18.11: Interchapter- A Preview of Carbonyl Chemistry
- Last updated
- Save as PDF
- Page ID
- 429667
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
So far, in our survey of the oxygen-containing organic compounds, we have studied the chemistry of ethers and alcohols. Now we turn our attention to a much larger group of compounds—those containing a carbonyl group:
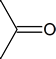
The variety of carbonyl compounds is so wide, and their chemistry so extensive, that we shall need the next five chapters to discuss them thoroughly. One advantage of this course is that we have divided up the discussion of carbonyl compounds so that this large amount of factual material can be studied in small, readily digestible blocks.
The purpose of this chapter is to provide a general outline of what is to follow in the next five chapters: the various types of carbonyl compounds are introduced, the nature of the carbonyl group is explained, and the four most common mechanisms by which carbonyl groups react are described in general terms.
Chapter Objectives
When you have completed this chapter, you should be able to
- fulfill all of the detailed objectives listed under each individual section.
- identify the various types of carbonyl compounds.
- write the mechanism of each of the four general types of reactions discussed in the chapter.
- define, and use in context, the key terms introduced in this chapter.
Introduction
Objectives
After completing this section, you should be able to give examples of the wide variety of biologically important, pharmaceutical and industrial compounds that contain one or more carbonyl groups.
Key Terms
Make certain that you can define, and use in context, the key term below.
- carbonyl group
Study Notes
A “carbonyl group” consists of an sp2‑hybridized carbon atom that is joined to an oxygen atom by a double bond.
Natural Occurrence of Aldehydes and Ketones
Aldehydes and ketones are widespread in nature and are often combined with other functional groups. Examples of naturally occurring molecules which contain a aldehyde or ketone functional group are shown in the following two figures. The compounds in the figure 1 are found chiefly in plants or microorganisms and those in the figure 2 have animal origins. Many of these molecular structures are chiral.
When chiral compounds are found in nature they are usually enantiomerically pure, although different sources may yield different enantiomers. For example, carvone is found as its levorotatory (R)-enantiomer in spearmint oil, whereas, caraway seeds contain the dextrorotatory (S)-enantiomer. In this case the change of the stereochemistry causes a drastic change in the perceived scent. Aldehydes and ketones are known for their sweet and sometimes pungent odors. The odor from vanilla extract comes from the molecule vanillin. Likewise, benzaldehyde provides a strong scent of almonds and is this author’s favorite chemical smell. Because of their pleasant fragrances aldehyde and ketone containing molecules are often found in perfumes. However, not all of the fragrances are pleasing. In particular, 2-heptanone provides part of the sharp scent from blue cheese and (R)-Muscone is part of the musky smell from the Himalayan musk deer. Lastly, ketones show up in many important hormones such as progesterone (a female sex hormone) and testosterone (a male sex hormone). Notice how subtle differences in structure can cause drastic changes in biological activity. The ketone functionality also shows up in the anti-inflammatory steroid, Cortisone.
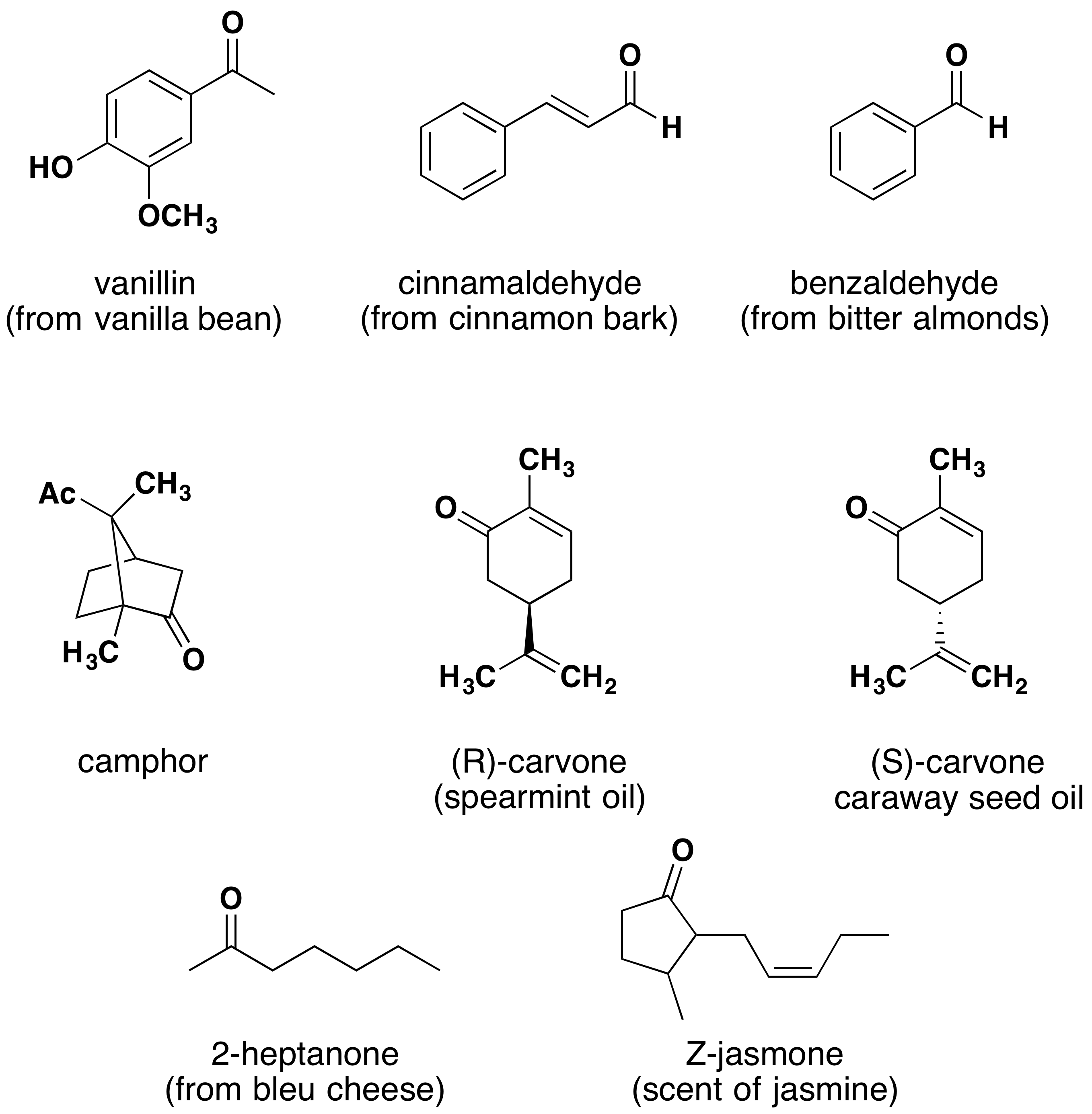
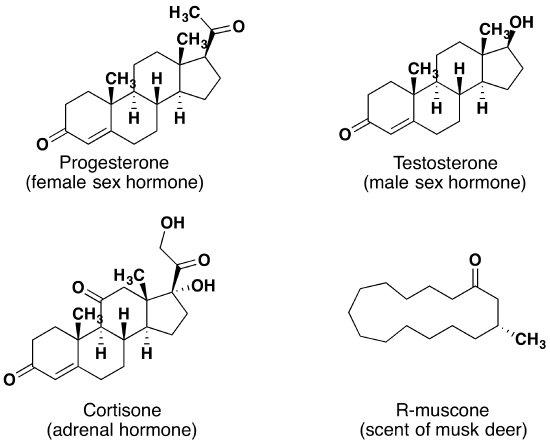
I Kinds of Carbonyl Compounds
Objectives
After completing this section, you should be able to
- identify the following types of compounds as containing carbonyl groups: aldehydes, ketones, carboxylic acids, acid chlorides, acid anhydrides, esters, lactones, amides and lactams.
- identify the important difference between aldehydes and ketones, and the other types of compounds listed under Objective 1, above.
Key Terms
Make certain that you can define, and use in context, the key term below.
- acyl fragment or acyl group
Study Notes
The idea of an “acyl group”
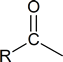
is not really new. Recall that in Chapter 16 we discussed the introduction of acyl groups into benzene rings by means of the Friedel‑Crafts acylation reaction.
Note the use of the word “moiety” as an alternative to “group” or
“fragment.”
Study the different types of carbonyl compounds listed in the table so that you can immediately recognize to which type a given compound belongs.
Some Carbonyl Compounds
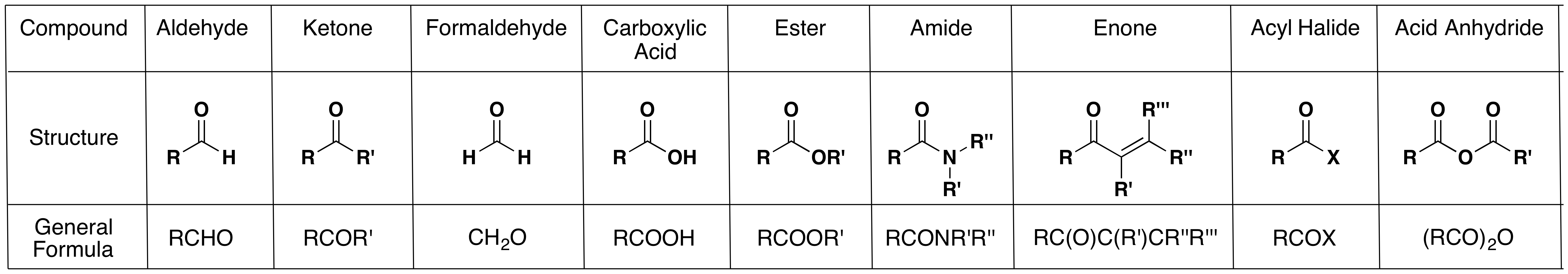
One should also add to this list lactones (cyclic esters) and lactams (cyclic amides).
II Nature of the Carbonyl Group
Objectives
After completing this section, you should be able to describe the electronic structure, polarity, and geometry of the carbonyl group.
A carbonyl group is a chemically organic functional group composed of a carbon atom double-bonded to an oxygen atom --> [C=O] The simplest carbonyl groups are aldehydes and ketones usually attached to another carbon compound. These structures can be found in many aromatic compounds contributing to smell and taste.
Introduction
Before going into anything in depth be sure to understand that the C=O entity itself is known as the "Carbonyl group" while the members of this group are called "carbonyl compounds" --> X-C=O. The carbon and oxygen are usually sp2 hybridized and planar.
Carbonyl Group Double Bonds
The double bonds in alkenes and double bonds in carbonyl groups are VERY different in terms of reactivity. The C=C is less reactive due to C=O electronegativity attributed to the oxygen and its two lone pairs of electrons. One pair of the oxygen lone pairs are located in 2s while the other pair are in 2p orbital where its axis is directed perpendicular to the direction of the pi orbitals. The Carbonyl groups properties are directly tied to its electronic structure as well as geometric positioning. For example, the electronegativity of oxygen also polarizes the pi bond allowing the single bonded substituent connected to become electron withdrawing.
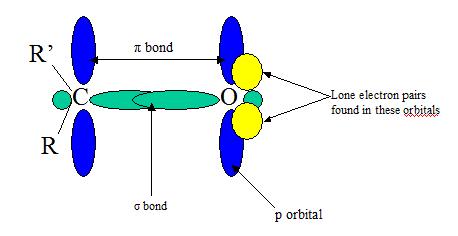
*Note: Both the pi bonds are in phase (top and botom blue ovals)
The double bond lengths of a carbonyl group is about 1.2 angstroms and the strength is about 176-179 kcal/mol). It is possible to correlate the length of a carbonyl bond with its polarity; the longer the bond meaing the lower the polarity. For example, the bond length in C=O is larger in acetaldehyde than in formaldehyde (this of course takes into account the inductive effect of CH3 in the compound).
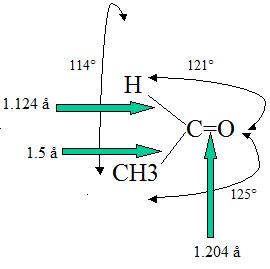
Polarization
As discussed before, we understand that oxygen has two lone pairs of electrons hanging around. These electrons make the oxygen more electronegative than carbon. The carbon is then partially postive (electrophillic) and the oxygen partially negative (nucleophillic). The polarizability is denoted by a lowercase delta and a positive or negative superscript depending. For example, carbon would have d+ and oxygen delta^(-). The polarization of carbonyl groups also effects the boiling point of aldehydes and ketones to be higher than those of hydrocarbons in the same amount. The larger the carbonyl compound the less soluble it is in water. If the compound exceeds six carbons it then becomes insoluble.
*For more information about carbonyl solubility, look in the "outside links" section
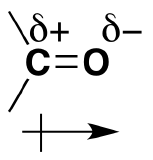
*Amides are the most stable of the carbonyl couplings due to the high-resonance stabilization between nitrogen-carbon and carbon-oxygen.
III General Reactions of Carbonyl Compounds
Objectives
After completing this section, you should be able to
- list the four general reaction mechanisms that dominate the chemistry of carbonyl compounds.
- discuss nucleophilic addition reactions of aldehydes and ketones (Chapter 19).
- write a general mechanism for nucleophilic addition to a carbonyl compound.
- write a detailed mechanism for the addition of a given nucleophile (e.g., a Grignard reagent) to an aldehyde or ketone.
- discuss nucleophilic acyl substitution reactions of carboxylic acid derivatives (Chapter 21).
- write a general mechanism to illustrate nucleophilic acyl substitution reactions.
- write a detailed mechanism for the reaction of a given carbonyl‑containing compound with a given nucleophile through a nucleophilic acyl substitution.
- discuss alpha substitution reactions (Chapter 22).
- write a general mechanism to illustrate alpha‑substitution of a carbonyl compound through the formation of an enol or enolate anion.
- explain the stability and ease of formation of enolate anions.
- write the detailed mechanism for the formation of an alpha‑substituted carbonyl compound from the overall reaction of a carbonyl compound with a primary alkyl halide.
- discuss carbonyl condensation reactions (Chapter 23).
- write a general mechanism to illustrate the condensation reaction that can occur between two molecules of a carbonyl compound; for example, the reaction of two molecules of acetaldehyde to form one molecule of aldol.
Key Terms
Note: All of these terms are defined in the “Study Notes,” below.
- aldol reaction
- alkylation
- alpha substitution
- carbonyl condensation
- enol
- enolate anion
- nucleophilic acyl substitution
- nucleophilic addition reaction
Study Notes
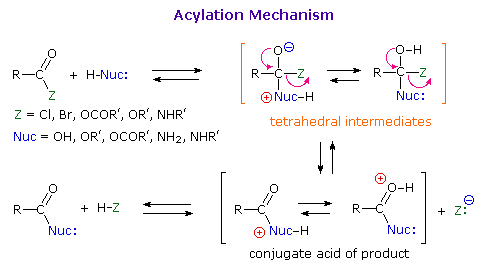
A “nucleophilic addition reaction” (of a carbonyl compound) involves the initial attack of a nucleophile on the slightly positive carbonyl‑carbon atom to form a tetrahedral intermediate.
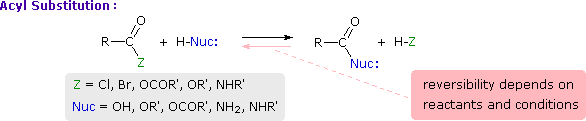
In a “nucleophilic acyl substitution reaction,” a nucleophile attacks the carbonyl carbon of a carboxylic acid derivative
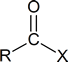
and initially produces a tetrahedral intermediate. The intermediate then reacts by expelling the leaving group, X, thereby forming a new carboxylic acid derivative.
In an “alpha substitution reaction” of a carbonyl compound, one of the hydrogen atoms attached to a carbon atom adjacent to the carbonyl group is removed and replaced by some other group.
Alpha substitution reactions of carbonyl compounds involve the “enol” form of the compound. Keto-enol tautomerism was briefly introduced in Section 9.4. Review this section if necessary.
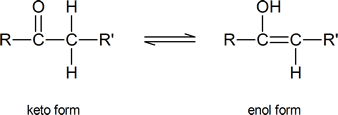
As you can see from the above diagram, the enol form has a hydroxyl group attached to one of the sp2 hybridized carbon atoms of a carbon-carbon double bond. Removal of the proton from the hydroxyl group produces a resonance-stabilized “enolate anion”:
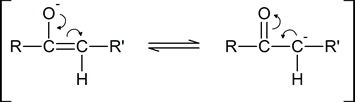
A “carbonyl condensation reaction” is one in which two carbonyl‑ containing molecules condense together (i.e., join together), often with the elimination of water. The reaction involves the formation of an enolate anion from one carbonyl‑containing molecule, followed by the subsequent nucleophilic attack by this enolate anion on the carbonyl carbon atom of the second molecule.
Nucleophilic Addition to a Carbonyl Group
C=O is prone to additions and nucleophillic attack because or carbon's positive charge and oxygen's negative charge. The resonance of the carbon partial positive charge allows the negative charge on the nucleophile to attack the Carbonyl group and become a part of the structure and a positive charge (usually a proton hydrogen) attacks the oxygen. Just a reminder, the nucleophile is a good acid therefore "likes protons" so it will attack the side with a positive charge.
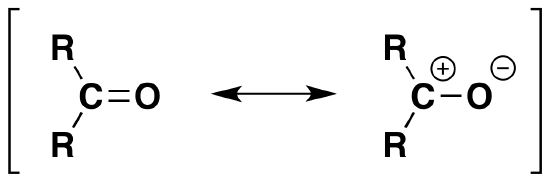
*Remember: due to the electronegative nature of oxygen the carbon is partially positive and oxygen is partially negative

1 2 3
- The Nucleophile (Nu) attacks the positively charged carbon and pushes one of the double bond electrons onto oxygen to give it a negative charge.
- The Nucleophile is now a part of the carbon structure with a negatively charged oxygen and a Na+ "floating" around.
- The negatively charged oxygen attacks the proton (H+) to give the resulting product above.
An Organometallic Example
Because organometallic reagents react as their corresponding carbanion, they are excellent nucleophiles. The basic reaction involves the nucleophilic attack of the carbanionic carbon in the organometallic reagent with the electrophilic carbon in the carbonyl to form alcohols.
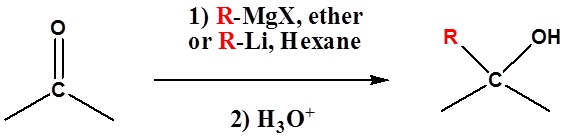
Nucleophilic Acyl Substitution
This is probably the single most important reaction of carboxylic acid derivatives. The overall transformation is defined by the following equation, and may be classified either as nucleophilic substitution at an acyl group or as acylation of a nucleophile. For certain nucleophilic reagents the reaction may assume other names as well. If Nuc-H is water the reaction is often called hydrolysis, if Nuc–H is an alcohol the reaction is called alcoholysis, and for ammonia and amines it is called aminolysis.
As illustrated in the following diagram, acylation reactions generally take place by an addition-elimination process in which a nucleophilic reactant bonds to the electrophilic carbonyl carbon atom to create a tetrahedral intermediate. This tetrahedral intermediate then undergoes an elimination to yield the products. In this two-stage mechanism bond formation occurs before bond cleavage, and the carbonyl carbon atom undergoes a hybridization change from sp2 to sp3 and back again. The facility with which nucleophilic reagents add to a carbonyl group was noted earlier for aldehydes and ketones.
Alpha-Substitution Reactions
Now we will investigate reactions which occur a the carbon alpha to the carbonyl groups. These reactions involve two new nucleophilic species the enol and the enolate.
Note! The electrophile replaces the hydrogen on the alpha carbon.
The following is a specific example of this type of reaction. Here we methylate the carbon alpha to the carbonyl group through an enolate intermediate. The hydrogen on the alpha carbon is acidic because of the presence of the C=O group and is therefore preferentially abstracted by the base.
Carbonyl Condensation Reactions
Enolizable aldehydes and enolizable ketones, in the presence of an acid or base catalyst usually in aqueous medium at low temperature, undergo a reaction, giving an aldol as the major product. This reaction is known as aldol reaction. The base-catalyzed aldol reaction, in which the catalyst is usually the hydroxide ion, is more common. Careful control of reaction temperature is critical because high temperatures promote aldol condensation. The optimum temperature depends on the nature of the aldehyde or ketone. Typically, heating the reaction mixture results in aldol condensation. For example:
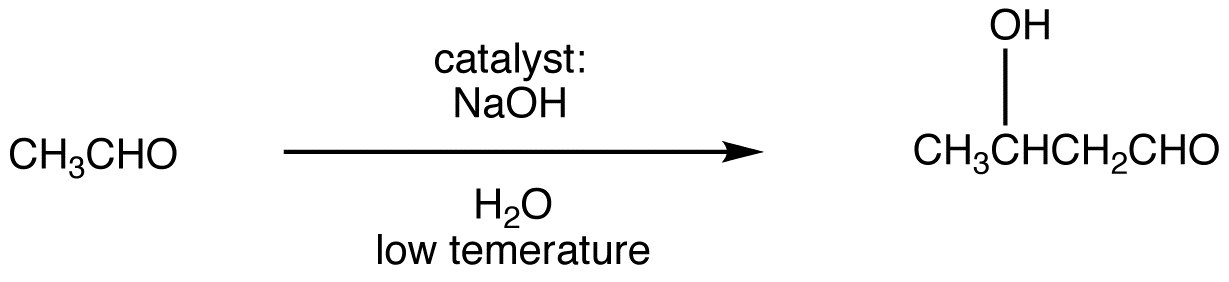
Mechanism:
Step 1: The hydroxide ion deprotanates the aldehyde reversibly.

Step 2: Enolate ion 1 adds to the unreacted aldehyde.

Step 3: Alkoxide ion 2 is protonated by water.
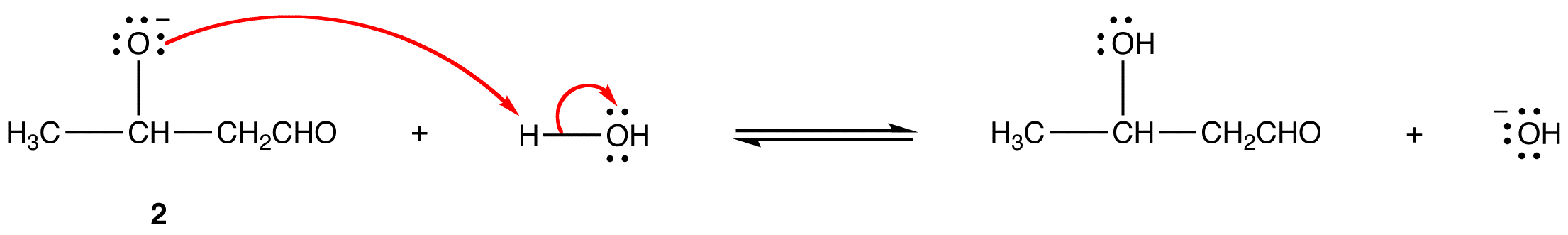
Since all three steps are reversible, the overall reaction is reversible and its equilibrium constant depends on the nature of the aldehyde or ketone. The reactions of aldehydes in which the α-carbon is a secondary carbon usually have large equilibrium constants; those of other aldehydes and ketones have small equilibrium constants. These observations suggest that the equilibrium constant of aldol reaction is sensitive to steric hindrance at the carbonyl carbon in the aldehyde or ketone.

