6.5: Chemical Formulas as Conversion Factors
- Page ID
- 48602
- Use chemical formulas as conversion factors.
Figure \(\PageIndex{1}\) shows that we need 2 hydrogen atoms and 1 oxygen atom to make one water molecule. If we want to make two water molecules, we will need 4 hydrogen atoms and 2 oxygen atoms. If we want to make five molecules of water, we need 10 hydrogen atoms and 5 oxygen atoms. The ratio of atoms we will need to make any number of water molecules is the same: 2 hydrogen atoms to 1 oxygen atom.
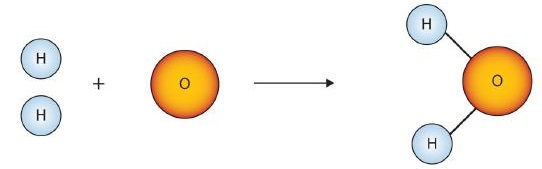
Using formulas to indicate how many atoms of each element we have in a substance, we can relate the number of moles of molecules to the number of moles of atoms. For example, in 1 mol of water (H2O) we can construct the relationships given in (Table \(\PageIndex{1}\)).
| 1 Molecule of \(H_2O\) Has | 1 Mol of \(H_2O\) Has | Molecular Relationships |
|---|---|---|
| 2 H atoms | 2 mol of H atoms | \(\mathrm{\dfrac{2\: mol\: H\: atoms}{1\: mol\: H_2O\: molecules}}\) or \(\mathrm{\dfrac{1\: mol\: H_2O\: molecules}{2\: mol\: H\: atoms}}\) |
| 1 O atom | 1 mol of O atoms | \(\mathrm{\dfrac{1\: mol\: O\: atoms}{1\: mol\: H_2O\: molecules}}\) or \(\mathrm{\dfrac{1\: mol\: H_2O\: molecules}{1\: mol\: O\: atoms}}\) |
A mole represents a very large number! The number 602,214,129,000,000,000,000,000 looks about twice as long as a trillion, which means it’s about a trillion trillion.
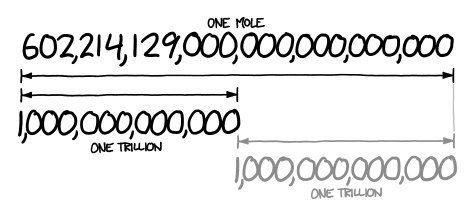
(CC BY-SA NC; what if? [what-if.xkcd.com]).
A trillion trillion kilograms is how much a planet weighs. If 1 mol of quarters were stacked in a column, it could stretch back and forth between Earth and the sun 6.8 billion times.
| 1 Molecule of \(C_2H_6O\) Has | 1 Mol of \(C_2H_6O\) Has | Molecular and Mass Relationships |
|---|---|---|
| 2 C atoms | 2 mol of C atoms | \(\mathrm{\dfrac{2\: mol\: C\: atoms}{1\: mol\: C_2H_6O\: molecules}}\) or \(\mathrm{\dfrac{1\: mol\: C_2H_6O\: molecules}{2\: mol\: C\: atoms}}\) |
| 6 H atoms | 6 mol of H atoms | \(\mathrm{\dfrac{6\: mol\: H\: atoms}{1\: mol\: C_2H_6O\: molecules}}\) or \(\mathrm{\dfrac{1\: mol\: C_2H_6O\: molecules}{6\: mol\: H\: atoms}}\) |
| 1 O atom | 1 mol of O atoms | \(\mathrm{\dfrac{1\: mol\: O\: atoms}{1\: mol\: C_2H_6O\: molecules}}\) or \(\mathrm{\dfrac{1\: mol\: C_2H_6O\: molecules}{1\: mol\: O\: atoms}}\) |
|
2 (12.01 amu) C 24.02 amu C |
2 (12.01 g) C 24.02 g C |
\(\mathrm{\dfrac{24.02\: g\: C\: }{1\: mol\: C_2H_6O\: molecules}}\) or \(\mathrm{\dfrac{1\: mol\: C_2H_6O\: molecules}{24.02\: g\: C\: }}\) |
|
6 (1.008 amu) H 6.048 amu H |
6 (1.008 g) H 6.048 g H |
\(\mathrm{\dfrac{6.048\: g\: H\: }{1\: mol\: C_2H_6O\: molecules}}\) or \(\mathrm{\dfrac{1\: mol\: C_2H_6O\: molecules}{6.048\: g\: H\: }}\) |
|
1 (16.00 amu) O 16.00 amu O |
1 (16.00 g) O 16.00 g O |
\(\mathrm{\dfrac{16.00\: g\: O\: }{1\: mol\: C_2H_6O\: molecules}}\) or \(\mathrm{\dfrac{1\: mol\: C_2H_6O\: molecules}{16.00\: g\: O\: }}\) |
The following example illustrates how we can use the relationships in Table \(\PageIndex{2}\) as conversion factors.
If a sample consists of 2.5 mol of ethanol (C2H6O), how many moles of carbon atoms does it have?
Solution
| Steps for Problem Solving | If a sample consists of 2.5 mol of ethanol (C2H6O), how many moles of carbon atoms does it have? |
|---|---|
| Identify the "given" information and what the problem is asking you to "find." |
Given: 2.5 mol C2H6O |
| List other known quantities. | 1 mol C2H6O = 2 mol C |
| Prepare a concept map and use the proper conversion factor. | |
| Cancel units and calculate. |
Note how the unit mol C2H6O molecules cancels algebraically. \(\mathrm{2.5\: \cancel{mol\: C_2H_6O\: molecules}\times\dfrac{2\: mol\: C\: atoms}{1\: \cancel{mol\: C_2H_6O\: molecules}}=5.0\: mol\: C\: atoms}\) |
| Think about your result. | There are twice as many C atoms in one C2H6O molecule, so the final amount should be double. |
If a sample contains 6.75 mol of Na2SO4, how many moles of sodium atoms, sulfur atoms, and oxygen atoms does it have?
- Answer
- 13.5 mol Na atoms, 6.75 mol S atoms, and 27.0 mol O atoms
The fact that 1 mol equals 6.022 × 1023 items can also be used as a conversion factor.
Determine the mass of Oxygen in 75.0g of C2H6O.
Solution
| Steps for Problem Solving | Determine the mass of Oxygen in 75.0g of C2H6O |
|---|---|
| Identify the "given" information and what the problem is asking you to "find." |
Given: 75.0g C2H6O |
| List other known quantities. |
1 mol O = 16.0g O 1 mol C2H6O = 1 mol O 1 mol C2H6O = 46.07g C2H6O |
| Prepare a concept map and use the proper conversion factor. |
|
| Cancel units and calculate. | \(\require{cancel}\mathrm{75.0\: \cancel{g\: C_2H_6O}\times\dfrac{1\: \cancel{mol\: C_2H_6O}}{46.07\:\cancel{g\: C_2H_6O}}\times\dfrac{1\: \cancel{mol\:O}}{1\: \cancel{mol\:C_2H_6O}}\times\dfrac{16.00\: g\: O}{1\: \cancel{mol\:O}}=26.0\: g\: O}\) |
| Think about your result. |
- How many molecules are present in 16.02 mol of C4H10? How many C atoms are in 16.02 mol?
- How many moles of each type of atom are in 2.58 mol of Na2SO4?
- Answer a:
- 9.647 x 1024 C4H10 molecules and 3.859 x 1025 C atoms
- Answer b:
- 5.16 mol Na atoms, 2.58 mol S atoms, and 10.3 mol O atoms
Summary
In any given formula, the ratio of the number of moles of molecules (or formula units) to the number of moles of atoms can be used as a conversion factor.

