8.12: Outdoor Air Pollution
- Page ID
- 189499
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
Air pollution refers to the introduction, into the atmosphere, of substances that have harmful effects on humans, other living organisms, and the environment either as solid particles, liquid droplets or gases. Air pollution can result from natural processes such as dust storms, forest fires, and volcanic eruptions, or from human activities such as biomass burning, vehicular emissions, mining, agriculture, and industrial processes. Improved technology and government policies have helped reduce most types of outdoor air pollution in many industrialized countries including the United States, in recent decades. However, outdoor air quality is still a problem in less industrialized nations, especially in megacities of rapidly industrializing nations such as China and India.
Outdoor pollutants can come from stationary (point) sources or mobile (nonpoint) sources (Figure \(\PageIndex{1}\)). Stationary sources have a fixed location, for example power plant smokestacks, burning, construction sites, farmlands and surface mines among others. Mobile sources of air pollutants move from place to place while emitting pollutants. Examples of mobile sources include vehicles, aircrafts, ships, and trains.
.png?revision=1&size=bestfit&width=777&height=372) Figure \(\PageIndex{1}\): Images showing various sources of air pollution, including natural and anthropogenic, stationary and mobile. Source: All images obtained from Wiki Commons commons.wikimedia.org/wiki/Air_pollution
Figure \(\PageIndex{1}\): Images showing various sources of air pollution, including natural and anthropogenic, stationary and mobile. Source: All images obtained from Wiki Commons commons.wikimedia.org/wiki/Air_pollution
Pollutants are categorized into two major types based on how they originated namely primary and secondary pollutants. Primary pollutants are those released directly from the source into the air in a harmful form. The primary pollutants that account for nearly all air pollution problems are carbon monoxide (58%), volatile organic compounds (VOCs, 11%), nitrogen oxides (15%), sulfur dioxides (13%), and particulate material (3%). Secondary pollutants are produced through reactions between primary pollutants and normal atmospheric compounds. For example, ground-level ozone forms over urban areas through reactions, powered by sunlight, between primary pollutants (oxides of nitrogen) and other atmospheric gases such as VOCs.
6.3.1: Criteria pollutants
Under the Clean Air Act (section 6.7.1), the Environmental Protection Agency (EPA) establishes air quality standards to protect public health and the environment. EPA has set national air quality standards for six common air pollutants namely: 1) carbon monoxide; 2) ground-level ozone; 3) nitrogen dioxide; 4) Sulfur dioxide; 5) lead; and 6) particulate matter (also known as particle pollution). Of the six pollutants, particle pollution and ground-level ozone are the most widespread health threats. EPA calls these pollutants "criteria" air pollutants because it regulates them by developing human health-based and/or environmentally-based criteria (science-based guidelines) for setting permissible levels. The set of limits based on human health is called primary standards. Another set of limits intended to prevent environmental and property damage is called secondary standards.
- Carbon Monoxide (CO): is a colorless, odorless gas emitted from combustion processes, specifically, the incomplete combustion of fuel. Nationally and, particularly in urban areas, the majority of CO emissions to ambient air come from mobile sources. CO can cause harmful health effects by reducing oxygen delivery to the body's organs (like the heart and brain) and tissues. At extremely high levels, CO can cause death.
- Ground-level ozone (O3): is a colorless gas with a slightly sweet odor that is not emitted directly into the air, but is created by the interaction of sunlight, heat, oxides of nitrogen (NOx) and volatile organic compounds (VOCs). Ozone is likely to reach unhealthy levels on hot sunny days in urban environments. Emissions from industrial facilities and electric utilities, motor vehicle exhaust, gasoline vapors, and chemical solvents are some of the major sources of NOx and VOCs.
- Nitrogen dioxide (NO2): is one of a group of highly reactive gasses known as "oxides of nitrogen," or "nitrogen oxides (NOx)." Other nitrogen oxides include nitrous acid and nitric acid. NO2 is a yellowish-brown to reddish-brown foul-smelling gas that is a major contributor to smog and acid rain. Nitrogen oxides result when atmospheric nitrogen and oxygen react at the high temperatures created by combustion engines. Most emissions in the U.S. result from combustion in vehicle engines, electrical utility, and industrial combustion.
- Sulfur dioxide (SO2): Sulfur dioxide is one of a group of highly reactive gasses known as “oxides of sulfur.” The largest sources of SO2 emissions are from fossil fuel combustion at power plants (73%) and other industrial facilities (20%). Smaller sources of SO2emissions include industrial processes such as extracting metals from their ores, and the burning of high sulfur containing fuels by locomotives, large ships, and non-road equipment.
- Lead (Pb): is a metal found naturally in the environment as well as in manufactured products. The major sources of lead emissions have historically been from fuels in motor vehicles (such as cars and trucks) and industrial sources. As a result of EPA's regulatory efforts to remove lead from gasoline, emissions of lead from the transportation sector dramatically declined by 95 percent between 1980 and 1999, and levels of lead in the air decreased by 94 percent during that time period. The major sources of lead emissions today are ore and metal processing and piston-engine aircraft operating on leaded aviation gasoline. Today, the highest levels of lead in air are usually found near lead smelters.
- Particulate material (PM), sometimes known simply as “particulates” refers to solid particles and liquid droplets suspended in the air we breathe. Particulate pollution is made up of a variety of components, including acids (nitrates and sulfates), organic chemicals, metals, soil or dust particles, and allergens (pollen and mold spores). The size of the particles in directly linked to their potential for causing health problems. Particles that are 10 micrometers in diameter or smaller generally pass through the throat and nose and enter the lungs. EPA groups these into two types: "inhalable coarse particles," with diameters larger than 2.5 micrometers and smaller than 10 micrometers and "fine particles," with diameters that are 2.5 micrometers and smaller. How small is 2.5 micrometers? Think about a single hair from your head. The average human hair is about 70 micrometers in diameter – making it 30 times larger than the largest fine particle (Figure \(\PageIndex{2}\)). Our respiratory systems are equipped to filter larger particles out of the air once it is inhaled. However, the lungs are vulnerable to both coarse particles (PM10), and fine particles (PM2.5). These can slip past the respiratory system's natural defenses and get deep into the lungs and some may even get into the bloodstream. Coarse particles come from road dust while fine particles come from combustion processes.
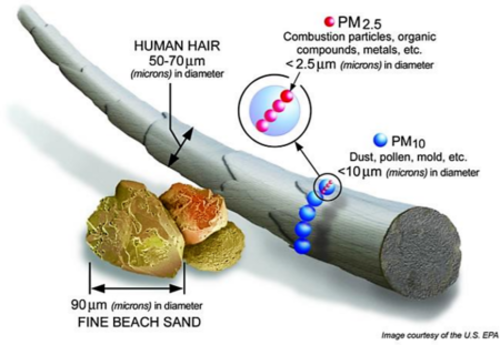 Figure \(\PageIndex{2}\): Graphic showing size comparison of particulate material (PM10 and PM2.5) compared to fine beach sand and human hair. Source: US EPA www.epa.gov/air/particlepollution/basic.html
Figure \(\PageIndex{2}\): Graphic showing size comparison of particulate material (PM10 and PM2.5) compared to fine beach sand and human hair. Source: US EPA www.epa.gov/air/particlepollution/basic.html
6.3.2: Volatile Organic Compounds
Volatile organic compounds (VOCs) are carbon-containing chemicals emitted as gases from natural and human-made sources. Natural sources of VOCs include plants, the largest source, and bacteria in the guts of termites and ruminant animals. These compounds are generally oxidized to carbon monoxide and carbon dioxide in the atmosphere. VOCs are of great concern because they are precursors for the formation of ozone, a secondary air pollutant.
A large number of synthetic organic chemicals such as benzene, toluene, formaldehyde, vinyl chloride, chloroform, and phenols are widely used as ingredients in countless household products. Paints, paint strippers, varnishes, many cleaning, disinfecting, cosmetic, degreasing, and hobby products all contain VOCs. Fuels are also made up of organic chemicals. All of these products can release organic compounds while you are using them, and, to some degree, when they are stored. The “new car smell” characteristic of new cars is from a complex mix of dozens of VOC. Also, concentrations of many VOCs are consistently higher indoors (up to ten times higher) than outdoors. They are often held responsible for sick building syndrome, an illness resulting from indoor pollution in which the specific cause is not identified.
6.3.3: Smog
Smog is a mixture of air pollutants (sulfur dioxide, nitrogen oxides, ozone, and particulates) that often form over urban areas as a result of fossil fuel combustion. The term was coined from the terms “smoke” and “fog” referring to a brownish haze that pollutes the air, greatly reducing visibility and making it difficult for some people to breathe (Figure \(\PageIndex{3}\) and \(\PageIndex{4}\)). There are two main types of smog: industrial and photochemical smog. Industrial smog is produced primarily by the burning of fossil fuels which produces carbon dioxide (from complete combustion), carbon monoxides (from partial combustion), sulfur, and mercury. The sulfur reacts with other chemicals in the atmosphere producing several sulfur compounds including sulfur dioxide. These compounds along with particulate material make up industrial smog. Photochemical smog is formed when sunlight drives chemical reactions between primary pollutants from automobiles and normal atmospheric compounds. The product is a mix of over 100 different chemicals with the most abundant being ground-level ozone.
.png?revision=1&size=bestfit&width=486&height=317) Figure \(\PageIndex{3}\): Smog over Almaty city, Kazakhstan. Photo by Igors Jefimovs. Source: Wikicommons commons.wikimedia.org/wiki/C...dia/File:Smog_ over_Almaty.jpg
Figure \(\PageIndex{3}\): Smog over Almaty city, Kazakhstan. Photo by Igors Jefimovs. Source: Wikicommons commons.wikimedia.org/wiki/C...dia/File:Smog_ over_Almaty.jpg
.png?revision=1) Figure \(\PageIndex{4}\): Smog over Santiago in Chile. Source: German Wikipedia commons.wikimedia.org/wiki/C...#/media/File:S antiago30std.jpg
Figure \(\PageIndex{4}\): Smog over Santiago in Chile. Source: German Wikipedia commons.wikimedia.org/wiki/C...#/media/File:S antiago30std.jpg
6.3.4: Toxic pollutants
Toxic air pollutants, also known as hazardous air pollutants, are those pollutants that are known or suspected to cause cancer or other serious health effects, such as reproductive effects or birth defects, or adverse environmental effects. Examples of toxic air pollutants include benzene, which is found in gasoline; perchloroethylene, which is emitted from some dry cleaning facilities; methylene chloride, which is used as a solvent and paint stripper by a number of industries; and others such as dioxin, asbestos, toluene, and metals such as cadmium, mercury, chromium, and lead compounds.
Most air toxics originate from human-made sources, including mobile sources (e.g., cars, trucks, buses) and stationary sources (e.g., factories, refineries, power plants), as well as indoor sources (e.g., some building materials and cleaning solvents). Some air toxics are also released from natural sources such as volcanic eruptions and forest fires. Exposure to air toxics is mainly through breathing but some toxic air pollutants such as mercury can deposit onto soils or surface waters, where they are taken up by plants and ingested by animals and are eventually magnified up through the food chain. Like humans, animals may experience health problems if exposed to sufficiently high quantities of air toxics over time.

