2.7: The Carbonyl Group
- Page ID
- 364136
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Let’s look at the electron transitions of H2CO, formaldyhyde, for a typical description of the MO of an organic molecule.

Pretty valence-bond theory
(first year description of formaldehyde): https://www.chemtube3d.com/orbitalsformaldehyde
View 1: Before (but after hybridization)
Carbon and the oxygen are both \(\mathrm{sp}^{2}\) hybridized.
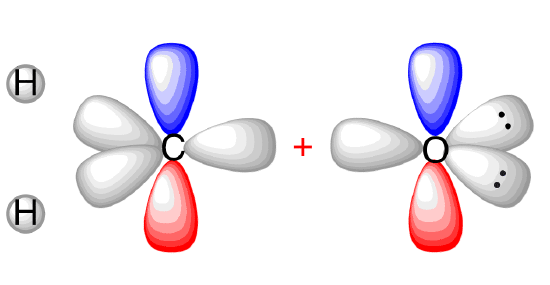
(CC BY-SA-NC; Nick Graves via ChemTube3D)
View 2: Moved together to demonstrate overlap
When moved together, there is overlap of the \(\pi\) and \(\mathrm{sp}^{2}\) orbitals.

(CC BY-SA-NC; Nick Graves via ChemTube3D)
View 3: Generation of new bonds to make the carbonyl bond)
Formation of bonding interactions to form the sigma bond (from the \(\mathrm{sp}^{2}\) orbitals on \(\ce{C}\) and \(\ce{O}\) AND an \(\pi\) bond from the two non-hybridized \(p\) orbitals on \(\ce{C}\) and \(\ce{O}\).
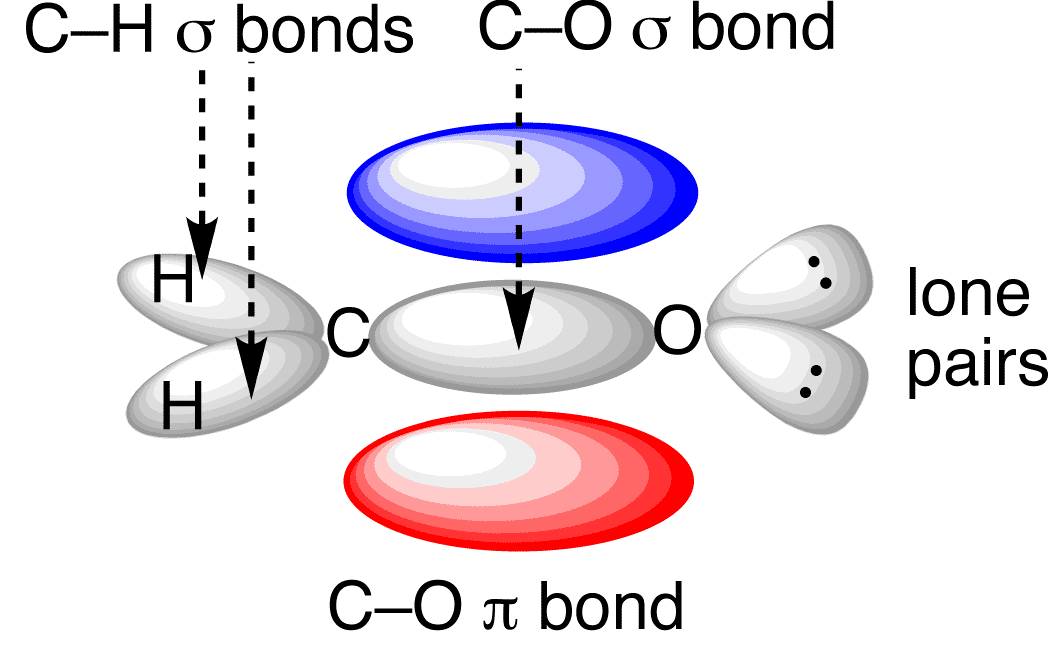.png?revision=1&size=bestfit&width=337&height=207)
(CC BY-SA-NC; Nick Graves via ChemTube3D)
H2CO is a planar molecule with \(C_{2v}\) symmetry. We ignore the n(1s) core electrons on C and O as well as the \(σ\) electrons in the C-H bonds. Very low energy excitations will not be involved in the UV-VIS region. The ground state valence electronic configuration of interest is:
\[n_{a}^{2} \sigma^{2} \pi^{2} n_{b}^{2}\left(\pi^{*}\right)^{0}\left(\sigma^{*}\right)^0 \nonumber \]
This is the lowest energy MO’s that originate from the valence atomic orbitals of the \(\ce{C}\) and \(\ce{O}\).
What do they look like? http://pubs.acs.org/doi/pdf/10.1021/ed050p400

Figure XX: http://pubs.acs.org/doi/pdf/10.1021/ed050p400. All Rights Reserved ACS
The MO energies are
\[n_{a}<\sigma<\pi<n_{b}<\pi^{*}<\sigma^{*} \nonumber \]
from a simple MO calculation. \(n < \sigma\) because \(2 s \ll 2p\). Let's ignore the lowest non-bodning state and focus on the "frontier orbitals" - the higher lying MOs.
Possible Electronic Transitions
There are six outer valence e- are involved in major electronic transitions. The lowest energy transition is a \(n \rightarrow \pi^{*}\) transition, while \(\pi \rightarrow \pi^{*}\) and \(n_a \rightarrow \sigma^{*}\) occur at higher \(ΔE\) and shorter \(λ\).

Energy levels of molecular orbitals in formaldehyde.
Molecular orbitals of formaldehyde emphasizing the transitions (after Guillet [19]).

In addition to change in the orbital type, the excited electron may keep the same \(m_s\), spin quantum number producing a singlet excited state. If \(m_s\) changes during the transition, the excited state had two ↑↑ electrons and is a triplet state. For instance, the \(n \rightarrow \pi^{*}\) transition can lead to a \(^1\left(n_{1} \pi^{*}\right)\) if the \(m_s\) does not change, but to \(^3\left(n_{1} \pi^{*}\right)\) if it does change (flip). Both the singlet and triplet states have the same configuration, but lies lower in energy because ↑↑ electrons are on average further apart than ↓↑ electrons (reducing e-e correlation).
This MO description is oversimplified. e-e interactions are different in an excited state than in the ground state. So A single configuration description is not very good; however this descriptions can be improved by allowing configurations to exit together to minimize the energy of the excited state (e.g. CI-doubles, CI-triples). We will discuss this later.

