6: Magnetism and Colors in Coordination Complexes (Worksheet)
- Page ID
- 81813
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Name: ______________________________
Section: _____________________________
Student ID#:__________________________
Work in groups on these problems. You should try to answer the questions without referring to your textbook. If you get stuck, try asking another group for help.
Introduction
According to Crystal Field Theory, the interaction between a transition metal and ligands arises from the attraction between the positively charged metal cation and negative charge on the non-bonding electrons of the ligand. The theory is developed by considering energy changes of the five degenerate d-orbitals upon being surrounded by an array of point charges consisting of the ligands. As a ligand approaches the metal ion, the electrons from the ligand will be closer to some of the d-orbitals and farther away from others, causing a loss of degeneracy.
Q1: Diagrams
Below draw the d-orbital splitting diagrams for an octahedral ligand field, a tetrahedral ligand field and a square planar ligand field. For the octahedral and tetrahedral fields, label the \(t_{2g}\) and \(e_g\) orbitals and identify \(\Delta _o\) (for octahedral field) and \(\Delta _t\) (for the tetrhedral field). Identify which specific orbitals are stabilized and which are destabilized.
Magnitude of Splitting Varies
The electrons in the d-orbitals and those in the ligand repel each other due to repulsion between like charges. Thus the d-electrons closer to the ligands will have a higher energy than those further away which results in the d-orbitals splitting in energy. This splitting is affected by the following factors:
- the nature of the metal ion.
- the metal's oxidation state. A higher oxidation state leads to a larger splitting.
- the arrangement of the ligands around the metal ion.
- the nature of the ligands surrounding the metal ion. The stronger the effect of the ligands then the greater the difference between the high and low energy d groups.
A ligands can be placed in order from those producing the smallest to the largest d-orbital splitting in coordination compounds. This ordering of ligands is called a Spectrochemical Series:
|
I- < Br- < SCN- < Cl- < F- ≤ OH- , ONO- < OH2 < NCS- < NCCH3 < NH3 , py < NO2- < CN- , NO , CO |
|
| weak-field ligands | strong-field ligands |
Magnetism in Coordination Complexes
Ligands which cause a larger splitting of the d-orbitals are referred to as strong-field ligands, such as \(\ce{CN^{−}}\) and \(\ce{CO}\) from the spectrochemical series. In complexes with these ligands, it is unfavourable to put electrons into the high energy orbitals. Therefore, the lower energy orbitals are completely filled before population of the upper sets starts according to the Aufbau principle. Complexes such as this are called "low spin". For example, NO2− is a strong-field ligand and produces a large \(Δ_o\). The octahedral ion [Fe(NO2)6]3−, which has 5 d-electrons, would have the octahedral splitting diagram shown at right with all five electrons in the t2g level. The low spin state therefore does not follow Hund's rule.
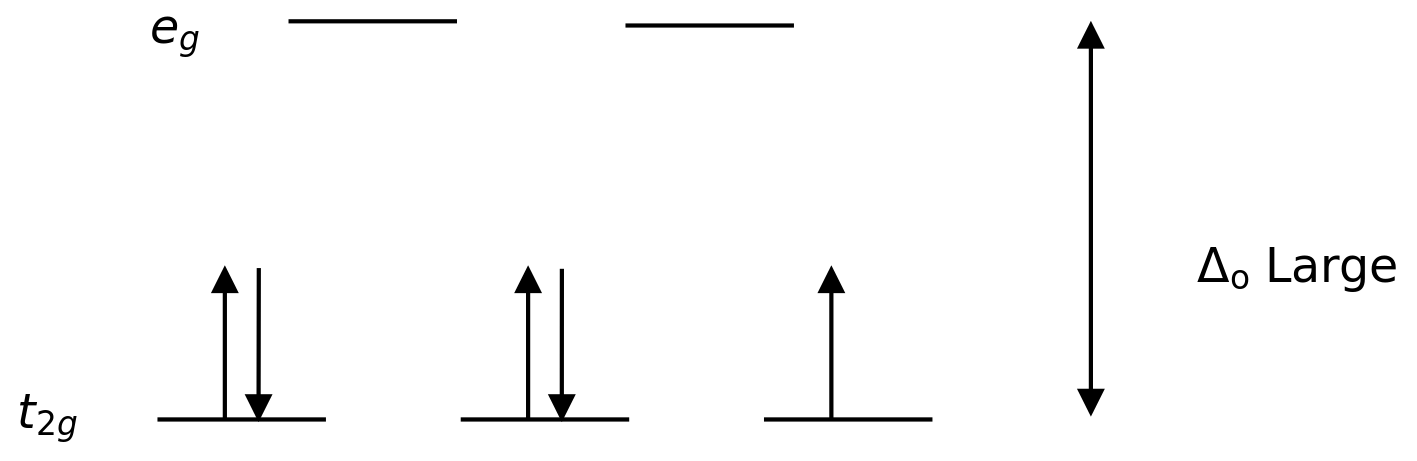
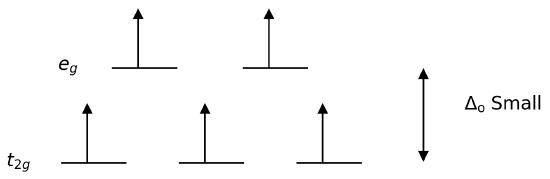
Conversely, ligands (like I− and Br−) which cause a small splitting Δ of the d-orbitals are referred to as weak-field ligands. In this case, it is easier to put electrons into the higher energy set of orbitals than it is to put two into the same low-energy orbital, because two electrons in the same orbital repel each other. So, one electron is put into each of the five d-orbitals before any pairing occurs in accord with Hund's rule and "high spin" complexes are formed. For example, Br− is a weak-field ligand and produces a small \(Δ_o\). So, the ion [FeBr6]3−, again with five d-electrons, would have an octahedral splitting diagram where all five orbitals are singly occupied.
The difference between strong- and weak-field, or low and high- spin cases comes down to the magnitude of the crystal field splitting energy
- Geometry is one factor, \(Δ_o\) is large than \(Δ_t\). In almost all cases, the \(Δ_t\) is smaller than P (pairing energy), so tetrahedral complexes are always weak-field (high spin). Square planar complexes may either be weak- or strong-field.
- Oxidation State of Metal Cation – A greater charge on cation results in a greater magnitude of \(Δ_o\). A greater charge pulls ligands more strongly towards the metal, therefore influences the splitting of the energy levels more.
- Size of the Metal Cation – For second and third-row transition metal ions, \(Δ_o\) is larger. Less steric hindrance between the ligands – better overlap w/orbitals.
In order for low spin splitting to occur, the energy cost of placing an electron into an already singly occupied orbital must be less than the cost of placing the additional electron into an eg orbital at an energy cost of Δ.
There are three type of magnetism were are addressing in this class:
- Ferromagnetism occurs in certain materials (such as iron) form permanent magnets. This is a bulk property associated with aligned domain in a material.
- Paramagnetism occurs in materials that are weakly attracted by an externally applied magnetic field.
- Diamagnetism occurs in materials that are weakly repeal by an externally applied magnetic field.
For a coordination complex to be paramagentic is needs unpaired electrons. The more unpaired electrons, the greater the paramagnetic behavior. If all electrons are paired, then the complex is diamagnetic. Ignore ferromagnetism for this worksheet
Q2
Sketch the d-orbital energy level diagrams for \(\ce{[Fe(H2O)6]^{3+}}\) and \(\ce{[Fe(CN)6]^{3–}}\) and predict the number of unpaired electrons in each.
Q3
The \(\ce{[CoF6]^{3-}}\) ion is paramagnetic and the \(\ce{[Co(CN)6]^{3-}}\) ion is diamagnetic. Explain this observation. Is the \(\ce{F^{-}}\) ligand inducing a strong or weak field?
Q4
\(\ce{[PdCl4]^{-2}}\) is found to be diamagnetic. Is this structure tetrahedral or square planar according to crystal field theory? Draw the energy orbital diagram.
Q5
Would each of the following complexes be expected to be high-spin or low spin, \(\ce{[MnCl4]^{2–}}\) (tetrahedral) and \(\ce{[FeCl6]^{3–}}\) ? Justify your answer using orbital energy diagrams from Q1.
Colors in Coordination Complexes
If you pass white light through a prism it splits into all the colors of the rainbow. Visible light is simply a small part of an electromagnetic spectrum most of which we cannot see - gamma rays, X-rays, infra-red, radio waves and so on. Each of these has a particular wavelength, ranging from 10-16 meters for gamma rays to several hundred meters for radio waves. Visible light has wavelengths from about 400 to 750 nm. (1 nanometer = 10-9 meters.). Transition metal ions show many intense colors in host crystals or solution The color of light absorbed by the complex ion is related to electronic energy changes in the structure of the complex the electron “leaping” from a lower energy state to a higher energy state. The observed color is complementary color of one that is absorbed (Figure \(\PageIndex{2}\)).
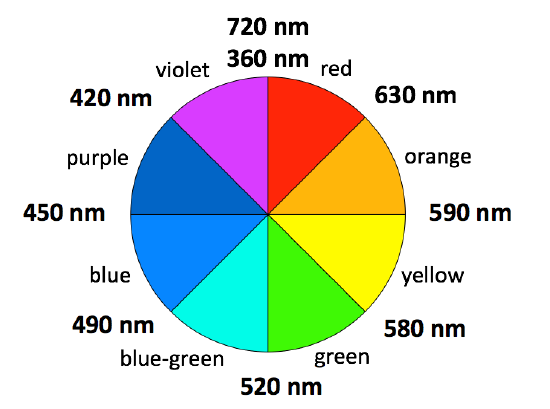
The colors directly opposite each other on the color wheel are said to be complementary colors. Blue and orange are complementary colors; red and blue-green (i.e. cyan) are complementary; and so are green and magenta.
For example, if white light (ordinary sunlight, for example) passes through copper(II) sulfate solution, some wavelengths in the light are absorbed by the solution. Copper(II) ions in solution absorb light in the red region of the spectrum. The light which passes through the solution and out the other side will have all the colors in it except for the red. We see this mixture of wavelengths as pale blue (cyan). The diagram gives an impression of what happens if you pass white light through copper(II) sulfate solution. If you arrange some colors in a circle, you get a "color wheel".
When white light is passed through a solution of this ion, some of the energy in the light is used to promote an electron from the lower set of orbitals into a space in the upper set. Due to electronic transitions between split d sublevel orbitals. The wavelength of maximum absorbance can be used to determine the size of the energy gap between split d sublevel orbitals
\[E_{photon} = h\nu = \dfrac{hc}{\lambda} = \Delta_o \nonumber\]
where Planck Constant (h) is \(6.6262 \times 10^{-34}\, J·s\) and the speed of light in a vacuum (c) is \(3.00 \times 10^8 m/s\).
Q6
The \(\ce{[Cr(Cl)6]^{-3}}\) complex ion has a maximum absorption spectrum at 735 nm. Calculate the crystal field spitting energy in J/photon and kJ/mol.
Q7
Which of the following complex ions is expected to absorb light of the longest wavelength: \(\ce{[Cr(H2O)6]^{3+}}\), \(\ce{[Cr(en)3]^{3+}}\), or \(\ce{[Cr(CN)6]^{3–}}\)? Which would absorb the shortest wavelength? Explain.
Q8
The five following hexaaqua metal complex ions are not colored. For each complex, explain why.
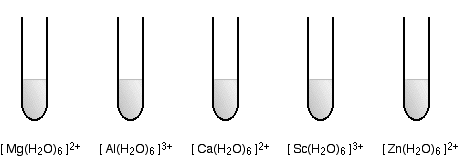
Q9
The \(\ce{[CoA6]^{2+}}\) complex is red to the eye and the \(\ce{[CoB6]^{2+}}\) complex is green to the eye. Which ligand (\(\ce{A}\) or \(\ce{B}\)) produces the larger crystal field splitting, \(∆_o\)? Both ligands are neutral. Explain your answer. Why does the charge on the ligand matter (if at all)?
Q10
Referring to the ligands in the previous problem, which complex would be expected to be more easily oxidized: \(\ce{[CoA6]^{2+}}\) or \(\ce{[CoB6]^{2+}}\)? Think about the similarity of oxidation and ionization energy.
Q11
Three complex ions of cobalt (III) absorb light at wavelengths at 290 nm, 440 nm, 770 nm. Match each complex ion to the appropriate wavelength and predict the color you would expect each solution to be. Refer to the color wheel for complements, 400 nm to 700 nm is visible. Choices:
- \(\ce{[Co(CN)6]^{-3}}\)
- \(\ce{[CoF6]^{-3}}\)
- \(\ce{[Co(NH3)6]^{+3}}\)
Solve for the crystal field splitting energy in kJ/mol for \(\ce{[Co(NH3)6]^{+3}}\)


