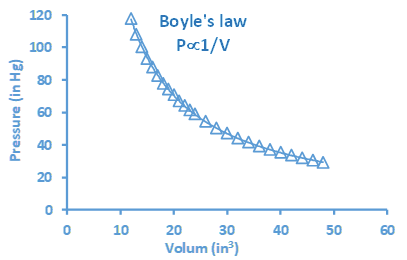
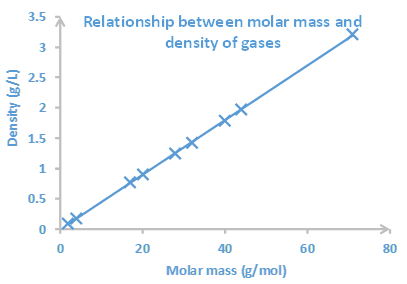
The Kelvin scale
Kelvin is the base unit of temperature in the SI. The freezing point of water is 273.15 K, and the boiling point of water is 373.15 K. For most practical purposes, the freezing point of water is reported as 273 K and the boiling point 373 K, i.e., accurate to three significant figures. Celsius scale units are the same size but shifted up by 273 compared to the Kelvin scale. So, the relationship between Kelvin and Celsius is:
\begin{equation}
T_{K}= T_{C}+273,\nonumber
\end{equation}
where TK is the temperature in Kelvin, and TC is the temperature in degrees Celsius. This equation converts temperature in Kelvin to temperature in Celsius.
A 0 K, also called absolute zero, is the temperature of a matter at which no energy can be removed as heat from the matter. There is no negative temperature on the Kelvin scale.
Fahrenheit (oF)
Fahrenheit is the base unit of the English system, with 32 oF at the freezing point of water and 212 oF at the boiling point of water. Fahrenheit is \(\frac{5}{9}\) times shorter and shifted up by 32 than Celsius. So the relationship between the two is:
\begin{equation}
T_{F}=\frac{9}{5} \times T_{C}+32,\nonumber
\end{equation}
where TF is the temperature in Fahrenheit, and TC is the temperature in degrees Celsius. This equation converts temperature in Celsius to temperature in Fahrenheit.
Manipulating temperature conversion equations
The equation for converting Celsius to Fahrenheit is:
\begin{equation}
T_{F}=\frac{9}{5} \times T_{C}+32,\nonumber
\end{equation}
Addition or subtraction of the same number on the two sides of an equation does not change the equality. Subtracting 32 from both sides of the above equation leads to:
\begin{equation}
T_{F} -32=\frac{9}{5} \times T_{C}\cancel{+32}\cancel{-32},\nonumber
\end{equation}
\begin{equation}
T_{F} -32=\frac{9}{5} \times T_{C}.\nonumber
\end{equation}
Multiplication or division by the same number on both sides of an equation does not change equality. Remember that multiplication or division should apply to every term on either side of the equality. Enclose the side with more than one term in small brackets and then do the multiplication of division operation so that it applies to each term in the bracket. Multiplying both sides of the above equation with \(\frac{5}{9}\) leads to:
\begin{equation}
\frac{5}{9} \times\left(T_{F}-32\right)=\cancel{\frac{5}{9}} \times \cancel{\frac{9}{5}} \times T_{C}\nonumber
\end{equation}
\begin{equation}
\frac{5}{9} \times\left(T_{F}-32\right)=T_{C}\nonumber
\end{equation}
Swapping the sides of an equation does not change equality. Swapping the sides in the above equation to bring TC to the left:
\begin{equation}
T_{C}=\frac{5}{9} \times\left(T_{F}-32\right)\nonumber
\end{equation}
This is the equation for Fahrenheit to Celsius conversion.
The procedure of rearranging an equation described above applies to all algebraic equations. For example, start with a relationship that converts Celsius to Kelvin:
\begin{equation}
T_{K}= T_{C}+273,\nonumber
\end{equation}
subtract 273 from both sides:
\begin{equation}
T_{K}-273= T_{C}\cancel{+273}\cancel{-273},\nonumber
\end{equation}
\begin{equation}
T_{K}-273= T_{C},\nonumber
\end{equation}
and finally swap the left and right side to bring TC to the left:
\begin{equation}
T_{C}=T_{K}-273.\nonumber
\end{equation}
This is the equation for Kelvin to Celsius conversion.