24.11: Radioisotopes in Medical Diagnosis and Treatment
- Page ID
- 53990
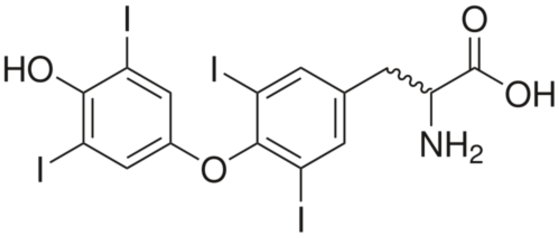
The molecule picture below is thyroxine, a compound produced by the thyroid gland. This molecule regulates how the body uses energy. In a condition known as hypothyroidism, the thyroid makes less thyroxine than normal. A person with this disease feels tired all the time and often puts on weight. Hypothyroidism is treated with thyroid hormone supplements.
Radioisotopes in Medical Diagnosis and Treatment
Radioisotopes are widely used to diagnose disease and as effective treatment tools. For diagnosis, the isotope is administered, and then located in the body using a scanner of some sort. The decay product (often gamma emission) can be located, and the intensity measured. The amount of isotope taken up by the body can then give information as to the extent of the medical problem.
An isotope of iodine (\(\ce{I}\)-131) is used in both the diagnosis and treatment of thyroid cancer. The thyroid will normally absorb iodine to produce the iodine-containing thyroid hormones. An overactive thyroid gland will absorb the radioactive material, which can then destroy excess thyroid tissue or any cancer of the thyroid. The material is sometimes used to image cancers in other parts of the body.
Technetium-99m is perhaps the most widely used radioisotope in diagnosis and treatment (the "m" stands for metastable). This isotope decays to \(\ce{Tc}\)-99 and a gamma emission of low intensity, making the radiation damage fairly negligible. The half-life is about six hours, so it will remain in the body for some time. \(\ce{Tc}\)-99m can be used to look at cardiac damage. The isotope flows in the bloodstream; if there is less blood flow in the heart, there will be less isotope concentrated in the heart muscle. Similar information can be obtained for blood flow in the brain.
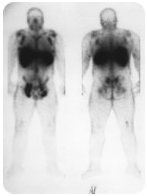
Isotopes can be very useful in scans to locate cancer cells. The patient in the above image has multiple tumors that have spread (metastasized) from the main tumor. A radioisotope has been attached to antibodies that bind to specific cancer cells. The very dark spots in the armpits, neck, and groin represent areas where tumor cells exist.
Many other examples could be presented. There are presently over 25 different isotopes in use for diagnosis and treatment. A very partial list can be seen in the table below.
| Table \(\PageIndex{1}\): Radioisotopes Employed in Diagnosis and/or Treatment | ||
|---|---|---|
| Isotope | Half-Life | Application |
| \(\ce{Cr}\)-51 | 28 days | Label red blood cells. |
| \(\ce{Fe}\)-59 | 446 days | Study iron metabolism in spleen. |
| \(\ce{Xe}\)-133 | 5 days | Study lung function. |
| \(\ce{Ho}\)-166 | 26 hours | Cancer treatment. |

