25.1: Organic Chemistry
- Page ID
- 53993
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
How many carbon-containing molecules are there? The current estimate is around 20 million different known organic compounds. Why the uncertainty? Every day, scientists are coming up with new compounds. Some of these materials are of interest for a research project, while others are destined to be developed for a commercial market. As soon as we think we know how many organic compounds exist, more are discovered and our number quickly becomes out of date.
Organic Chemistry
At one time in history, it was thought that only living things were capable of synthesizing the carbon-containing compounds present in cells. For that reason, the term organic was applied to those compounds. Eventually it was proved that carbon-containing compounds could be synthesized from inorganic substances, but the term "organic" has remained. Currently, organic compounds are defined as covalently bonded compounds containing carbon, excluding carbonates and oxides. By this definition, compounds such as carbon dioxide \(\left( \ce{CO_2} \right)\) and sodium carbonate \(\left( \ce{Na_2CO_3} \right)\) are considered to be inorganic. Organic chemistry is the study of all organic compounds.
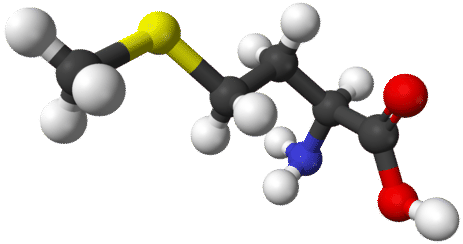
Organic chemistry is a very vast and complex subject. There are millions of known organic compounds—far more than the number of inorganic compounds. The reason lies within the uniqueness of carbon's structure and bonding capabilities. Carbon has four valence electrons, and therefore makes four separate covalent bonds in compounds. Carbon has the ability to bond to itself repeatedly, making long chains of carbon atoms, as well as ringed structures. These bonds can be single, double, or triple covalent bonds. Carbon readily makes covalent bonds with other elements—primarily hydrogen, oxygen, nitrogen, halogens, and several other nonmetals. The figures below show ball-and-stick models of two of the many organic compounds.

The related field of biochemistry overlaps to some extent with organic chemistry. Biochemistry is the study of the chemistry of living systems. Many biochemical compounds are considered to be organic chemicals. Both of the molecules shown above are biochemical materials in terms of their use in the body, but organic chemicals in terms of their structure and chemical reactivity.
Summary
- Organic compounds are defined as covalently bonded compounds containing carbon, excluding carbonates and oxides. (By this definition, compounds such as carbon dioxide \(\left( \ce{CO_2} \right)\) and sodium carbonate \(\left( \ce{Na_2CO_3} \right)\) are considered to be inorganic.)
- Organic chemistry is the study of all organic compounds.
- Biochemistry is the study of the chemistry of living systems.

