20.15: Secondary Protein Structure
- Page ID
- 49625
One might expect a long-chain protein molecule to be rather floppy, adopting a variety of molecular shapes and changing rapidly from one conformation to another. In practice this seldom happens. Instead the protein chain stays more or less in the same conformation all the time. It is held in this shape by the cooperative effect of a large number of hydrogen bonds between different segments of the chain.
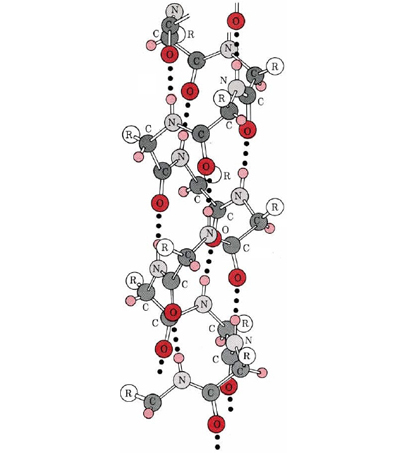
A particularly important conformation of the polypeptide chain is the spiral structure shown in Figure \(\PageIndex{1}\). This is called an α helix. Many fibrous proteins like hair, skin, and nails consist almost entirely of α helices. In globular proteins too, although the overall structure is more complex, short lengths of the chain often have this configuration. In an a helix the polypeptide chain is twisted into a right-hand spiral—the chain turns around clockwise as one moves along it. The spiral is held together by hydrogen bonds from the amido ( ) group of one peptide bond to the carbonyl group of a peptide bond three residues farther along the chain. Two factors 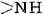 contribute toward making this a particularly stable
contribute toward making this a particularly stable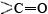 structure. One is the involvement of all the
structure. One is the involvement of all the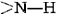 and
and 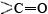 groups in the chain in the hydrogen bonding. Spirals with slightly more or slightly less twist do not permit this. The second factor is the way in which the side chains project outward from an α helix. Bulky side chains therefore do not interfere with the hydrogen bonding, enabling a fairly rigid cylinder to be formed.
groups in the chain in the hydrogen bonding. Spirals with slightly more or slightly less twist do not permit this. The second factor is the way in which the side chains project outward from an α helix. Bulky side chains therefore do not interfere with the hydrogen bonding, enabling a fairly rigid cylinder to be formed.
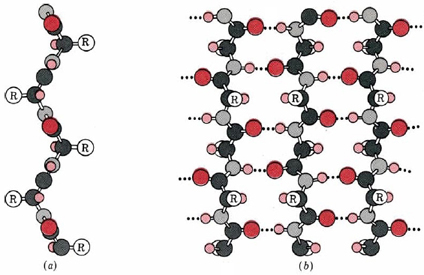
A second regular arrangement of the polypeptide chain is the β sheet, the β-keratin structure found in silk and shown in Figure \(\PageIndex{2}\). As in the α helix, this structure allows all the amido and carbonyl groups to participate in hydrogen bonds. This hydrogen bonding structure can be accomplished in two manners, either a parallel or antiparallel β sheet, which are compared in Figure \(\PageIndex{3}\). Unlike the α helix, though, the side chains are squeezed rather close together in a pleated-sheet arrangement. In consequence very bulky side chains make the structure unstable. This explains why silk is composed almost entirely of glycine, alanine, and serine, the three amino acids with the smallest side chains. Most other proteins contain a much more haphazard collection of amino acid residues.


