2.3: The Wave-Particle Duality of Matter
- Page ID
- 483510
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Extend the concept of wave-particle duality to electrons
Photons of light are individual packets of energy having characteristics of particles. This idea, that energy is concentrated in localized bundles, was a significant departure from the classical view that energy is spread out continuously as a wave. Our modern understanding of electronic structure builds on this concept by recognizing that not only does energy have particle-like properties, but matter also has wave-like properties.
The Wave Character of Electrons
In 1924, a young French physicist, Louis de Broglie (1892–1972; Nobel Prize, 1929), proposed that particles—such as electrons—could exhibit wave-like behaviour. He suggested that an electron could be described by a wave with a wavelength (λ) given by:
\[\lambda =\dfrac{h}{m u} \label{6.4.3} \]
where
- \(h\) is Planck’s constant,
- \(m\) is the mass of the particle, and
- \(u\) is the velocity of the particle.
Although you are not required to calculate de Broglie wavelengths in CHM 135H, this equation helps illustrate the concept that wavelength is a fundamental property of matter, not just electromagnetic radiation. Unlike the wavelengths discussed earlier in this chapter (which relate to electromagnetic waves), the de Broglie wavelength applies to physical particles and depends on velocity rather than frequency.
de Broglie also sought to explain why only certain orbits were allowed in Bohr’s model of the hydrogen atom. While Bohr assumed electrons followed fixed circular orbits, de Broglie suggested that an electron behaves as a standing wave around the nucleus. For the wave to be stable, only whole numbers of wavelengths could fit perfectly into the orbit (Figure \(\PageIndex{1}\)). In this model, the lowest energy level (n=1) corresponds to a wave that forms one complete wavelength around the nucleus. Higher energy levels contain more wavelengths, with nodes—regions where the amplitude is zero—appearing as n increases. As you will see, some of de Broglie’s ideas are retained in the modern theory of the electronic structure of the atom: the wave behavior of the electron and the presence of nodes that increase in number as the energy level increases. While de Broglie’s model helped explain why Bohr’s quantization worked, it was still incomplete. Later models showed that electrons do not move in fixed circular orbits.
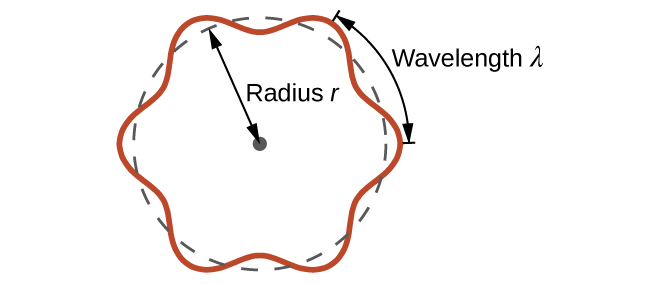
Figure \(\PageIndex{1}\): If an electron is viewed as a standing wave around the nucleus, an integer number of wavelengths must fit into the orbit for this standing wave behavior to be possible. The standing wave shown here corresponds to n = 6 with six wavelengths around the nucleus.
Shortly after de Broglie’s proposal, C. J. Davisson and L. H. Germer at Bell Laboratories provided experimental evidence for the wave-like nature of electrons. They observed interference patterns when electrons passed through a nickel crystal, a phenomenon typically seen with waves (Figure \(\PageIndex{2}\)). This confirmed that electrons exhibit wavelike behaviour.

Figure \(\PageIndex{2}\): The interference pattern for electrons passing through very closely spaced slits demonstrates that quantum particles such as electrons can exhibit wavelike behavior.
If electrons have wave properties, why hadn’t this been observed before? The answer lies in Planck’s constant (h), which is an extremely small number. For objects with large masses, such as a baseball or even a neutron, the de Broglie wavelength is so short (smaller than an atomic nucleus) that it is undetectable. However, for very small particles like electrons, the wavelength is significant, meaning they behave as both particles and waves. This principle is applied in electron microscopes, which use the short wavelength of electrons to resolve details far beyond what is possible with visible light (Figure \(\PageIndex{3}\)).
The Heisenberg Uncertainty Principle
Waves do not have a fixed position—they are spread out in space. If electrons behave as waves, it should be difficult to define their exact location at any given moment. This idea was formulated mathematically by the German physicist Werner Heisenberg (1901–1976; Nobel Prize in Physics, 1932).
Heisenberg’s Uncertainty Principle states that:
There is a fundamental limit to how precisely we can simultaneously know certain pairs of properties of a particle, such as its position and momentum.
For electrons, this means that the more accurately we know their position, the less accurately we can know their velocity (or energy), and vice versa. This uncertainty is not due to flaws in measurement but is an inherent property of quantum systems. Here is a mathematical description of the Heisenberg uncertainty principle if you're interested.
Bohr’s model of the hydrogen atom violated Heisenberg’s principle by assuming that electrons follow precise circular orbits with both a known radius (position) and energy. Given the wave-like nature of electrons, this is impossible. Modern atomic models replace fixed orbits with probability distributions, where an electron’s location is described in terms of likelihood rather than a definite path.
Summary
Electrons exhibit both particle and wave properties, a concept known as wave-particle duality. Louis de Broglie proposed that electrons have wavelengths, helping to explain Bohr’s quantized orbits in terms of standing waves. Experimental confirmation came from Davisson and Germer, who showed that electrons create interference patterns just like waves.
However, Bohr’s model incorrectly described electrons as moving in fixed circular orbits. The Heisenberg Uncertainty Principle showed that it is impossible to precisely determine both an electron’s position and momentum simultaneously.
Contributors and Attributions
Modified by Joshua Halpern (Howard University)

