4.8: Formula Mass, Molecular Mass, and Molar Mass
- Page ID
- 525765
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Calculate the formula masses of ionic compounds and molecular masses of molecules.
- Calculate the molar mass of an element or compound.
- Determine the number of atoms, formula units, or molecules in one mole of a substance.
Formula Mass (for Ionic Compounds) and Molecular Mass (for Molecules)
Ionic compounds are composed of discrete cations and anions combined in ratios to yield electrically neutral bulk matter. The formula mass for an ionic compound is calculated in the same way as the formula mass for covalent compounds: by summing the average atomic masses of all the atoms in the compound’s formula. Keep in mind, however, that the formula for an ionic compound does not represent the composition of a discrete molecule, so it may not correctly be referred to as the “molecular mass.”
As an example, consider sodium chloride, NaCl, the chemical name for common table salt. Sodium chloride is an ionic compound composed of sodium cations, Na+, and chloride anions, Cl−, combined in a 1:1 ratio. As you may recall, the periodic table shows the average mass of the atoms for each element. We refer to this mass as the atomic mass. The atomic mass of sodium is 22.99 amu, and the atomic mass of chlorine is 35.45 amu.
\(\boxed{\;\overset{11}{\underset{22.99}{\mathrm{Na}}}\;}\;\;\;\boxed{\;\overset{17}{\underset{35.45}{\mathrm{Cl}}}\;}\)
The formula mass for this compound is computed as 58.44 amu (Figure \(\PageIndex{1}\)).
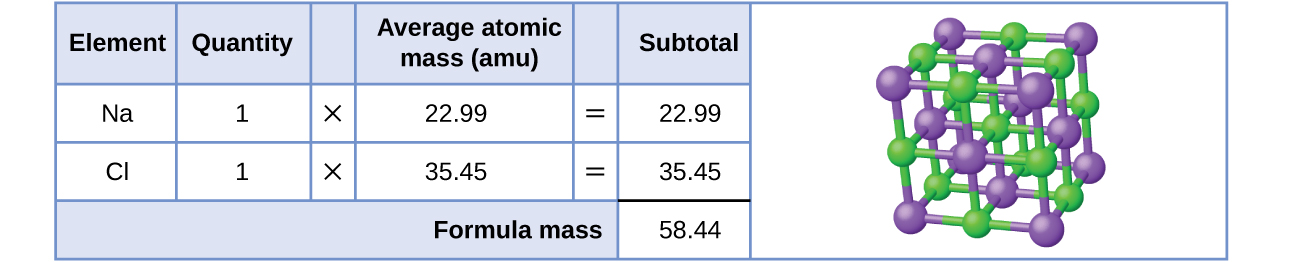
Note that the average masses of neutral sodium and chlorine atoms were used in this computation, rather than the masses for sodium cations and chlorine anions. This approach is perfectly acceptable when computing the formula mass of an ionic compound. Even though a sodium cation has a slightly smaller mass than a sodium atom (since it is missing an electron), this difference will be offset by the fact that a chloride anion is slightly more massive than a chloride atom (due to the extra electron). Moreover, the mass of an electron is negligibly small with respect to the mass of a typical atom. Even when calculating the mass of an isolated ion, the missing or additional electrons can generally be ignored, as their contribution to the overall mass is negligible and reflected only in the non-significant digits that will be lost when the computed mass is properly rounded. The few exceptions to this guideline are very light ions derived from elements with precisely known atomic masses.
Calculate the molecular mass or formula mass for each compound: (Periodic Table)
- C12H22O11
- Ca(NO3)2
Solution
| C12H22O11 | Ca(NO3)2 | |
|---|---|---|
|
12 C = 12x(12.01) = 144.1 amu 22 H = 22x(1.008) = 22.18 amu 11 O = 11x(16.00) = 176.0 amu |
1 Ca = 1x(40.08) = 40.08 amu 2 N = 2x(14.01) = 28.02 amu 6 O = 6x(16.00) = 96.00 amu |
|
molecular mass = 342.3 amu | formula mass = 164.10 amu |
|
It is called a molecular mass since C12H22O11 is a molecular compound. | It is called a formula mass since Ca(NO3)2 is an ionic compound. |
|
One molecule of C12H22O11 has a mass of 342.3 amu. | One formula unit of Ca(NO3)2 has a mass of 164.10 amu. |
Aluminum sulfate, Al2(SO4)3, is an ionic compound that is used in the manufacture of paper and in various water purification processes. What is the formula mass (amu) of this compound?
Solution
The formula for this compound indicates it contains Al3+ and SO42− ions combined in a 2:3 ratio. For the purpose of computing a formula mass, it is helpful to rewrite the formula in a simpler format, Al2S3O12. Following the approach outlined above, the formula mass for this compound is calculated as follows:

Molar Mass of an Element
According to the periodic table, the atomic mass of aluminum is 26.98 amu, copper is 63.55 amu, and carbon is 12.01 amu. Since 1 amu is only 1.674 × 10−24 g, these masses would be way too small to measure on ordinary laboratory equipment. However, if we have 6.022 × 1023 atoms, or 1 mole of each of these elements, we would have 26.98 g of Al, 63.55 g of Cu, and 12.01 g of C.
In other words, one mole of any element will have a mass that is numerically equal to its atomic mass and expressed in units of grams. This mass is called the molar mass.

We might find these easier to use if the relationships are written out like this (recall that mole is abbreviated as mol):
| Substance | Atomic Mass | Molar Mass | Atoms in a Mol |
|---|---|---|---|
| Aluminum (Al) | 1 atom Al = 26.98 amu | 1 mol Al = 26.98 g | 1 mol Al = 6.022 × 1023 atoms Al |
| Copper (Cu) | 1 atom Cu = 63.55 amu | 1 mol Cu = 63.55 g | 1 mol Cu = 6.022 × 1023 atoms Cu |
| Carbon (C) | 1 atom C = 12.01 amu | 1 mol C = 12.01 g | 1 mol C = 6.022 × 1023 atoms C |
Molar Mass of a Compound
One mole of any compound will have a mass that is numerically equal to its molecular mass or formula mass and expressed in units of grams. This mass is also called the molar mass.
The formula mass for NaCl is 58.44 amu. This means that the molar mass of NaCl is 58.44 g, and it will contain 6.022 × 1023 formula units of NaCl, or one mole of NaCl. We also calculated the molecular mass for C12H22O11 to be 342.3 amu. This means that the molar mass of C12H22O11 is 342.3 g, and it will contain 6.022 × 1023 molecules of C12H22O11, or one mole of C12H22O11.
We might find these easier to use if the relationships are written out like this (recall that mole is abbreviated as mol):
- 1 mol NaCl = 58.44 g NaCl = 6.022 × 1023 formula units NaCl
- 1 mol C12H22O11 = 342.3 g C12H22O11 = 6.022 × 1023 molecules C12H22O11

Summary of Relevant Terminology
Keeping the terminology straight (see Table \(\PageIndex{1}\)) is crucial for understanding the mole concept and accurately performing mole calculations.
| Substance | Fundamental Unit | Atomic, Molecular, or Formula Mass | Molar Mass |
|---|---|---|---|
| aluminum (Al) | atom | atomic mass = 26.98 amu | molar mass = 26.98 g |
| sucrose (C12H22O11) | molecule | molecular mass = 342.3 amu | molar mass = 342.3 g |
| sodium chloride (NaCl) | formula unit | formula mass = 58.44 amu | molar mass = 58.44 g |
Find the molar mass of each element. Then determine how many atoms are contained in the calculated molar mass. (Periodic Table)
- K
- Pb
Solution
- Since the atomic mass of K is 39.10 amu, it has a molar mass of 39.10 g. There are 6.022 × 1023 atoms K, which is also 1 mole of K, in 39.10 g K.
- Since the atomic mass of Pb is 207.2 amu, it has a molar mass of 207.2 g. There are 6.022 × 1023 atoms Pb, which is also 1 mole of Pb, in 207.2 g Pb.
Calculate the molar mass for each compound. Then determine how many formula units or molecules are contained in the calculated molar mass. (Periodic Table)
- H2O
- Ca(NO3)2
Solution
| H2O | Ca(NO3)2 | |
|---|---|---|
|
2 H = 2(1.008) = 2.016 g H 1 O = 1(16.00) = 16.00 g O |
1 Ca = 1(40.08) = 40.08 g Ca 2 N = 2(14.01) = 28.02 g N 6 O = 6(16.00) = 96.00 g O |
|
molar mass of H2O = 18.02 g | molar mass of Ca(NO3)2 = 164.10 g |
|
H2O is a molecular compound. | Ca(NO3)2 is an ionic compound. |
|
6.022 × 1023 molecules H2O, which is also 1 mol H2O, are in 18.02 g H2O. | 6.022 × 1023 formula units Ca(NO3)2, which is also 1 mol Ca(NO3)2, are in 164.10 g Ca(NO3)2. |
Calcium phosphate, \(\ce{Ca3(PO4)2}\), is an ionic compound and a common anti-caking agent added to food products. What is the formula mass (amu) of calcium phosphate?
- Answer
-
310.18 amu
Calculate the molecular mass or formula mass for each compound. (Periodic Table)
- TiO2
- C6H12O6
- Fe3(PO4)2
- Answer
-
- The formula mass is 79.90 amu.
- The molecular mass is 180.16 amu.
- The formula mass is 357.5 amu.
Calculate the molar mass for each substance. Then show the number of grams in 1 mol of the substance, as well as the number of atoms, molecules, or formula units that are contained 1 mol of the substance. (Periodic Table)
- Na
- Na2S
- N2O
- Answer
-
- molar mass = 22.99 g; Therefore, 1 mol Na = 22.99 g Na = 6.022 × 1023 atoms Na.
- molar mass = 78.05 g; Therefore, 1 mol Na2S = 78.05 g Na2S = 6.022 × 1023 formula units Na2S.
- molar mass = 44.02 g; Therefore, 1 mol N2O = 44.02 g N2O = 6.022 × 1023 molecules N2O.
Take away
- Formula masses of ionic compounds and molecular masses of molecular compounds can be determined from the masses of the atoms in their formulas.
- The molar mass of an element can be found by referring to the atomic mass on a periodic table with units of grams.
- The molar mass of compounds can be determined by the molar masses of the atoms in their formulas.


