2.6: Drawing Resonance Forms
- Page ID
- 67057
Objective
After completing this section, you should be able to draw all possible resonance structures for a given species.
Resonance is a mental exercise and method within the Valence Bond Theory of bonding that describes the delocalization of electrons within molecules. It compares and contrasts two or more possible Lewis structures that can represent a particular molecule. Resonance structures are used when one Lewis structure for a single molecule cannot fully describe the bonding that takes place between neighboring atoms relative to the empirical data for the actual bond lengths between those atoms. The net sum of valid resonance structures is defined as a resonance hybrid, which represents the overall delocalization of electrons within the molecule. A molecule that has several resonance structures is more stable than one with fewer. Some resonance structures are more favorable than others.
Introduction
Electrons have no fixed position in atoms, compounds and molecules (see image below) but have probabilities of being found in certain spaces (orbitals). Resonance forms illustrate areas of higher probabilities (electron densities). This is like holding your hat in either your right hand or your left. The term Resonance is applied when there are two or more possibilities available. Resonance structures do not change the relative positions of the atoms like your arms in the metaphor. The skeleton of the Lewis Structure remains the same, only the electron locations change. A double headed arrow on both ends of the arrow ( \( \leftrightarrow \) ) between Lewis structures is used to show their inter-connectivity. It is different from the double harpoons ( \( \rightleftharpoons\) ) used for designating equilibria. A double headed arrow on only one end ( \( \rightarrow \) ) is used to indicate the movement of two electrons in a single resonance structure.
Consider ozone (O3)
Solution
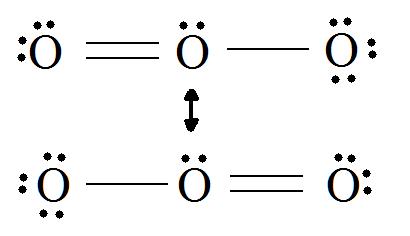
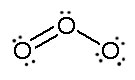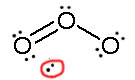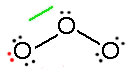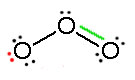
Figure: This is an animation of how one can do a resonance with ozone by moving electrons.
Delocalization and Resonance Structures Rules
In resonance structures, the electrons are able to move to help stabilize the molecule. This movement of the electrons is called delocalization.
- Resonance structures should have the same number of electrons, do not add or subtract any electrons. (You can check the number of electrons by counting them)
- All resonance structures must follow the rules of writing Lewis Structures.
- The hybridization of the structure must stay the same.
- The skeleton of the structure can not be changed (only the electrons move).
- Resonance structures must also have the same amount of lone pairs.
Formal Charge
Even though the structures look the same, the formal charge (FC) may not be. Formal charges are charges that are assigned to a specific atom in a molecule. If computed correctly, the overall formal charge of the molecule should be the same as the oxidation charge of the molecule (the charge when you write out the empirical and molecular formula) We want to choose the resonance structure with the least formal charges that add up to zero or the charge of the overall molecule.
The equation for finding Formal Charge is:
Formal Charge = (number of valence electrons in free orbital) - (number of lone-pair electrons) - ( \( \frac{1}{2} \) number bond pair electrons)
The formal charge has to equal the molecule's overall charge.
Ex.) CNS- has an overall charge of -1, so the Lewis structure's formal charge has to equal -1. See Lewis Structure for more information.
Consider the thiocyanate (CNS-) ion.
Solution
1. Find the Lewis Structure of the molecule. (Remember the Lewis Structure rules.)
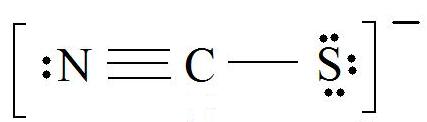
2. Resonance: All elements want an octet, and we can do that in multiple ways by moving the terminal atom's electrons around (bonds too).
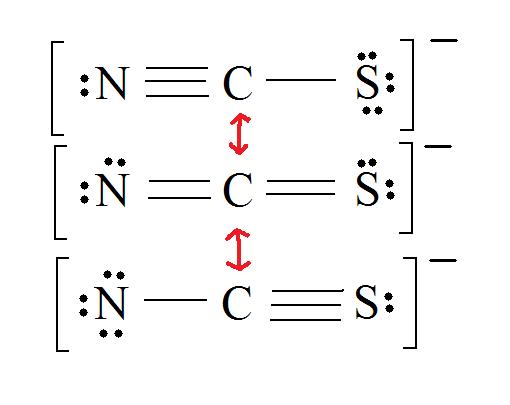
3. Assign Formal Charges
Formal Charge = (number of valence electrons in free orbital) - (number of lone-pair electrons) - ( \( \frac{1}{2} \) number bond pair electrons)
Remember to determine the number of valence electron each atom has before assigning Formal Charges
C = 4 valence e-, N = 5 valence e-, S = 6 valence e-, also add an extra electron for the (-1) charge. The total of valence electrons is 16.
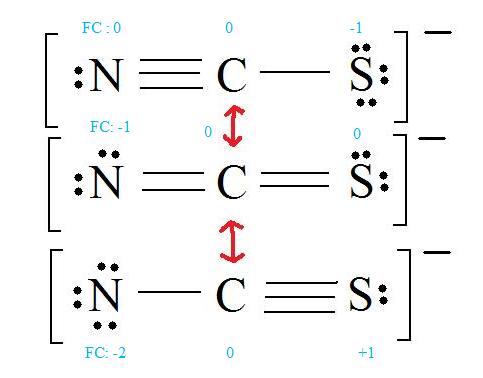
4. Find the most ideal resonance structure. (Note: It is the one with the least formal charges that adds up to zero or to the molecule's overall charge.)
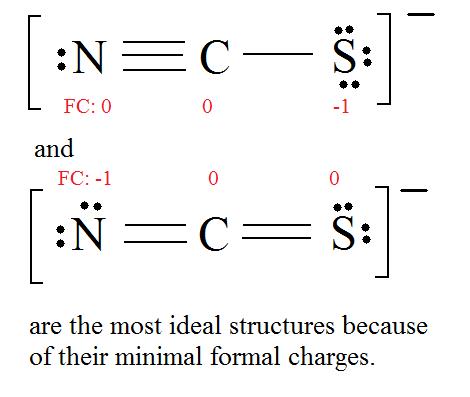
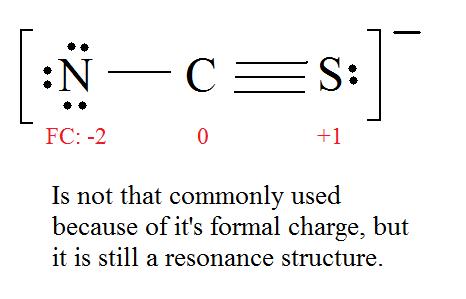
5. Now we have to look at electronegativity for the "Correct" Lewis structure.
The most electronegative atom usually has the negative formal charge, while the least electronegative atom usually has the positive formal charges.
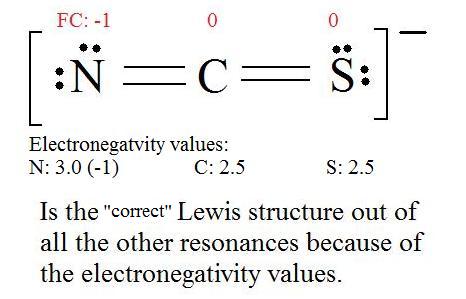
Resonance Hybrids
Resonance Structures are a representation of a Resonance Hybrid, which is the combination of all resonance structures. Though the Formal Charge closest to zero is the most accepted structure, in reality the correct Lewis structure is actually a combination of all the resonance structures (and hence is not solely describe as one).
- Draw the Lewis Structure & Resonance for the molecule (using solid lines for bonds).
- Where there can be a double or triple bond, draw a dotted line (-----) for a bond.
- Draw only the lone pairs found in all resonance structures, do not include the lone pairs that are not on all of the resonance structures.
Consider the carbonate ion: CO32-
Solution
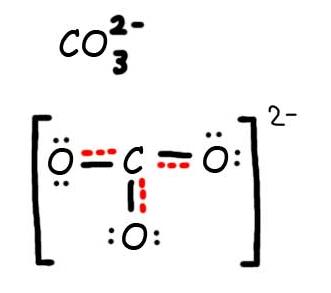
Step 1: Draw the Lewis Structure & Resonance.
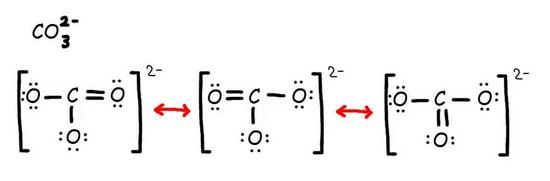
Step 2: Combine the resonance structures by adding (dotted) bonds where other resonance bonds can be formed.
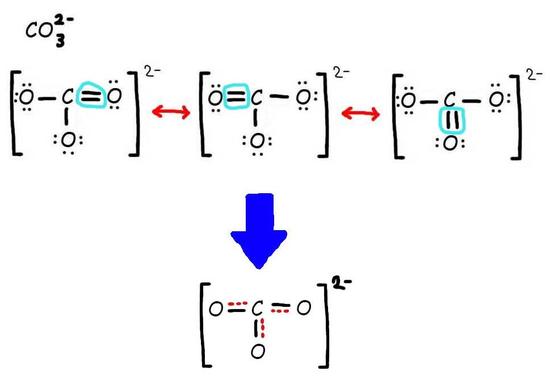
Step 3: Add only the lone pairs found on ALL resonance structures.
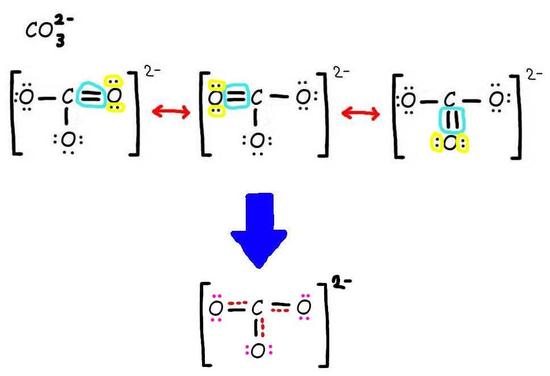
The bottom is the finished resonance hybrid for CO32-.
Rules for estimating stability of resonance structures
- The greater the number of covalent bonds, the greater the stability since more atoms will have complete octets
- The structure with the least number of formal charges is more stable
- The structure with the least separation of formal charge is more stable
- A structure with a negative charge on the more electronegative atom will be more stable
- Positive charges on the least electronegative atom (most electropositive) is more stable
- Resonance forms that are equivalent have no difference in stability and contribute equally. (eg. benzene)
Draw the resonance contributors that correspond to the curved, two-electron movement arrows in the resonance expressions below. Then identify the type of resonance motion in each structure below.
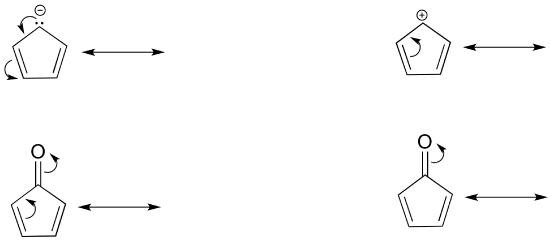
- Answer
-
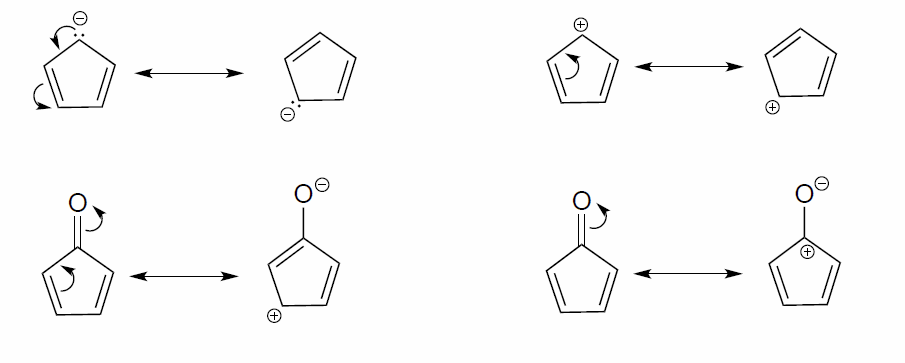
In each resonance expression, identify the type of resonance motion. Then draw curved arrows on the left-side contributor that shows how we get to the right-side contributor.
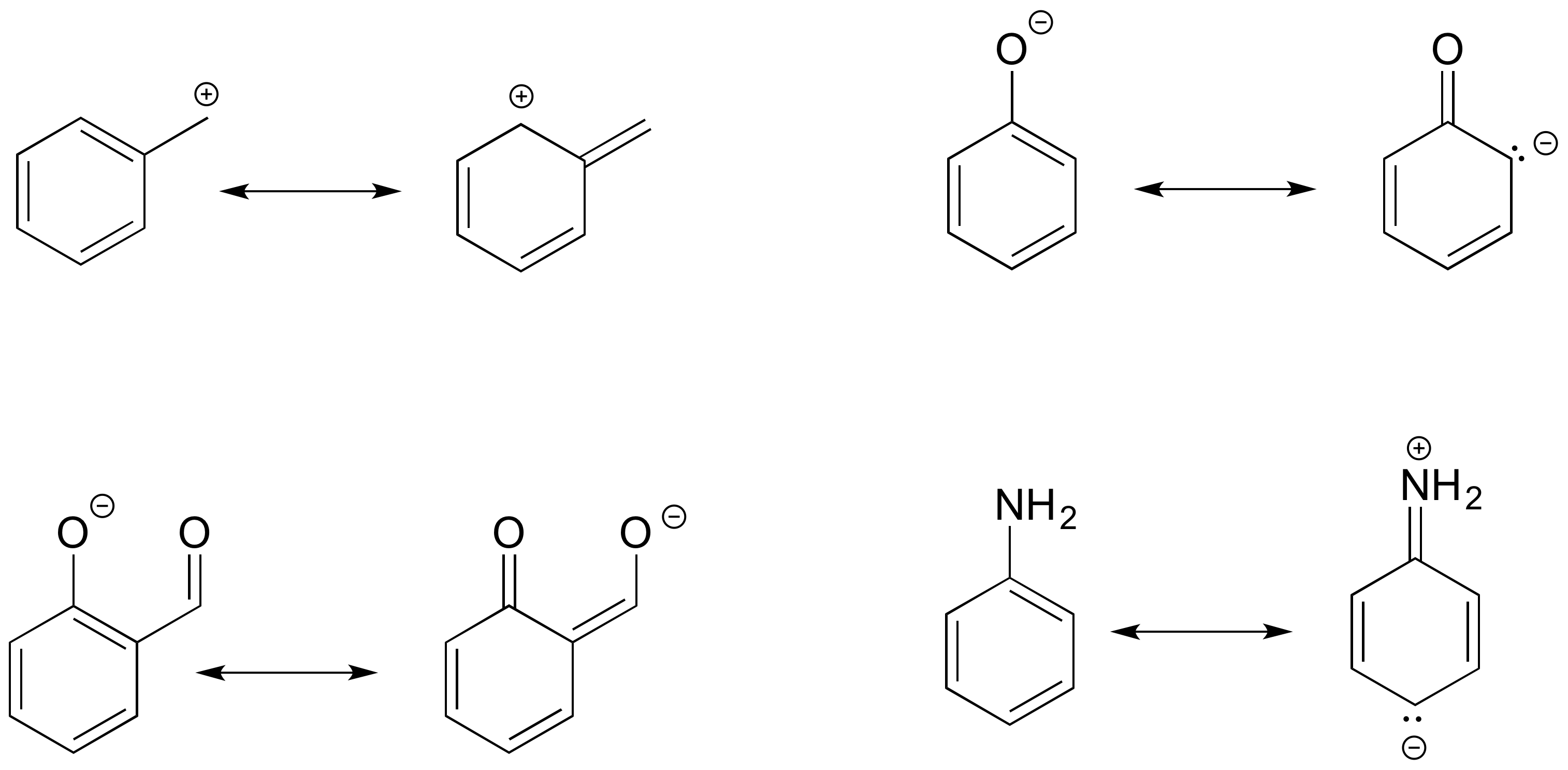
- Answer
-
-
bond.
a) Draw three additional resonance contributors for the carbocation below. Include in your figure the appropriate curved arrows showing how one contributor is converted to the next.

b) Fill in the blanks: the conjugated pi system in this carbocation is composed of ______ 2p orbitals sharing ________ delocalized pi electrons.
- Answer
-
a)
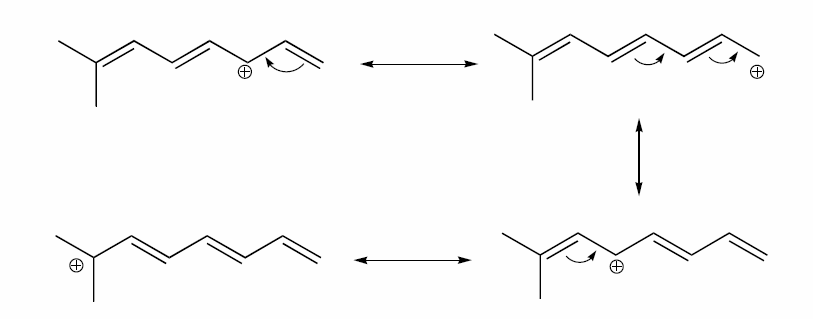
b) The conjugated pi system in this carbocation is composed of seven p orbitals containing six delocalized pi electrons.
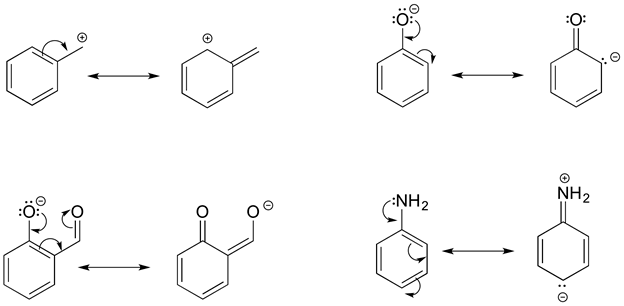
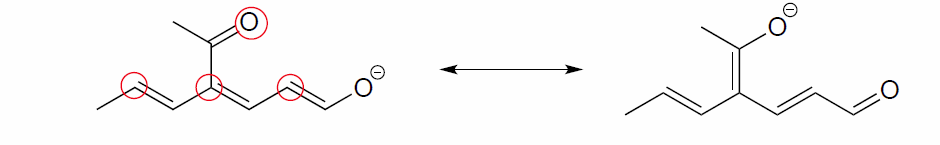
The figure below shows how the negative formal charge on an oxygen (of an enol) can be delocalized to the carbon indicated by an arrow. More resonance contributors can be drawn in which negative charge is delocalized to three other atoms on the molecule.
a) Circle these atoms that can also have a resonance structure with a negative charge.
b) Draw the two most important resonance contributors for the enolate ion.
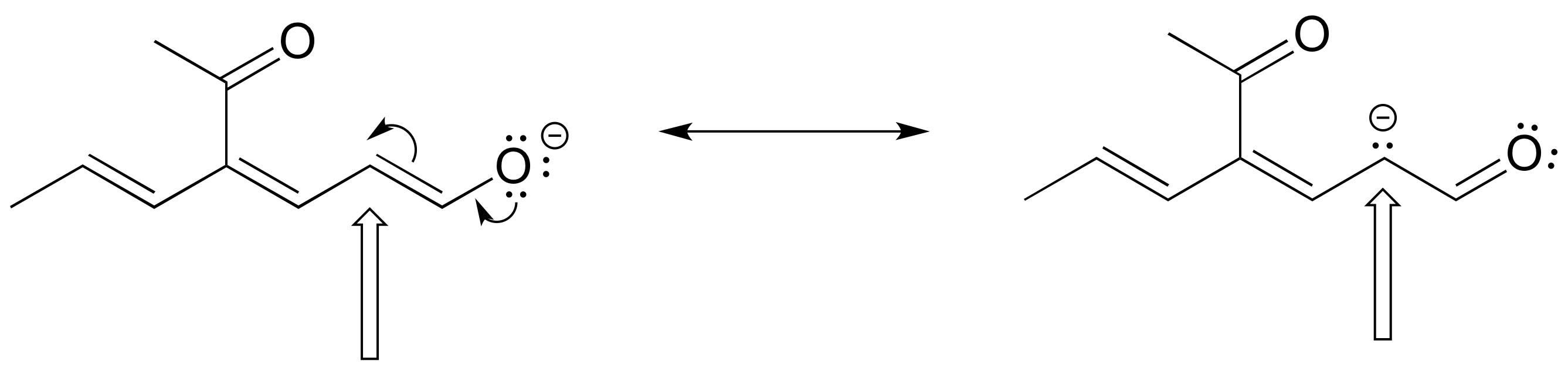
- Answer
-
The two major contributors are those in which the negative formal charge is located on an oxygen rather than on a carbon.
References
- Petrucci, Ralph H., et al. General Chemistry: Principles and Modern Applications. New Jersey: Pearson Prentice Hall, 2007. Print.
- Ahmad, Wan-Yaacob and Zakaria, Mat B. "Drawing Lewis Structures from Lewis Symbols: A Direct Electron Pairing Approach." Journal of Chemical Education: Journal 77.3: n. pag. Web. March 2000. Link to this journal: pkukmweb.ukm.my/~mbz/c_penerbitan/jurnal/jce2k77%283%29/p329.pdf
Outside links
- http://en.Wikipedia.org/wiki/Resonance_(chemistry)
- http://www.absoluteastronomy.com/topics/Resonance_(chemistry)#encyclopedia
- http://www.nku.edu/~russellk/tutorial/reson/resonance.html
- http://en.Wikipedia.org/wiki/Formal_charge
- http://commons.wikimedia.org/wiki/Main_Page (for the electronegatvity chart)
- http://misterguch.brinkster.net/PRA037.pdf (for problem 5)
- http://commons.wikimedia.org/wiki/File:Phosphite-ion-resonance-structures-2D.png (for the (HPO32-) problem 4 answer)
- http://commons.wikimedia.org/wiki/File:Sulfate-resonance-2D.png (for the Sulfate answer)
- http://www.mpcfaculty.net/mark_bishop/resonance.htm
- www.chem.ucla.edu/harding/tutorials/resonance/draw_res_str.html
Problems
- True or False, The picture below is a resonance structure?
- Draw the Lewis Dot Structure for SO42- and all possible resonance structures. Which of the following resonance structure is not favored among the Lewis Structures? Explain why. Assign Formal Charges.
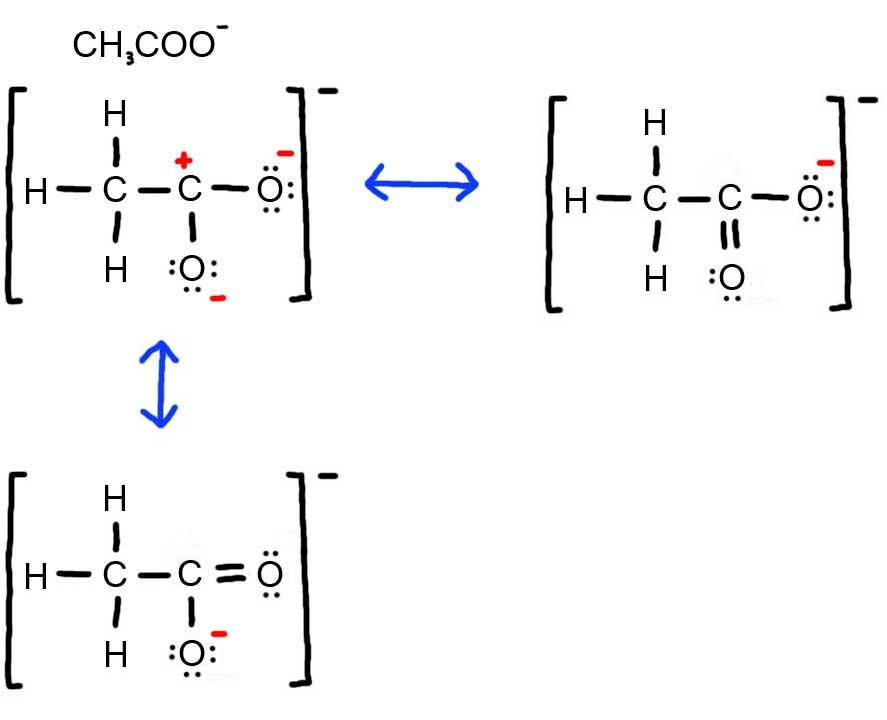
- Draw the Lewis Dot Structure for CH3COO- and all possible resonance structures. Assign Formal Charges. Choose the most favorable Lewis Structure.
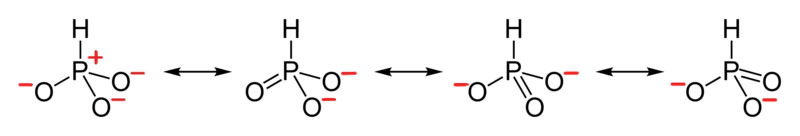
- Draw the Lewis Dot Structure for HPO32- and all possible resonance structures. Assign Formal Charges.
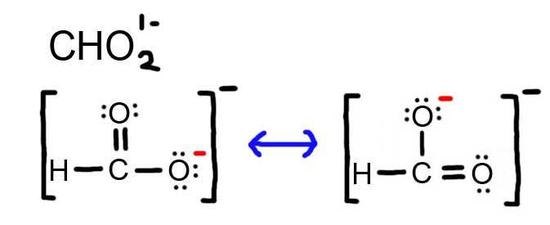
- Draw the Lewis Dot Structure for CHO21- and all possible resonance structures. Assign Formal Charges.
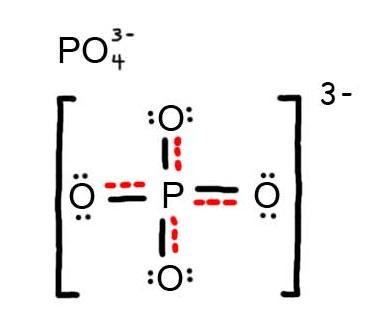
- Draw the Resonance Hybrid Structure for PO43-.
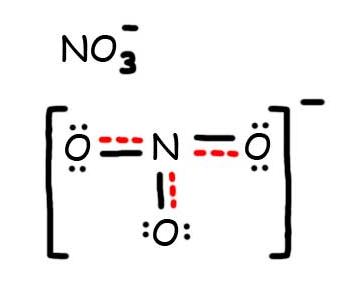
- Draw the Resonance Hybrid Structure for NO3-.
Answers
1. False, because the electrons were not moved around, only the atoms (this violates the Resonance Structure Rules).
2. Below are the all Lewis dot structure with formal charges (in red) for Sulfate (SO42-). There isn't a most favorable resonance of the Sulfate ion because they are all identical in charge and there is no change in Electronegativity between the Oxygen atoms.
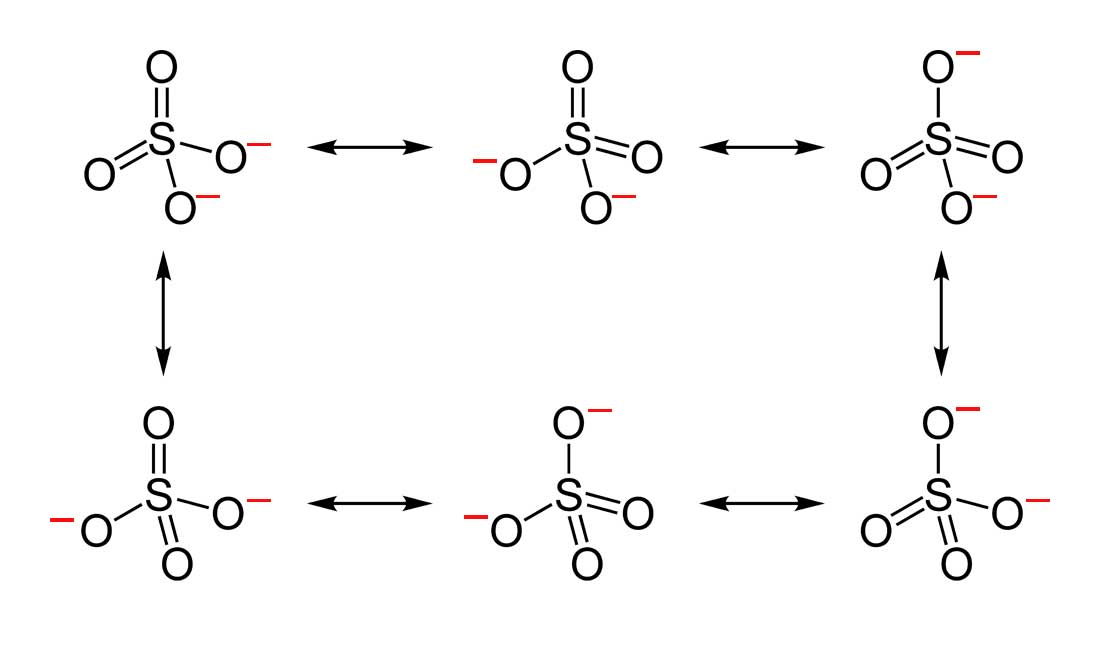
3. Below is the resonance for CH3COO-, formal charges are displayed in red. The Lewis Structure with the most formal charges is not desirable, because we want the Lewis Structure with the least formal charge.
4. The resonance for HPO32-, and the formal charges (in red).
5. The resonance for CHO21-, and the formal charges (in red).
6. The resonance hybrid for PO43-, hybrid bonds are in red.
7. The resonance hybrid for NO3-, hybrid bonds are in red.
Contributors and Attributions
- Sharon Wei (UCD), Liza Chu (UCD)
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)

