8.1: The Schrödinger Equation
- Page ID
- 4527
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
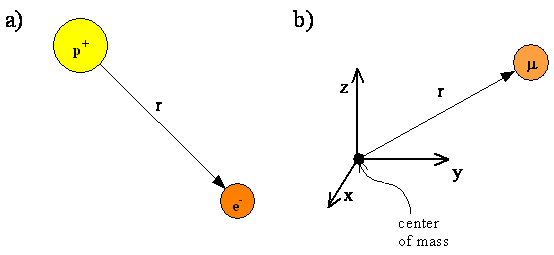
The hydrogen atom, consisting of an electron and a proton, is a two-particle system, and the internal motion of two particles around their center of mass is equivalent to the motion of a single particle with a reduced mass. This reduced particle is located at \(r\), where \(r\) is the vector specifying the position of the electron relative to the position of the proton. The length of \(r\) is the distance between the proton and the electron, and the direction of \(r\) and the direction of \(r\) is given by the orientation of the vector pointing from the proton to the electron. Since the proton is much more massive than the electron, we will assume throughout this chapter that the reduced mass equals the electron mass and the proton is located at the center of mass.
Exercise \(\PageIndex{1}\)
- Assuming the Bohr radius gives the distance between the proton and electron, calculate the distance of the proton from the center of mass, and calculate the distance of the electron from the center of mass.
- Calculate the reduced mass of the electron-proton system.
- In view of your calculations in (a) and (b), comment on the validity of a model in which the proton is located at the center of mass and the reduced mass equals the electron mass.
Since the internal motion of any two-particle system can be represented by the motion of a single particle with a reduced mass, the description of the hydrogen atom has much in common with the description of a diatomic molecule that we considered in Chapter 7. The Schrödinger Equation for the hydrogen atom
\[ \hat {H} (r , \theta , \varphi ) \psi (r , \theta , \varphi ) = E \psi ( r , \theta , \varphi) \label {8-1}\]
employs the same kinetic energy operator, \(\hat {T}\), written in spherical coordinates as developed in Chapter 7. For the hydrogen atom, however, the distance, r, between the two particles can vary, unlike the diatomic molecule where the bond length was fixed, and the rigid rotor model was used. The hydrogen atom Hamiltonian also contains a potential energy term, \(\hat {V}\), to describe the attraction between the proton and the electron. This term is the Coulomb potential energy,
\[ \hat {V} (r) = - \dfrac {e^2}{4 \pi \epsilon _0 r } \label {8-2}\]
where r is the distance between the electron and the proton. The Coulomb potential energy depends inversely on the distance between the electron and the nucleus and does not depend on any angles. Such a potential is called a central potential.
The full expression for \(\hat {H}\) in spherical coordinates is
\[\hat {H} (r , \theta , \varphi ) = - \dfrac {\hbar ^2}{2 \mu r^2} \left [ \dfrac {\partial}{\partial r} \left (r^2 \dfrac {\partial}{\partial r} \right ) + \dfrac {1}{\sin \theta } \dfrac {\partial}{\partial \theta } \left ( \sin \theta \dfrac {\partial}{\partial \theta} \right ) + \dfrac {1}{\sin ^2 \theta} \dfrac {\partial ^2}{\partial \varphi ^2} \right ] - \dfrac {e^2}{4 \pi \epsilon _0 r } \label {8-3}\]
The contributions from rotational and radial components of the motion become clearer if we write out the complete Schrödinger equation,
\[ \left \{ -\dfrac {\hbar ^2}{2 \mu r^2} \left [ \dfrac {\partial}{\partial r} \left (r^2 \dfrac {\partial}{\partial r} \right ) + \dfrac {1}{\sin \theta } \dfrac {\partial}{\partial \theta } \left ( \sin \theta \dfrac {\partial}{\partial \theta} \right ) + \dfrac {1}{\sin ^2 \theta} \dfrac {\partial ^2}{\partial \varphi ^2} \right ] - \dfrac {e^2}{4 \pi \epsilon _0 r } \right \} \psi (r , \theta , \varphi ) = E \psi (r , \theta , \varphi ) \label {8-4}\]
multiply both sides of Equation \ref{8-4} by \(2 \mu r \), and rearrange to obtain
\[\hbar ^2 \dfrac {\partial}{\partial r} \left ( r^2 \dfrac {\partial}{\partial r} \psi (r , \theta , \varphi ) \right ) + 2 \mu r^2 \left [ E + \dfrac {e^2}{4 \pi \epsilon _0 r } \right ] \psi (r , \theta , \varphi ) = \]
\[ - \hbar^2 \left [ \dfrac {1}{\sin \theta } \dfrac {\partial}{\partial \theta } \left ( \sin \theta \dfrac {\partial}{\partial \theta} \right ) + \dfrac {1}{\sin ^2 \theta} \dfrac {\partial ^2}{\partial \varphi ^2} \right ] \psi (r , \theta , \varphi ) \label {8-5}\]
Manipulating the Schrödinger equation in this way helps us recognize the square of the angular momentum operator in Equation \ref{8-5}. The square of the angular momentum operator, which was defined in Chapter 7, is repeated here as Equation \ref{8-6}.
\[\hat {M} ^2 = -\hbar ^2 \left [\dfrac {1}{\sin \theta } \dfrac {\partial}{\partial \theta } \left ( \sin \theta \dfrac {\partial}{\partial \theta} \right ) + \dfrac {1}{\sin ^2 \theta} \dfrac {\partial ^2}{\partial \varphi ^2} \right ] \label {8-6}\]
Substituting Equation \ref{8-6} into Equation \ref{8-5} produces
\[\hbar ^2 \dfrac {\partial}{\partial r } \left ( r^2 \dfrac {\partial}{\partial r} \psi (r , \theta , \varphi ) \right ) + 2 \mu r^2 [ E - \hat {V} ] \psi (r , \theta , \varphi ) = \hat {M} ^2 \psi (r, \theta , \varphi ) \label {8-7}\]
Exercise \(\PageIndex{1}\)
Show the algebraic steps going from Equation \ref{8-4} to Equation \ref{8-5} and finally to Equation \ref{8-7}. Justify the statement that the rotational and radial motion are separated in Equation \ref{8-7}.
Since the angular momentum operator does not involve the radial variable, \(r\), we can separate variables in Equation \ref{8-7} by using a product wavefunction, as we did previously in Chapter 7. From our work on the rigid rotor, Chapter 7, we know that the eigenfunctions of the angular momentum operator are the Spherical Harmonic functions, \(Y (\theta , \varphi )\), so a good choice for a product function is
\[ \psi (r , \theta , \varphi ) = R (r) Y (\theta , \varphi ) \label {8-8}\]
The Spherical Harmonic functions table provide information about where the electron is around the proton, and the radial function R(r) describes how far the electron is away from the proton.
To separate variables, substitute the product function, Equation \ref{8-8}, into Equation \ref{8-7}, evaluate partial derivatives, divide each side by R(r) \(Y (\theta, \varphi ) \), and set each side of that resulting equation equal to a constant \(\lambda\).
\[ \dfrac {\hbar ^2}{R (r)} \dfrac {\partial}{\partial r} r^2 \dfrac {\partial}{\partial r} R(r) + \dfrac {2 \mu r^2}{R (r)} [ E - V ] R (r) = \lambda \label {8-9}\]
\[ \dfrac {1}{Y (\theta , \varphi )} \hat {M} ^2 Y (\theta , \varphi ) = \lambda \label {8-10}\]
Equations \ref{8-9} and \ref{8-10} represent the radial differential equation and the angular differential equation, respectively. As we describe below, they are solved separately to give the \(Y (\theta , \varphi )\) angular functions and the \(R(r)\) radial functions.
Exercise \(\PageIndex{3}\)
Complete the steps leading from Equation \ref{8-7} to Equation \ref{8-9} and Equation \ref{8-10}.
Rearranging Equation \ref{8-10} yields
\[\hat {M} ^2 Y^{m_l}_l (\theta , \varphi ) = \lambda Y^{m_l}_l (\theta , \varphi ) \label {8-11}\]
where we have added the indices \(l\) and \(m_l\) to identify a particular spherical harmonic function. Note that the notation has changed from that used in Chapter 7. It is customary to use \(J\) and \(m_J\) to represent the angular momentum quantum numbers for rotational states, but for electronic states, it is customary to use \(l\) and \(m_l\) to represent the same thing. Further, the electronic angular momentum is designated by L and the corresponding operator is called \(\hat {L}\). In complete electronic notation, Equation \ref{8-11} is
\[ \hat {L} ^2 Y^{m_l}_l (\theta , \varphi ) = \lambda Y^{m_l}_l (\theta , \varphi ) \label {8-12}\]
Equation \ref{8-12} says that \(Y^{m_l}_l (\theta , \varphi )\) must be an eigenfunction of the angular momentum operator \(\hat {L} ^2\) with eigenvalue \(λ\). We know from the discussion of the Rigid Rotor that the eigenvalue λ is \(J(J+1)ħ^2\), or in electronic notation, \(l (l + 1) \hbar ^2\). Consequently, Equation \ref{8-12} becomes
\[ \hat {L} ^2 Y^{m_l}_l (\theta , \varphi ) = l (l + 1) \hbar ^2 Y^{m_l}_l (\theta , \varphi ) \label {8-13}\]
Using this value for λ and rearranging Equation \ref{8-9}, we obtain
\[ - \dfrac {\hbar ^2}{2 \mu r^2} \dfrac {\partial}{\partial r} r^2 \dfrac {\partial}{\partial r} R(r) + \left [ \dfrac {l(l +1) \hbar ^2}{2 \mu r^2} + V (r) - E \right ] R (r) = 0 \label {8-14}\]
Exercise \(\PageIndex{4}\)
Write the steps leading from Equation \ref{8-9} to Equation \ref{8-14}.
The details for solving Equation \ref{8-14} are provided elsewhere, but the procedure and consequences are similar to previously examined cases. As for the harmonic oscillator, an asymptotic solution (valid at large \(r\)) is found, and then the complete solutions are written as products of the asymptotic solution and polynomials arising from sequential truncations of a power series.
The asymptotic solution is
\[ R_{asymptotic} (r) = e^{-\dfrac {r}{n} a_0} \label {8-15}\]
where n will turn out to be a quantum number and \(a_0\) is the Bohr radius. Note that this function decreases exponentially with distance, in a manner similar to the decaying exponential portion of the harmonic oscillator wavefunctions, but with a different distance dependence, \(r\) vs. \(r^2\).
Exercise \(\PageIndex{5}\)
What happens to the magnitude of \(R_{asymptotic}(r)\) as the distance \(r\) from the proton approaches infinity? Sketch a graph of the function, \(R_{asymptotic}(r)\). Why might this behavior be expected for an electron in a hydrogen atom?
ExercisE \(\PageIndex{6}\)
Why is exp(-r/na0) used instead of exp(+r/na0) as the exponential component of the hydrogen atom radial function?
The polynomials produced by the truncation of the power series are related to the associated Laguerre polynomials, \(L_n , _l(r)\), where the set of ci are constant coefficients.
\[L_n, _l (r) = \sum _{r=0}^{n-l-1} c_i r^i \label {8-16}\]
These polynomials are identified by two indices or quantum numbers, n and \(l\). Physically acceptable solutions require that n must be greater than or equal to \(l +1\). The smallest value for \(l\) is zero, so the smallest value for n is 1. The angular momentum quantum number affects the solution to the radial equation because it appears in the radial differential equation, Equation \ref{8-14}.
The \(R(r)\) functions, Equation \ref{9-17}, that solve the radial differential Equation \ref{8-14}, are products of the associated Laguerre polynomials times the exponential factor, multiplied by a normalization factor \((N_{n,l})\) and \(\left (\dfrac {r}{a_0} \right ) ^l\).
\[R (r) = N_{n,l} \left ( \dfrac {r}{a_0} \right ) ^l L_{n,l} (r) e^{-\dfrac {r}{n {a_0}}} \label {8-17}\]
As in Chapter 6, the decreasing exponential term overpowers the increasing polynomial term so that the overall wavefunction exhibits the desired approach to zero at large values of \(r\). The first six radial functions are provided in Table \(\PageIndex{1}\). Note that the functions in the table exhibit a dependence on \(Z\), the atomic number of the nucleus. As discussed later in this chapter, other one electron systems have electronic states analogous to those for the hydrogen atom, and inclusion of the charge on the nucleus allows the same wavefunctions to be used for all one-electron systems. For hydrogen, Z = 1.
| n | \(l\) | \(R_{n,l} (\rho)\) |
|---|---|---|
| 1 | 0 | \(2 \left (\dfrac {Z}{a_0} \right ) ^{3/2} e^{-\rho}\) |
| 2 | 0 | \( \dfrac {1}{2 \sqrt {2}}\left (\dfrac {Z}{a_0} \right ) ^{3/2} (2 - \rho) e^{-\rho/2}\) |
| 2 | 1 | \( \dfrac {1}{2 \sqrt {6}}\left (\dfrac {Z}{a_0} \right ) ^{3/2} \rho e^{-\rho/2}\) |
| 3 | 0 | \( \dfrac {1}{81 \sqrt {3}}\left (\dfrac {Z}{a_0} \right ) ^{3/2} (27 - 18 \rho + 2\rho ^2) e^{-\rho/3}\) |
| 3 | 1 | \( \dfrac {1}{81 \sqrt {6}}\left (\dfrac {Z}{a_0} \right ) ^{3/2} (6 \rho - \rho ^2) e^{-\rho/3}\) |
| 3 | 2 | \( \dfrac {1}{81 \sqrt {30}}\left (\dfrac {Z}{a_0} \right ) ^{3/2} \rho ^2 e^{-\rho/3}\) |
The constraint that n be greater than or equal to \(l +1\) also turns out to quantize the energy, producing the same quantized expression for hydrogen atom energy levels that was obtained from the Bohr model of the hydrogen atom discussed in Chapter 2.
\[ E_n = - \dfrac {\mu e^4}{8 \epsilon ^2_0 h^2 n^2} \]
It is interesting to compare the results obtained by solving the Schrödinger equation with Bohr’s model of the hydrogen atom. There are several ways in which the Schrödinger model and Bohr model differ. First, and perhaps most strikingly, the Schrödinger model does not produce well-defined orbits for the electron. The wavefunctions only give us the probability for the electron to be at various directions and distances from the proton. Second, the quantization of angular momentum is different from that proposed by Bohr. Bohr proposed that the angular momentum is quantized in integer units of \(\hbar\), while the Schrödinger model leads to an angular momentum of \(\sqrt{(l (l +1)} \hbar\). Third, the quantum numbers appear naturally during solution of the Schrödinger equation while Bohr had to postulate the existence of quantized energy states. Although more complex, the Schrödinger model leads to a better correspondence between theory and experiment over a range of applications that was not possible for the Bohr model.
Exercise \(\PageIndex{7}\)
Explain how the Schrödinger equation leads to the conclusion that the angular momentum of the hydrogen atom can be zero, and explain how the existence of such states with zero angular momentum contradicts Bohr's idea that the electron is orbiting around the proton in the hydrogen atom.


