18.6 Reactions of Epoxides: Ring-opening
- Page ID
- 90957
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Objectives
After completing this section, you should be able to
- write an equation to describe the opening of an epoxide ring under mildly acidic conditions.
- identify the product formed from the hydrolysis of an epoxide.
- write the mechanism for the opening of an epoxide ring by an aqueous acid, paying particular attention to the stereochemistry of the product.
- identify the product formed when an epoxide ring is opened by a hydrogen halide under anhydrous conditions.
- predict the major product from the acidic cleavage of a given unsymmetrical epoxide.
- write an equation to illustrate the cleavage of an epoxide ring by a base.
- identify the product formed from the reaction of a given epoxide with given base.
- explain why epoxides are susceptible to cleavage by bases, whereas other cyclic ethers are not.
In the discussion on base‑catalyzed epoxide opening, the mechanism is essentially SN2. While oxygen is a poor leaving group, the ring strain of the epoxide really helps to drive this reaction to completion. Indeed, larger cyclic ethers would not be susceptible to either acid‑catalyzed or base‑catalyzed cleavage under the same conditions because the ring strain is not as great as in the three‑membered epoxide ring.
Epoxide ring-opening reactions - SN1 vs. SN2, regioselectivity, and stereoselectivity
The nonenzymatic ring-opening reactions of epoxides provide a nice overview of many of the concepts we have seen already in this chapter. Ring-opening reactions can proceed by either SN2 or SN1 mechanisms, depending on the nature of the epoxide and on the reaction conditions. If the epoxide is asymmetric, the structure of the product will vary according to which mechanism dominates. When an asymmetric epoxide undergoes solvolysis in basic methanol, ring-opening occurs by an SN2 mechanism, and the less substituted carbon is the site of nucleophilic attack, leading to what we will refer to as product B:

Conversely, when solvolysis occurs in acidic methanol, the reaction occurs by a mechanism with substantial SN1 character, and the more substituted carbon is the site of attack. As a result, product A predominates.

These are both good examples of regioselective reactions. In a regioselective reaction, two (or more) different constitutional isomers are possible as products, but one is formed preferentially (or sometimes exclusively).
Let us examine the basic, SN2 case first. The leaving group is an alkoxide anion, because there is no acid available to protonate the oxygen prior to ring opening. An alkoxide is a poor leaving group, and thus the ring is unlikely to open without a 'push' from the nucleophile.
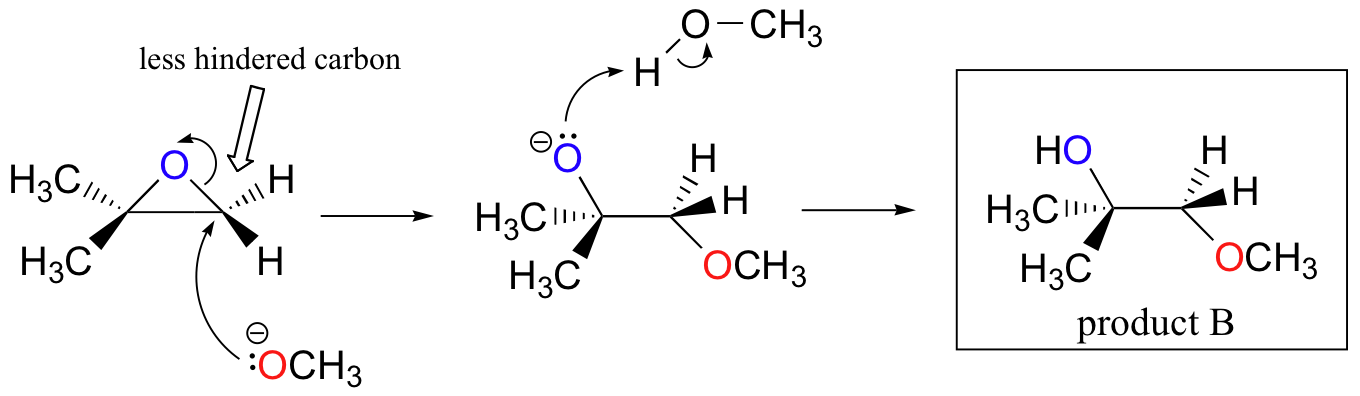
The nucleophile itself is potent: a deprotonated, negatively charged methoxide ion. When a nucleophilic substitution reaction involves a poor leaving group and a powerful nucleophile, it is very likely to proceed by an SN2 mechanism.
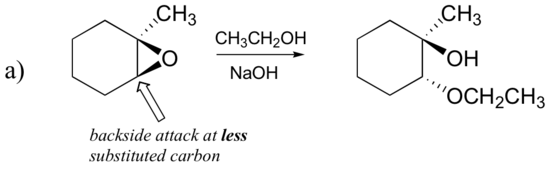
What about the electrophile? There are two electrophilic carbons in the epoxide, but the best target for the nucleophile in an SN2 reaction is the carbon that is least hindered. This accounts for the observed regiochemical outcome. Like in other SN2 reactions, nucleophilic attack takes place from the backside, resulting in inversion at the electrophilic carbon.
Probably the best way to depict the acid-catalyzed epoxide ring-opening reaction is as a hybrid, or cross, between an SN2 and SN1 mechanism. First, the oxygen is protonated, creating a good leaving group (step 1 below) . Then the carbon-oxygen bond begins to break (step 2) and positive charge begins to build up on the more substituted carbon (recall the discussion from section 8.4B about carbocation stability).

Unlike in an SN1 reaction, the nucleophile attacks the electrophilic carbon (step 3) before a complete carbocation intermediate has a chance to form.
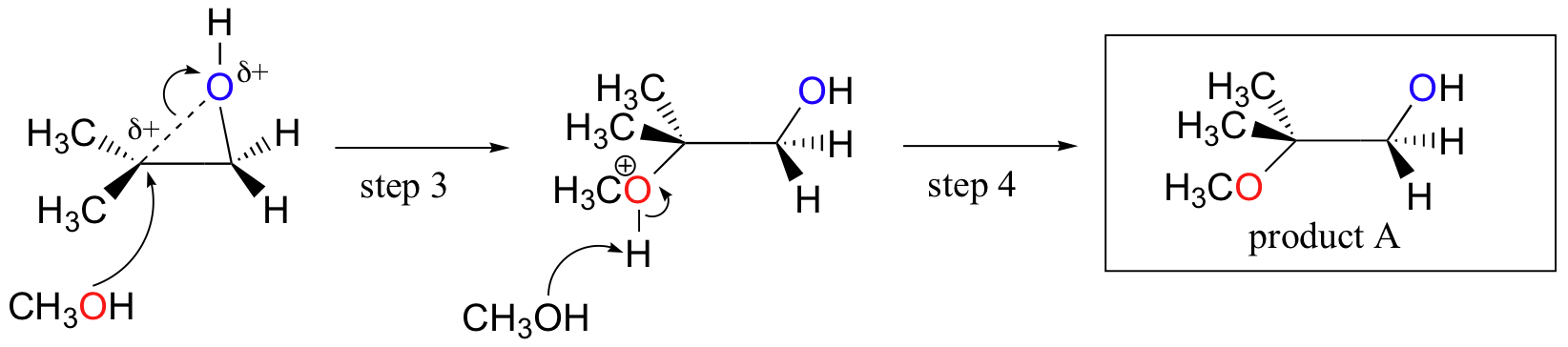
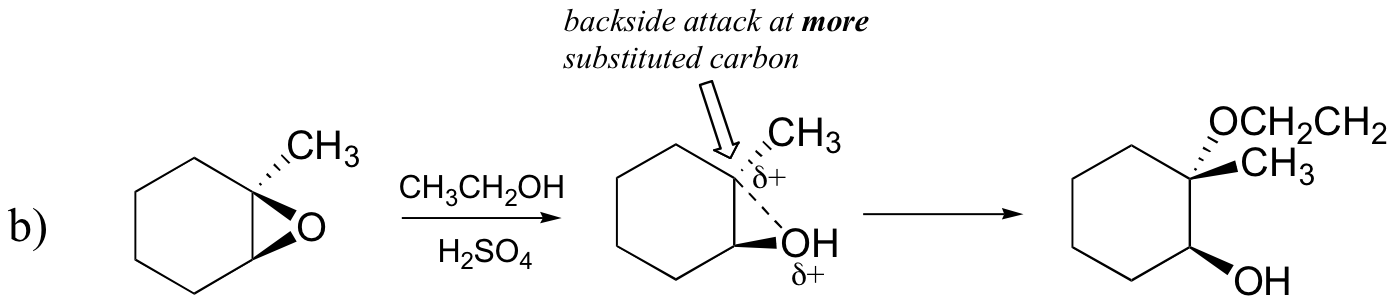
Attack takes place preferentially from the backside (like in an SN2 reaction) because the carbon-oxygen bond is still to some degree in place, and the oxygen blocks attack from the front side. Notice, however, how the regiochemical outcome is different from the base-catalyzed reaction: in the acid-catalyzed process, the nucleophile attacks the more substituted carbon because it is this carbon that holds a greater degree of positive charge.
Predict the major product(s) of the ring opening reaction that occurs when the epoxide shown below is treated with:
- ethanol and a small amount of sodium hydroxide
- ethanol and a small amount of sulfuric acid
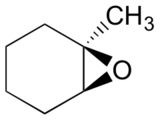
Hint: be sure to consider both regiochemistry and stereochemistry!
- Answer
-
Anti Dihydroxylation
Epoxides may be cleaved by aqueous acid to give glycols that are often diastereomeric with those prepared by the syn-hydroxylation reaction described above. Proton transfer from the acid catalyst generates the conjugate acid of the epoxide, which is attacked by nucleophiles such as water in the same way that the cyclic bromonium ion described above undergoes reaction. The result is anti-hydroxylation of the double bond, in contrast to the syn-stereoselectivity of the earlier method. In the following equation this procedure is illustrated for a cis-disubstituted epoxide, which, of course, could be prepared from the corresponding cis-alkene. This hydration of an epoxide does not change the oxidation state of any atoms or groups.
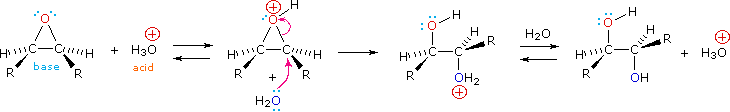
Addition of HX
Epoxides can also be opened by other anhydrous acids (HX) to form a trans halohydrin. When both the epoxide carbons are either primary or secondary the halogen anion will attack the less substituted carbon and an SN2 like reaction. However, if one of the epoxide carbons is tertiary, the halogen anion will primarily attack the tertiary carbon in a SN1 like reaction.

Exercises
Provide the structure of the product of the following reaction. Be sure to include proper stereochemistry.
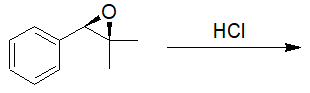
- Answer
-
The ring side of the protonated epoxide intermediate will better stabilize a partial positive charge, so would be the more likely carbon for the chloride ion to attack.
Predict the product of the following reaction.
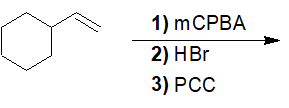
- Answer
-

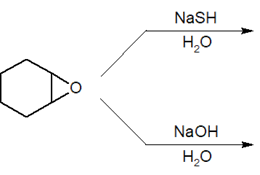
Provide the final products of the following reactions.
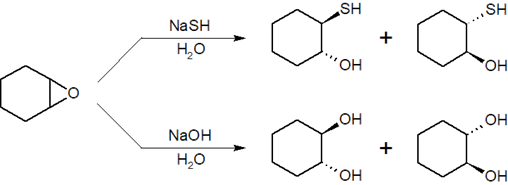
- Answer
-
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry

