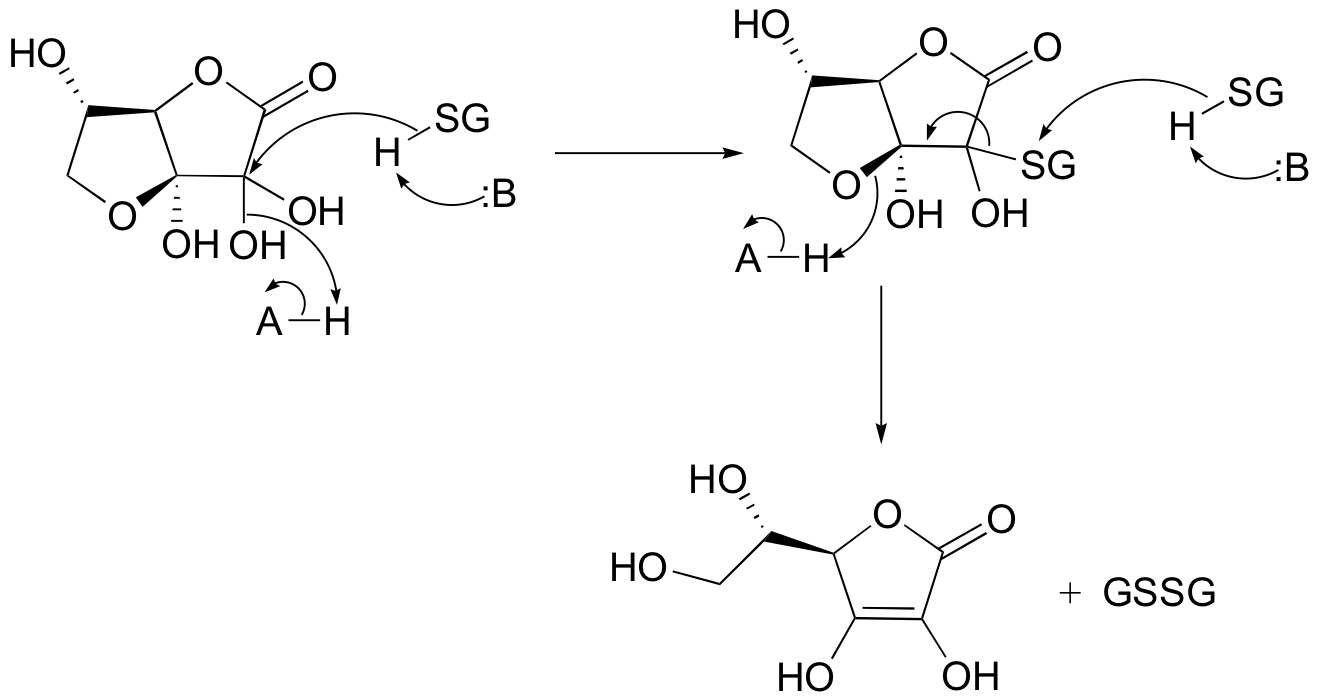
P16.14:
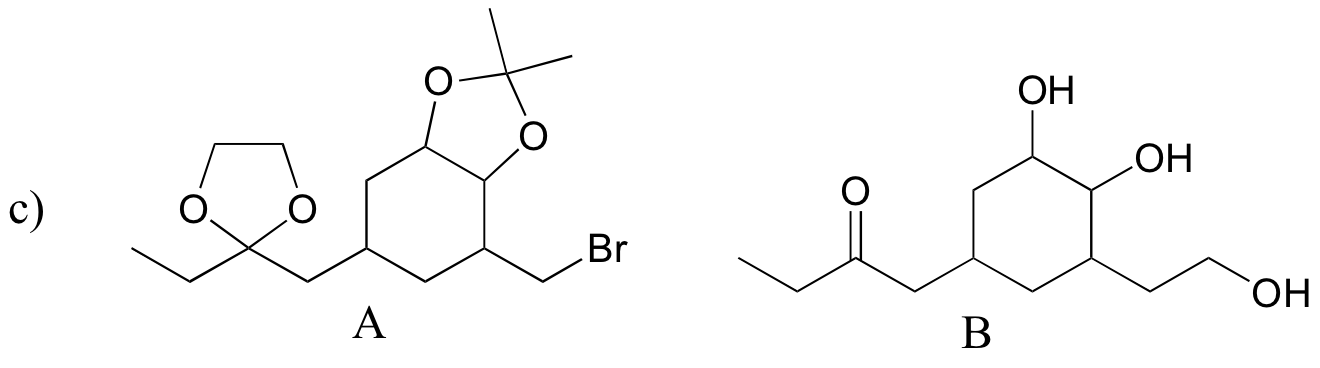
d)
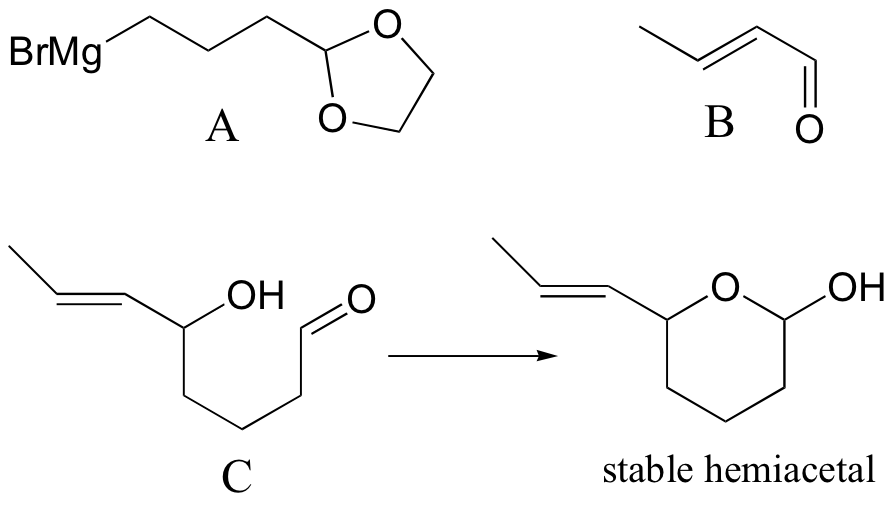
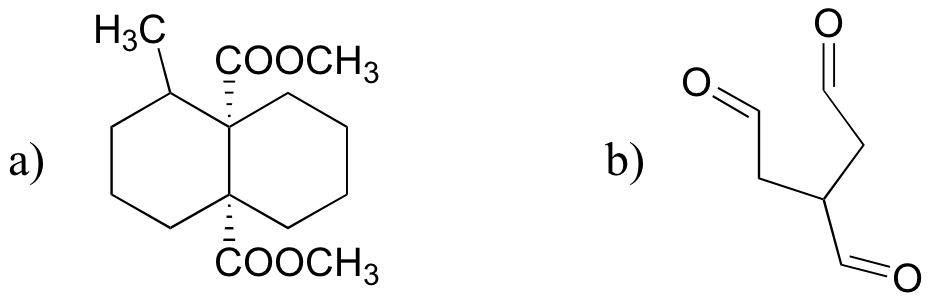
P16.15:
a)
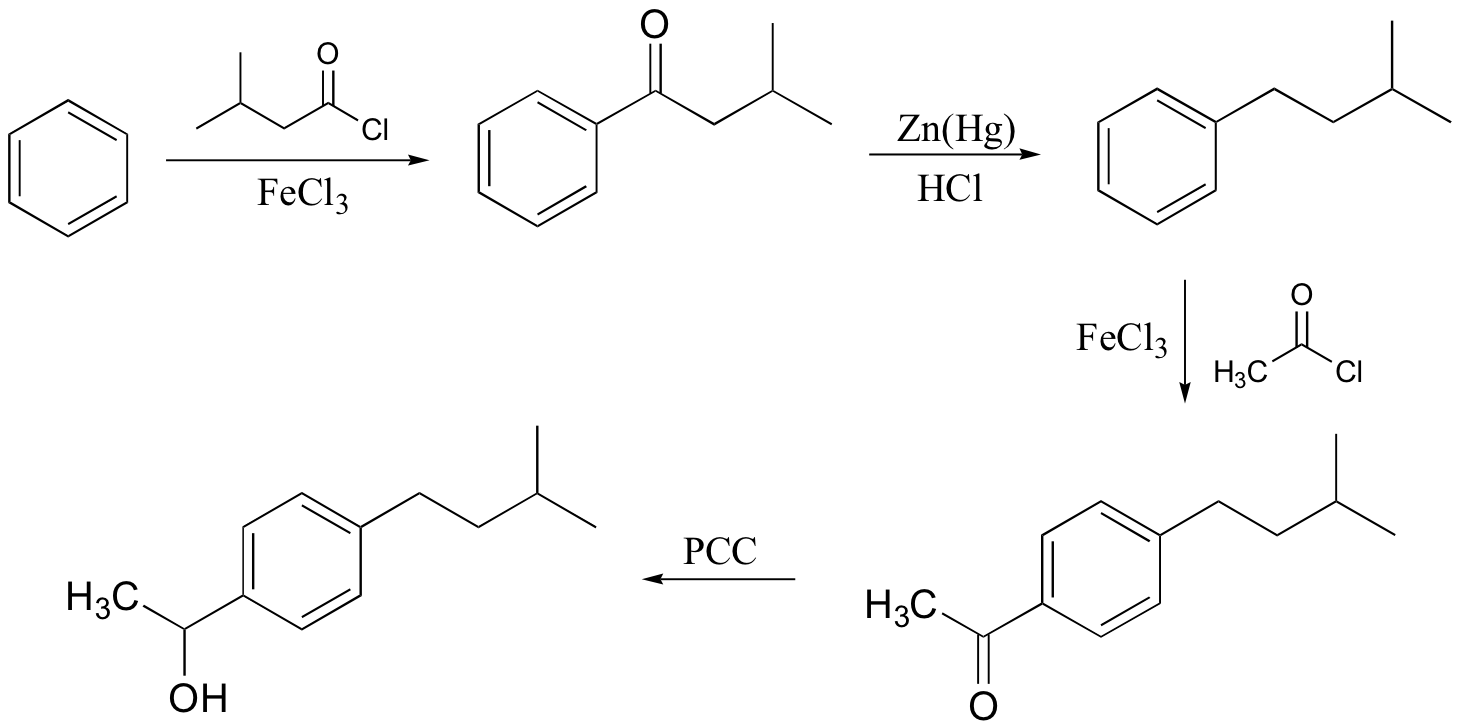
b)
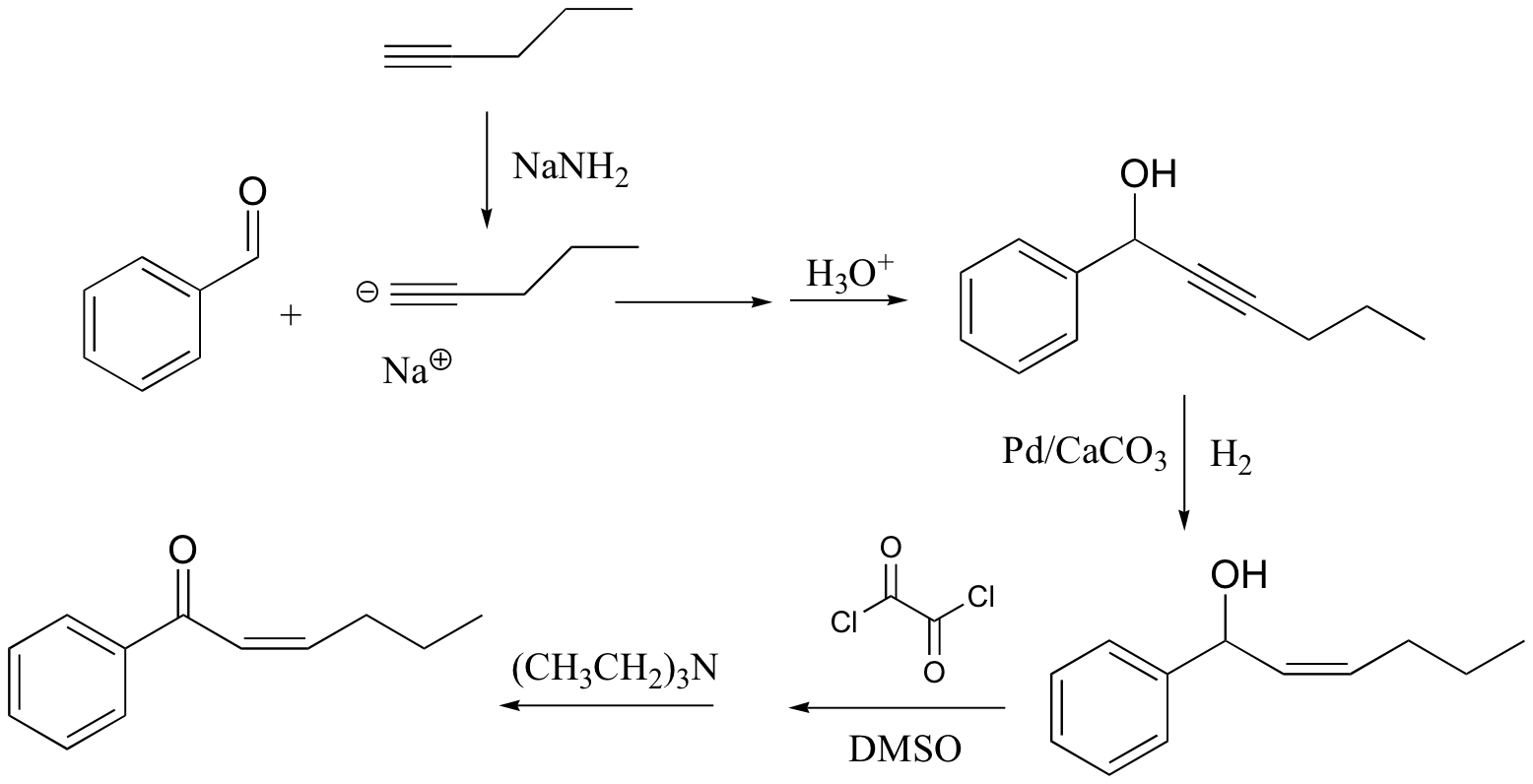
c)
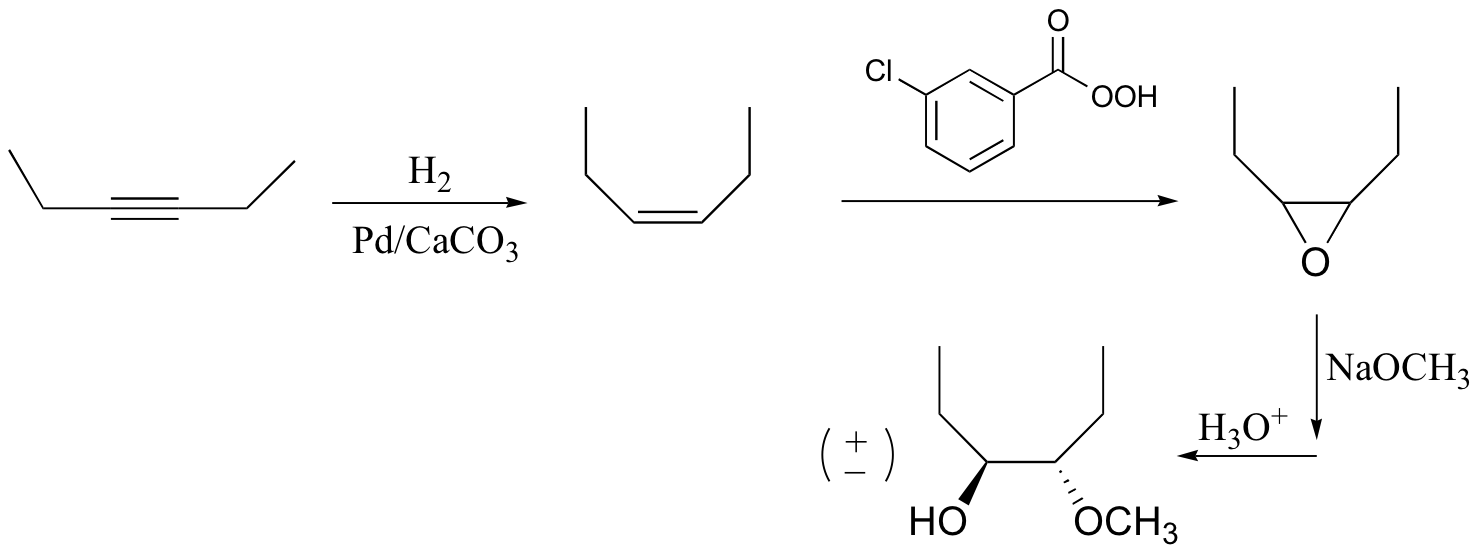
P16.16:
a)
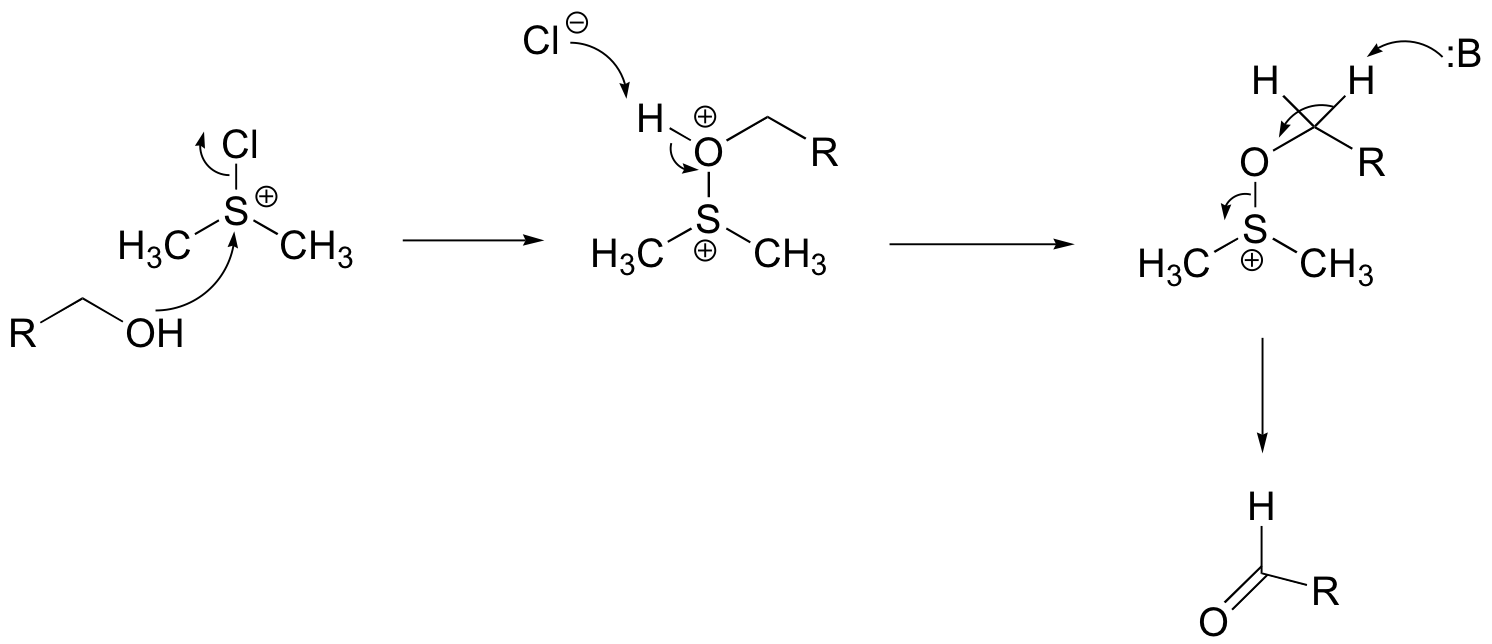
b)
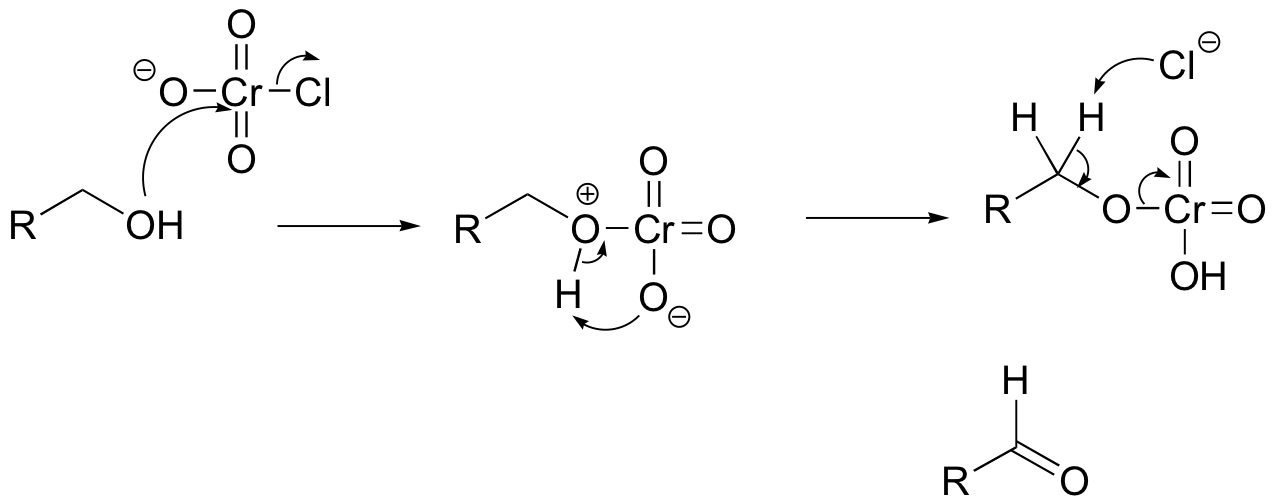
Challenge problems
C16.1: See J. Mol. Biol. 2004, 342, 183, fig 7. Note that the electron accepting-donating mechanism for NAD+ is the same as what we have seen in redox reactions throughout this chapter, except that the electrons being accepted (and then given back) come from a carbon-carbon pi bond rather than a hydride ion.


