11.9: Addition of Water to Alkynes
- Page ID
- 28238
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)After completing this section, you should be able to
- write the equation for the reaction of water with an alkyne in the presence of sulfuric acid and mercury(II) sulfate.
- describe keto-enol tautomerism.
- predict the structure of the ketone formed when a given alkyne reacts with sulfuric acid in the presence of mercury(II) sulfate.
- identify the reagents needed to convert a given alkyne to a given ketone.
- identify the alkyne needed to prepare a given ketone by hydration of the triple bond.
- write an equation for the reaction of an alkyne with borane.
- write the equation for the reaction of a vinylic borane with basic hydrogen peroxide or hot acetic acid.
- identify the reagents, the alkyne, or both, needed to prepare a given ketone or a given cis alkene through a vinylic borane intermediate.
- identify the ketone produced when a given alkyne is reacted with borane followed by basic hydrogen peroxide.
- identify the cis alkene produced when a given alkyne is reacted with borane followed by hot acetic acid.
- explain why it is necessary to use a bulky, sterically hindered borane when preparing vinylic boranes from terminal alkynes.
- predict the product formed when the vinylic borane produced from a terminal alkyne is treated with basic hydrogen peroxide.
- identify the alkyne needed to prepare a given aldehyde by a vinylic borane.
Make certain that you can define, and use in context, the key terms below.
- enol
- keto-enol tautomeric equilibrium
- tautomerism
- tautomers
Rapid interconversion between tautomers is called tautomerism; however, as the two tautomers are in equilibrium, the term tautomeric equilibrium may be used. This section demonstrates the equilibrium between a ketone and an enol; hence, the term keto-enol tautomeric equilibrium is appropriate. The term “enol” indicates the presence of a carbon-carbon double bond and a hydroxyl (i.e., alcohol) group. Later in the course, you will see the importance of keto-enol tautomerism in discussions of the reactions of ketones, carbohydrates and nucleic acids.
It is important to note that tautomerism is not restricted to keto-enol systems. Other examples include imine-enamine tautomerism
and nitroso-oxime tautomerism
However, at the moment you need only concern yourself with keto-enol tautomerism.
Notice how hydroboration complements hydration in the chemistry of both alkenes and alkynes.
Mercury(II)-Catalyzed Hydration of Alkynes
As with alkenes, hydration (addition of water) of alkynes requires a strong acid, usually sulfuric acid, and is facilitated by the mercuric ion (Hg2+). However, the hydration of alkynes gives ketone products while the hydration of alkenes gives alcohol products. Notice that the addition of oxygen in both reactions follows Markovnikov rule.
During the hydration of an alkyne, the initial product is an enol intermediate (a compound having a hydroxyl substituent attached to a double-bond), which immediately rearranges to the more stable ketone through a process called enol-keto tautomerization.
Tautomers are defined as rapidly inter-converted constitutional isomers, usually distinguished by a different bonding location for a labile hydrogen atom and a differently located double bond. The keto and enol tautomers are in equilibrium with each other and with few exceptions the keto tautomer is more thermodynamically stable and therefor favored by the equilibrium. This mechanism for tautomerization will be discussed in greater detail in Section 22-1.
General Reaction
For terminal alkynes, the addition of water follows the Markovnikov rule, and the final product is a methyl ketone. For internal alkynes the addition of water is not regioselective. Hydration of symmetrical internal alkynes produces a single ketone product. However, hydration of asymmetrical alkynes, (i.e. if R & R' are not the same ) produces two isomeric ketone products.
Mechanism
The mechanism starts with the eletrophilic addition of the mercuric ion (Hg2+) to the alkyne producing a mercury-containing vinylic carbocation intermediate. Nucleophilic attack of water on the vinylic carbocation forms a C-O bond to produce a protonated enol. Deprotonation of the enol by water then produces a organomurcury enol. The murcury is substituted with H+ to produced a neutral enol and regenerate the Hg2+ catalyst. The enol is converted to the ketone product through keto-enol tautomerization the mechanism of which is provided in Section 22-1.
1) Electrophilic addition of Mg2+
2) Nucleophilic attack by water
3) Deprotonation
4) Substitution
5) Tautomerization
Hydroboration–Oxidation of Alkynes
The hydroboration-oxidation of alkynes is analagous to the reaction with alkenes. However, where alkenes for alcohol products, alkynes for aldehyde or ketone products. In both cases the addition is anti-Markovnikov and an oxygen is placed on the less alkyl substituted carbon. With the hydroboration of an alkyne the presences of a second pi bond allows the initial product to under tautomerization to become the final aldehyde product.
Alkynes have two pi bonds both of which are capable of reacting with borane (BH3). To limit the reactivity to only one alkyne pi bond, a dialkyl borane reagent (R2BH) is used. Replacing two of the hydrogens on the borane with alkyl groups also creates steric hindrance which enhances the anti-Markovnikov regioselective of the reaction. Disiamylborane (Sia2BH) and 9-borabicyclo[3.3.1]nonane (9-BBN) are two common reagents for this hydroboration reaction. The oxidation reagents (a basic hydrogen peroxide solution) are the same for both alkenes and alkynes.
General Reaction
The hydroboration of terminal alkynes produces aldehyde products while internal alkynes produce ketone products. The hydroboration of symmetrical alkynes produces one ketone product and asymmetrical alkynes produce a mixture of product ketones.
Mechanism
The mechanism starts with the electrophilic addition of the B-H bond of the borane. The hydrogen atom and the borane all on the same side of the alkyne creating a syn addition configuration in the alkene poduct. Also, the addition is anti-Markovnikov regioselective which means the borane adds to the less substituted carbon of the alkyne and the hydrogen atom adds to the more substituted. The oxidative work-up replaces the borane with a hydroxy group (-OH) creating an enol intermediate. The enol immediately tautomerizes to the product aldehyde for terminal alkynes and the product ketone for internal alkynes.
Comparison of Mercury(II)-Catalyzed Hydration and Hydroboration–Oxidation of Alkynes
These two reactions are complementary for the reaction of a terminal alkyne because the produce distinctly different products. The mercury(II) catalyzed hydration of a terminal alkyne produces a methyl ketone, while the hydroboration-oxidation produces an aldehyde.
For internal alkynes, the regioslectivity of these reactions are rendered ineffective. The reactions are redundant in that they both produce the same ketone products.
1) Draw the structure of the product formed when each of the substances below is treated with H2O/H2SO4 in the presence of HgSO4.
a)
b)
2) Draw the structure of the keto form of the compound shown below. Which form would you expect to be the most stable?
3) What alkyne would you start with to gain the following products?
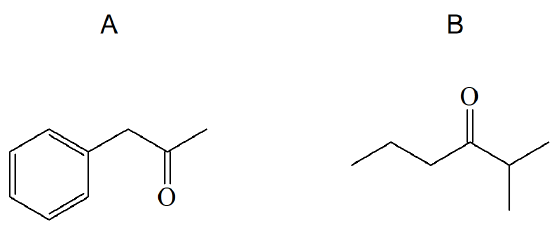
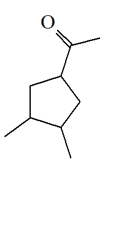
4) What alkyne would you start with to gain the following product?
5) Draw the product(s) of the following reactions:
- Answer
-
Answers:
1)
a)
b)
2.
The keto form should be the most stable.
3)
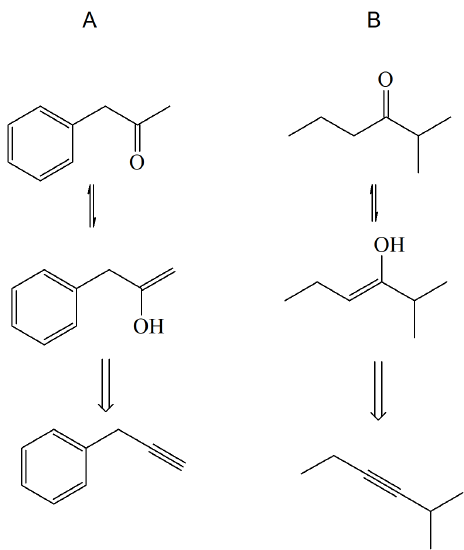
4)
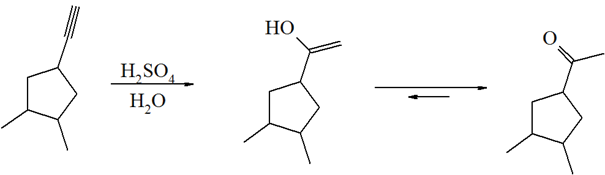
5)
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry


