3.3: Alkylation and Acylation of Aromatic Rings - The Friedel-Crafts Reaction
- Page ID
- 500371
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)After completing this section, you should be able to
- write the equation and the detailed mechanism for a typical Friedel-Crafts alkylation, then identify the similarities between this reaction and the previous presented electrophilic aromatic substitution reactions.
- identify the product formed from the Friedel-Crafts alkylation of a given aromatic compound.
- identify the aromatic compound needed to prepare a given arene by a Friedel-Crafts alkylation.
- identify the alkyl halide and catalyst needed to form a specified arene from a given aromatic compound.
- show how alkyl halides and acyl-halides can be used as alkylating agents in Friedel-Crafts alkylation reactions.
- discuss the limitations of the Friedel-Crafts alkylation reaction, paying particular attention to the structure of the alkyl halide, the structure of the aromatic substrate, and the problem of polyalkylation.
- write an equation and the detailed mechanism for a typical Friedel-Crafts acylation.
- identify the product formed by the Friedel-Crafts acylation of a given aromatic compound.
- identify the aromatic compound, and the reagent and catalyst needed to prepare a given ketone through a Friedel-Crafts acylation reaction.
Friedel-Crafts Alkylation
Among the most useful electrophilic aromatic substitution reactions in the laboratory is alkylation—the introduction of an alkyl group onto the benzene ring. Called the Friedel–Crafts reaction after its founders in 1877, Charles Friedel and James Crafts, the reaction is carried out by treating an aromatic compound with an alkyl chloride, RCl, in the presence of AlCl3 to generate a carbocation electrophile, R+.
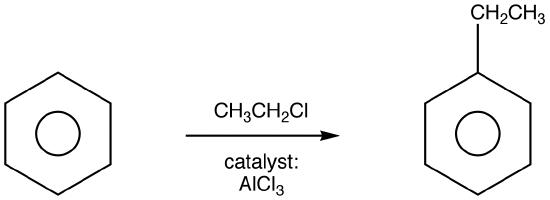
Aluminum chloride catalyzes the reaction by helping the alkyl halide to generate a carbocation in much the same way that FeBr3 catalyzes aromatic brominations by polarizing Br2.
The reactivity of haloalkanes increases as we move up the periodic table and increase polarity. This means that an RF haloalkane is most reactive followed by RCl then RBr and finally RI. This means that the Lewis acids used as catalysts in Friedel-Crafts Alkylation reactions tend to have similar halogen combinations, such as BF3, SbCl5, AlCl3, SbCl5, and AlBr3, all of which are commonly used in these reactions.
Mechanism
Step 1: An electron pair from the aromatic ring attacks the carbonation, forming a new C-C bond. The arenium ion intermediate results with stabilization from multiple resonance forms.
Steps 2: The loss of H+ completes the reaction giving the neutral alkylated substitution product (Figure \(\PageIndex{1}\)).
Some limitations of Friedel-Crafts Alkylation
Despite its utility, the Friedel–Crafts alkylation has several limitations. For one thing, only alkyl halides can be used. Aromatic (aryl) halides and vinylic halides don’t react because aryl and vinylic carbocations are too high in energy to form under Friedel–Crafts conditions.
Another limitation is that Friedel–Crafts reactions don’t succeed on aromatic rings that are substituted by a strongly electron-withdrawing group, which will deactivate the ring, such as carbonyl (C=O) or nitro group (NO2).
Adding to the list of substituents that will not allow the reaction to proceed are basic amino groups that can be protonated. The lone pair electrons on the amines react with the Lewis acid AlCl3. This places a positive charge next to the benzene ring, which is so strongly activating that the Friedel-Crafts reaction cannot occur.
We’ll see in the next section that the presence of a substituent group already on a ring can have a dramatic effect on that ring’s reactivity to further electrophilic substitution. Rings that contain any of the substituents listed in Figure \(\PageIndex{2}\) do not undergo Friedel–Crafts alkylation.
A third limitation to the Friedel–Crafts alkylation is that it’s often difficult to stop the reaction after a single substitution. Once the first alkyl group is on the ring, a second substitution reaction is facilitated for reasons we’ll discuss in the next section. Thus, we often observe polyalkylation. Reaction of benzene with 1 mol equivalent of 2-chloro-2-methylpropane, for example, yields p-di-tert-butylbenzene as the major product, along with small amounts of tert-butylbenzene and unreacted benzene. A high yield of mono-alkylation product is obtained only when a large excess of benzene is used.
A final limitation to the Friedel–Crafts reaction is that a skeletal rearrangement of the alkyl carbocation electrophile sometimes occurs during reaction, particularly when a primary alkyl halide is used. For instance, treating benzene with 1-chlorobutane at 0 °C gives an approximately 2 : 1 ratio of rearranged (sec-butyl) to unrearranged (butyl) products.
The carbocation rearrangements that accompany Friedel–Crafts reactions are like those that accompany electrophilic additions to alkenes and occur either by hydride shift or alkyl shift. For example, the relatively unstable primary butyl carbocation produced by reaction of 1-chlorobutane with AlCl3 rearranges to the more stable secondary butyl carbocation by the shift of a hydrogen atom and its electron pair (a hydride ion, H:–) from C2 to C1. Similarly, alkylation of benzene with 1-chloro-2,2-dimethylpropane yields (1,1-dimethylpropyl)benzene. The initially formed primary carbocation rearranges to a tertiary carbocation by shift of a methyl group and its electron pair from C2 to C1.
The Friedel–Crafts reaction of benzene with 2-chloro-3-methylbutane in the presence of AlCl3 occurs with a carbocation rearrangement. What is the structure of the product?
Strategy
A Friedel–Crafts reaction involves initial formation of a carbocation, which can rearrange by either a hydride shift or an alkyl shift to give a more stable carbocation. Draw the initial carbocation, assess its stability, and see if the shift of a hydride ion or an alkyl group from a neighboring carbon will result in increased stability. In the present instance, the initial carbocation is a secondary one that can rearrange to a more stable tertiary one by a hydride shift.

Use this more stable tertiary carbocation to complete the Friedel–Crafts reaction.
Solution

Friedel-Crafts Acylation
Just as an aromatic ring can be alkylated by reaction with an alkyl chloride, it can be acylated by reaction with a carboxylic acid chloride, RCOCl, in the presence of AlCl3. That is, an acyl group (–COR; pronounced a-sil) is substituted onto the aromatic ring. For example, reaction of benzene with acetyl chloride yields the ketone acetophenone.
Mechanism
The mechanism of Friedel–Crafts acylation is similar to that of Friedel–Crafts alkylation, and the same limitations on the aromatic substrate noted previously in Figure \(\PageIndex{2}\) for alkylation also apply to acylation. The reactive electrophile is a resonance-stabilized acyl cation, generated by reaction between the acyl chloride and AlCl3 (Figure \(\PageIndex{3}\)). As the resonance structures in the figure indicate, an acyl cation is stabilized by interaction of the vacant orbital on carbon with lone-pair electrons on the neighboring oxygen. Because of this stabilization, no carbocation rearrangement occurs during acylation.
Unlike the multiple substitutions that often occur in Friedel–Crafts alkylations, acylations never occur more than once on a ring because the product acylbenzene is less reactive than the starting material.
Aromatic alkylations occur in numerous biological pathways, although there is of course no AlCl3 present in living systems to catalyze the reaction. Instead, the carbocation electrophile is typically formed by dissociation of an organodiphosphate. The dissociation is usually assisted by complexation to a divalent metal cation such as Mg2+, just as the dissociation of an alkyl chloride is assisted by AlCl3.
An example of a biological Friedel–Crafts reaction occurs during the biosynthesis of phylloquinone, or vitamin K1, the human blood-clotting factor. Phylloquinone is formed by reaction of 1,4-dihydroxynaphthoic acid with phytyl diphosphate. Phytyl diphosphate first dissociates to a resonance-stabilized allylic carbocation, which then substitutes onto the aromatic ring in the typical way. Several further transformations lead to phylloquinone (Figure \(\PageIndex{4}\)).
Exercises
Which of the following will NOT undergo a rearrangement in a Friedel-Crafts reaction?
- Answer
-
A, B, and E will not undergo a rearrangement.
What is the major monosubstitution product from the Friedel–Crafts reaction of benzene with 1-chloro-2-methylpropane in the presence of AlCl3?
- Answer
-
tert-Butylbenzene
Suggest an acyl chloride that was used to make the following compounds:
- Answer
-
Make certain that you can define, and use in context, the key terms below.
- acyl group
- Friedel-Crafts acylation reaction
- Friedel-Crafts alkylation reaction
- polyalkylation
A Friedel-Crafts alkylation reaction is an electrophilic aromatic substitution reaction in which a carbocation is attacked by a pi bond from an aromatic ring with the net result that one of the aromatic protons is replaced by an alkyl group. If you prefer, you may regard these reactions as involving an attack by an aromatic ring on a carbocation. The latter approach is the one used in the textbook, but the former approach is probably more common.
When more than one alkyl group is introduced into an aromatic ring during the course of a Friedel-Crafts alkylation reaction, polyalkylation is said to have occurred.
The four limitations on the use of Friedel-Crafts alkylations are as follows:
- vinyl and aryl halides cannot be used to form carbocations.
- the aromatic substrate must not contain a strongly deactivating group, or groups, such as NH2, NHR or NR2, which form complexes with the Lewis acid catalyst and in so doing become strongly deactivating.
- polyalkylation, which can be overcome by using a large excess of the aromatic substrate.
- carbocation rearrangements may occur in any reaction that involves a carbocation.
The reaction of an aromatic substrate with an acid chloride (or acid anhydride) in the presence of an aluminum chloride catalyst is used to introduce an acyl group (C=O) into the aromatic ring through an electrophilic aromatic substitution mechanism. Such reactions are Friedel-Crafts acylation reactions.


