12.12: Oxidation of Alcohols
- Page ID
- 28253
This page looks at the oxidation of alcohols using acidified sodium or potassium dichromate(VI) solution. This reaction is used to make aldehydes, ketones and carboxylic acids, and as a way of distinguishing between primary, secondary and tertiary alcohols.
Oxidizing the different types of alcohols
The oxidizing agent used in these reactions is normally a solution of sodium or potassium dichromate(VI) acidified with dilute sulfuric acid. If oxidation occurs, the orange solution containing the dichromate(VI) ions is reduced to a green solution containing chromium(III) ions. The electron-half-equation for this reaction is
Cr2O72- + 14 H+ → 2 Cr3+ + 7 H2O
Oxidizing agents
- K2Cr2O7 potassium dichromate
- CrO3 chromium trioxide
Both of these are used along with H2SO4, H2O
- 1o alcohol → Carboxylic acid
- 2o alcohol → Ketone
- 3o alcohol → No reaction
Primary alcohols
Primary alcohols can be oxidized to either aldehydes or carboxylic acids depending on the reaction conditions. In the case of the formation of carboxylic acids, the alcohol is first oxidized to an aldehyde which is then oxidized further to the acid.
Full oxidation to carboxylic acids
You need to use an excess of the oxidizing agent and make sure that the aldehyde formed as the half-way product stays in the mixture. The alcohol is heated under reflux with an excess of the oxidizing agent. When the reaction is complete, the carboxylic acid is distilled off. The full equation for the oxidation of ethanol to ethanoic acid is:
3 CH3CH2OH + 2 Cr2O72- + 16 H+ → 3 CH3COOH + 4 Cr3+ + 11 H2O
The more usual simplified version looks like this:
CH3CH2OH + 2[O] → CH3COOH + H2O
Alternatively, you could write separate equations for the two stages of the reaction - the formation of ethanal and then its subsequent oxidation.
CH3CH2OH + [O] → CH3CHO + H2O
CH3CHO + [O] → CH3COOH
This is what is happening in the second stage:

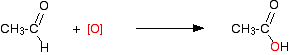
Secondary alcohols
Secondary alcohols are oxidized to ketones - and that's it. For example, if you heat the secondary alcohol propan-2-ol with sodium or potassium dichromate(VI) solution acidified with dilute sulfuric acid, you get propanone formed. Playing around with the reaction conditions makes no difference whatsoever to the product. Using the simple version of the equation and showing the relationship between the structures:

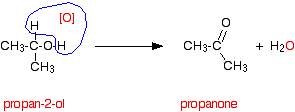
If you look back at the second stage of the primary alcohol reaction, you will see that an oxygen "slotted in" between the carbon and the hydrogen in the aldehyde group to produce the carboxylic acid. In this case, there is no such hydrogen - and the reaction has nowhere further to go.
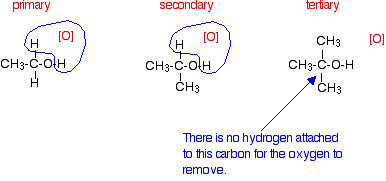
Tertiary alcohols
Tertiary alcohols are not oxidized by acidified sodium or potassium dichromate(VI) solution - there is no reaction whatsoever. If you look at what is happening with primary and secondary alcohols, you will see that the oxidizing agent is removing the hydrogen from the -OH group, and a hydrogen from the carbon atom attached to the -OH. Tertiary alcohols don't have a hydrogen atom attached to that carbon.
You need to be able to remove those two particular hydrogen atoms in order to set up the carbon-oxygen double bond.
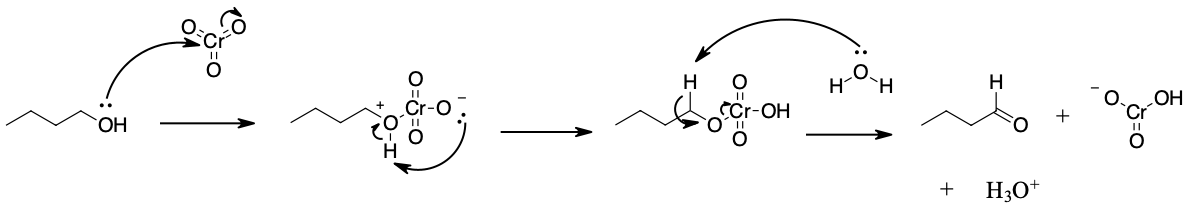
Mechanism
Examples
Using these reactions as a test for the different types of alcohol
First you have to be sure that you have actually got an alcohol by testing for the -OH group. You would need to show that it was a neutral liquid, free of water and that it reacted with solid phosphorus(V) chloride to produce a burst of acidic steamy hydrogen chloride fumes. You would then add a few drops of the alcohol to a test tube containing potassium dichromate(VI) solution acidified with dilute sulfuric acid. The tube would be warmed in a hot water bath.
Picking out the tertiary alcohol
In the case of a primary or secondary alcohol, the orange solution turns green. With a tertiary alcohol there is no color change. After heating:
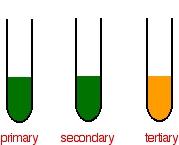
Distinguishing between the primary and secondary alcohols
You need to produce enough of the aldehyde (from oxidation of a primary alcohol) or ketone (from a secondary alcohol) to be able to test them. There are various things which aldehydes do which ketones don't. These include the reactions with Tollens' reagent, Fehling's solution and Benedict's solution, and are covered on a separate page.
These tests can be a bit of a bother to carry out and the results are not always as clear-cut as the books say. A much simpler but fairly reliable test is to use Schiff's reagent. Schiff's reagent is a fuchsin dye decolorized by passing sulfur dioxide through it. In the presence of even small amounts of an aldehyde, it turns bright magenta.
It must, however, be used absolutely cold, because ketones react with it very slowly to give the same color. If you heat it, obviously the change is faster - and potentially confusing. While you are warming the reaction mixture in the hot water bath, you can pass any vapours produced through some Schiff's reagent.
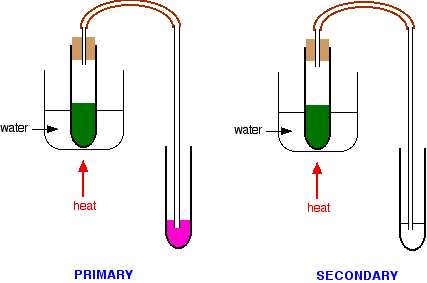
- If the Schiff's reagent quickly becomes magenta, then you are producing an aldehyde from a primary alcohol.
- If there is no color change in the Schiff's reagent, or only a trace of pink color within a minute or so, then you are not producing an aldehyde, and so haven't got a primary alcohol.
Because of the color change to the acidified potassium dichromate(VI) solution, you must therefore have a secondary alcohol. You should check the result as soon as the potassium dichromate(VI) solution turns green - if you leave it too long, the Schiff's reagent might start to change color in the secondary alcohol case as well.
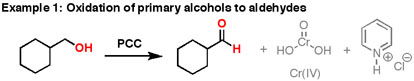
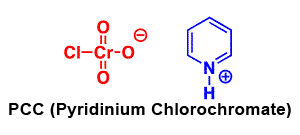
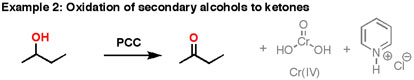
Formation of Aldehydes using PCC
Pyridinium chlorochromate (PCC) is a milder version of chromic acid.
PCC oxidizes alcohols one rung up the oxidation ladder, from primary alcohols to aldehydes and from secondary alcohols to ketones. Unlike chromic acid, PCC will not oxidize aldehydes to carboxylic acids. Similar to or the same as: \(CrO_3\) and pyridine (the Collins reagent) will also oxidize primary alcohols to aldehydes. Here are two examples of PCC in action.
- If you add one equivalent of PCC to either of these alcohols, you obtain the oxidized version. The byproducts (featured in grey) are Cr(IV) as well as pyridinium hydrochloride.
- One has to be careful with the amount of water present in the reaction. If water were present, it can ad to the aldehyde to make the hydrate, which could be further oxidized by a second equivalent of PCC were it present. This is not a concern with ketones, since there is no H directly bonded to C.
How does it work? Oxidation reactions of this sort are actually a kind of elimination reaction. We’re going from a carbon-oxygen single bond to a carbon-oxygen double bond. The elimination reaction can occur because we’re putting a good leaving group on the oxygen, namely the chromium, which will be displaced when the neighboring C-H bond is broken with a base.
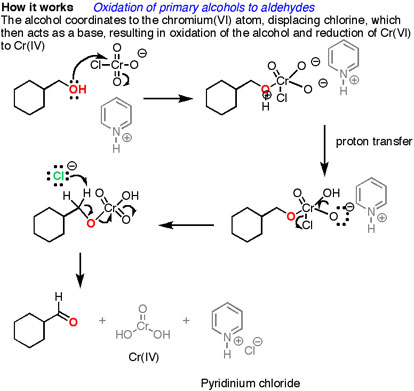
The first step is attack of oxygen on the chromium to form the Cr-O bond. Secondly, a proton on the (now positive) OH is transferred to one of the oxygens of the chromium, possibly through the intermediacy of the pyridinium salt. A chloride ion is then displaced, in a reaction reminiscent of a 1,2 elimination reaction, to form what is known as a chromate ester.
The C-O double bond is formed when a base removes the proton on the carbon adjacent to the oxygen. [aside: I've drawn the base as Cl(-) although there are certainly other species which could also act as bases here (such as an alcohol). It is also possible for pyridine to be used as the base here, although only very low concentrations of the deprotonated form will be present under these acidic conditions.] The electrons from the C-H bond move to form the C-O bond, and in the process break the O-Cr bond, and Cr(VI) becomes Cr(IV) in the process (drawn here as O=Cr(OH)2 ).
Real life notes: If you end up using PCC in the lab, don’t forget to add molecular sieves or Celite or some other solid to the bottom of the flask, because otherwise you get a nasty brown tar that is a real major pain to clean up. The toxicity and mess associated with chromium has spurred the development of other alternatives like TPAP, IBX, DMP, and a host of other neat reagents you generally don’t learn about until grad school.
Prof. Steven Farmer (Sonoma State University)
James Ashenhurst (MasterOrganicChemistry.com)
Jim Clark (Chemguide.co.uk)