Reactivity of Nitriles
- Page ID
- 5365
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
- Conversion of nitriles to 1° amines using LiAlH4
- Nitriles can be converted to 1° amines by reaction with LiAlH4. During this reaction the hydride nucleophile attacks the electrophilic carbon in the nitrile to form an imine anion. Once stabilized by a Lewis acid-base complexation the imine salt can accept a second hydride to form a dianion. The dianion can then be converted to an amine by addition of water.
- Conversion of nitriles to amides
- Nitriles can be converted to amides. This reaction can be acid or base catalyzed. In the case of acid catalysis the nitrile becomes protonated. Protonation increases the electrophilicity of the nitrile so that it will accept water, a poor nucleophile.
- Conversion of nitriles to carboxylic acids
- Nitriles can be converted to carboxylic acid with heating in sulfuric acid. During the reaction an amide intermediate is formed.
- Conversion to ketones using Grignard reagents
- Grignard reagents can attack the electophillic carbon in a nitrile to form an imine salt. This salt can then be hydrolyzed to become a ketone.
- The Hydrolysis of Nitriles
- This page looks at the hydrolysis of nitriles under either acidic or alkaline conditions to make carboxylic acids or their salts.
- The Reduction of Nitriles
- This page looks at the reduction of nitriles to primary amines using either lithium tetrahydridoaluminate(III) (lithium aluminium hydride) or hydrogen and a metal catalyst.
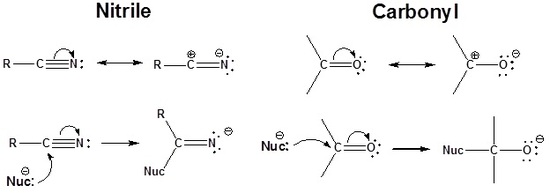
The carbon in a nitrile is electrophilic because a resonance structure can be drawn which places a positive charge on it. Because of this the triple bond of a nitrile accepts a nucleophile in a manner similar to a carbonyl.
Contributors
Prof. Steven Farmer (Sonoma State University)

