18.4 Reactions of Ethers - Claisen Rearrangement
- Page ID
- 90955
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
Objectives
After completing this section, you should be able to:
- write an equation to represent the Claisen rearrangement of allyl phenyl ester.
- account for the formation of a specific product from a Claisen rearrangement, without giving mechanistic details.
Make certain that you can define, and use in context, the key term below:
- Claisen rearrangement
Cope and Claisen rearrangements
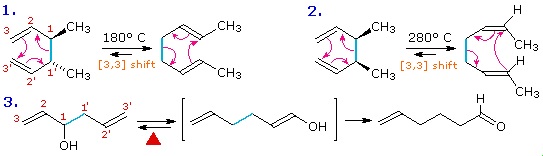
The [3,3] sigmatropic rearrangement of 1,5-dienes or allyl vinyl ethers, known respectively as the Cope and Claisen rearrangements, are among the most commonly used sigmatropic reactions. Three examples of the Cope rearrangement are shown in the following diagram. Reactions 1 and 2 (top row) demonstrate the stereospecificity of this reaction. The light blue σ-bond joins two allyl groups, oriented so their ends are near each other. Since each allyl segment is the locus of a [1,3] shift, the overall reaction is classified as a [3,3] rearrangement. The three pink colored curved arrows describe the redistribution of three bonding electron pairs in the course of this reversible rearrangement. The diene reactant in the third reaction is drawn in an extended conformation. This molecule must assume a coiled conformation (as above) before the [3,3] rearrangement can take place. The product of this rearrangement is an enol which immediately tautomerizes to its keto form. Such variants are termed the oxy-Cope rearrangement, and are useful because the reverse rearrangement is blocked by rapid ketonization. If the hydroxyl substituent is converted to an alkoxide salt, the activation energy of the rearrangement is lowered significantly.
The degenerate or self-replicating Cope rearrangement has been a fascinating subject of research. For examples .
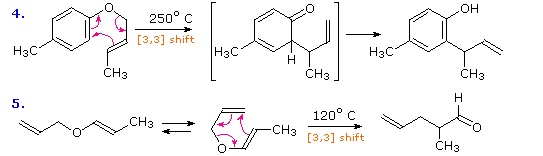
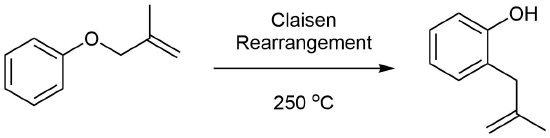
Two examples of the Claisen Rearrangement may be seen. Reaction 4 is the classic rearrangement of an allyl phenyl ether to an ortho-allyl phenol. The methyl substituent on the allyl moiety serves to demonstrate the bonding shift at that site. The initial cyclohexadienone product immediately tautomerizes to a phenol, regaining the stability of the aromatic ring. Reaction 5 is an aliphatic analog in which a vinyl group replaces the aromatic ring. In both cases three pairs of bonding electrons undergo a reorganization.
Exercises
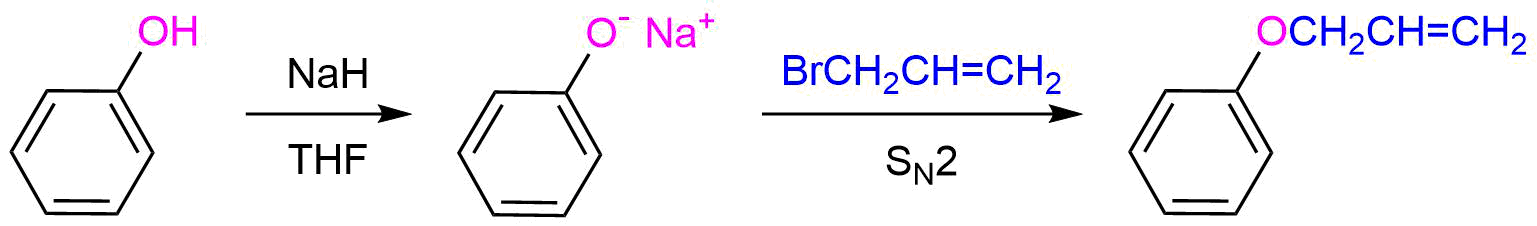
Show how you could synthesize allyl phenyl ether from allyl bromide and phenol.
- Answer
-
What would be the expected product of the following Claisen rearrangement?
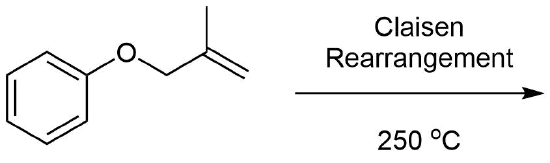
- Answer
-
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry

