5.6: Reactive intermediates
- Page ID
- 225789
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)In chemistry, a reactive intermediate or an intermediate is a short-lived, high-energy, highly reactive molecule. When generated in a chemical reaction, it will quickly convert into a more stable molecule. Only in exceptional cases can these compounds be isolated and stored, e.g. low temperatures, matrix isolation. When their existence is indicated, reactive intermediates can help explain how a chemical reaction takes place.
Most chemical reactions take more than one elementary step to complete, and a reactive intermediate is a high-energy, yet stable, product that exists only in one of the intermediate steps. The series of steps together make a reaction mechanism. A reactive intermediate differs from a reactant or product or a simple reaction intermediate only in that it cannot usually be isolated but is sometimes observable only through fast spectroscopic methods. It is stable in the sense that an elementary reaction forms the reactive intermediate and the elementary reaction in the next step is needed to destroy it.
When a reactive intermediate is not observable, its existence must be inferred through experimentation. This usually involves changing reaction conditions such as temperature or concentration and applying the techniques of chemical kinetics, chemical thermodynamics, or spectroscopy. We will often refer to certain reactive intermediates based on carbon, viz., carbocations, radicals, carbanions and carbenes.
Common features
Reactive intermediates have several features in common:
- low concentration with respect to reaction substrate and final reaction product
- often generated on chemical decomposition of a chemical compound
- it is often possible to prove the existence of this species by spectroscopic means
- cage effects have to be taken into account
- often stabilization by conjugation or resonance
- often difficult to distinguish from a transition state
- prove existence by means of chemical trapping
References
- ^ Carey, Francis A.; Sundberg, Richard J.; (1984). Advanced Organic Chemistry Part A Structure and Mechanisms (2nd ed.). New York N.Y.: Plenum Press. ISBN0-306-41198-9.
- ^ March Jerry; (1885). Advanced Organic Chemistry reactions, mechanisms and structure (3rd ed.). New York: John Wiley & Sons, inc. ISBN0-471-85472-7
- ^ Gilchrist, T. L. (1966). Carbenes nitrenes and arynes. Springer US. ISBN9780306500268.
- ^ Moss, Robert A.; Platz, Matthew S.; Jones, Jr., Maitland (2004). Reactive intermediate chemistry. Hoboken, N.J.: Wiley-Interscience. ISBN9780471721499.
Carbocations (R+)
- +3 in carbenium ions (protonated carbenes),
- +5 or +6 in the carbonium ions (protonated alkanes, named by analogy to ammonium). These are much less common.
Structure and properties
The charged carbon atom in a carbocation is a “sextet”, i.e. it has only six electrons in its outer valence shell instead of the eight valence electrons that ensures maximum stability (octet rule). Therefore, carbocations are often reactive, seeking to fill the octet of valence electrons as well as regain a neutral charge. One could reasonably assume a carbocation to have $$sp^3$$ hybridization with an empty $$sp_3$$ orbital giving positive charge. However, the reactivity of a carbocation more closely resembles $$sp^2$$ hybridization with atrigonal planar molecular geometry. An example is the methyl cation, $$CH_3^+$$.
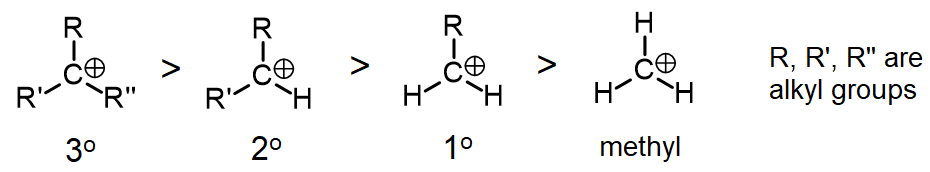
Order of stability of examples of tertiary (3o), secondary (2o), and primary (1o) alkylcarbeniumions, as well as the methyl cation (far right). The methyl group is so unstable it is only observed in the gas phase.
Carbocations are often the target of nucleophilic attack by nucleophiles such as water or halide ions.
Carbocations typically undergo rearrangement reactions from less stable structures to equally stable or more stable ones with rate constants in excess of 109/sec. This fact complicates synthetic pathways to many compounds. For example, when 3-pentanol is heated with aqueous HCl, the initially formed 3-pentyl carbocation rearranges to a statistical mixture of the 3-pentyl and 2-pentyl. These cations react with chloride ion to produce about 1/3 3-chloropentane and 2/3 2-chloropentane.
A carbocation may be stabilized by resonance by a carbon-carbon double bond next to the ionized carbon. Such cations as allyl cation CH2=CH–CH2+ and benzyl cation C6H5–CH2+ are more stable than most other carbocations. Molecules that can form allyl or benzyl carbocations are especially reactive. These carbocations where the C+ is adjacent to another carbon atom that has a double or triple bond have extra stability because of the overlap of the empty p orbital of the carbocation with the p orbitals of the π bond. This overlap of the orbitals allows the charge to be shared between multiple atoms – delocalization of the charge – and, therefore, stabilizes the carbocation.
References
- Gold Book definition for carbocation
Radicals
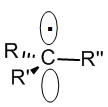
A radicals is a seven electron intermediate that adopts a flat, sp2 structure despite the fact that it has four electron groups; the lone electron resides in a half-filled p-orbital. This sp2 structure allows radicals to delocalize the single electron through resonance. We will study radical reactions in detail in the second semester.
Being short of the octet, radicals are electrophilic, and therefore they are stabilized by alkyl groups. Thus the order for stability is the same as for carbocations, namely tertiary > secondary > primary > methyl .
Carbanions

A carbanion is an eight electron intermediate with an sp3 structure as shown in A. Despite its full octet, it is very reactive due to the fact that carbon is not very electronegative. Although it is sp3, it can participate in resonance because it can easily re-hybridize to an sp2 structure (see B), which allows overlap.
Carbanions are electron-rich and nucleophilic, so in fact they are destabilized by alkyl groups. This means that the order for stability is the opposite of that for carbocations, namely methyl > primary > secondary > tertiary.
Carbenes
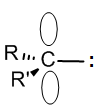
Carbenes are the least obvious of the four common intermediates; in most cases they have a six-electron sp2 structure that has a lone pair but no overall charge. Although they are short of a full octet, they also have a reactive lone pair, so (depending on structure) carbenes can be either electrophilic or nucleophilic, or sometimes both – they just like to react with almost anything! We will learn about carbene reactions in section 10.7.
- Reactive_intermediate. Authored by: Wikipedia contributors. Provided by: Wikimedia Foundation. Located at: https://en.wikipedia.org/wiki/Reactive_intermediate. Project: Wikipedia. License: CC BY-SA: Attribution-ShareAlike
- Carbocations. Authored by: Libretexts contributors. Provided by: UC Davis. Located at: https://chem.libretexts.org/Textbook_Maps/Organic_Chemistry/Supplemental_Modules_(Organic_Chemistry)/Fundamentals/Reactive_Intermediates/Carbocations. Project: Libretexts. License: CC BY-NC: Attribution-NonCommercial

