2.9: Determination of the Molar Mass by Freezing Point Depression (Experiment)-Home Version
- Page ID
- 386958
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- To determine the molar mass of an unknown solid using the colligative property of freezing point depression.
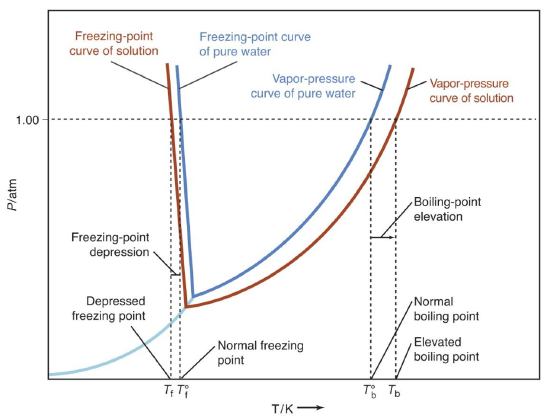
When a small amount of nonvolatile solute is dissolved in a volatile solvent, the vapor pressure of the solvent over the solution will be less than the vapor pressure of the pure solvent at the same temperature. Therefore, the temperature at which the equilibrium vapor pressure reaches atmospheric pressure is higher for the solution than for the pure solvent. As a result the boiling point of the solution, \(T_{b}\), is higher than the boiling point of the pure solvent, \(T^{o}_{b}\). The amount by which the boiling point of the solution exceeds the boiling point of the pure liquid, \(\Delta T_{b} = T_{b} - T^{o}_{b}\), is called the boiling point elevation. Similarly, because of the reduction in vapor pressure over the solution the freezing point of the solution, \(T_{f}\), is lower than the freezing point of the pure solvent, \(T^{o}_{f}\). The amount by which the freezing point of the solution is decreased from that of the pure liquid, \(\Delta T_{f} = T^{o}_{f} - T_{f}\), is called the freezing point depression. Figure 1 below illustrates this behavior for water.
Figure 1
The magnitude of the freezing depression produced by a solute is proportional to its colligative molality, \(m_{c}\):
\[\Delta T_{f} =T^{o}_{f} - T_{f} =K_{f} \times m_{c} \label{1}\]
\(K_{f}\) is known as the freezing point depression constant, and depends on the solvent used.
In this experiment you will determine the molar mass of an unknown solid by dissolving a pre-weighed sample in a solvent, and measuring the resulting freezing point depression of the solvent. From the measured \(\Delta T_{f}\) and the known \(K_{f}\) value of the solvent, you can then determine the value of \(m_{c}\) using the above Equation \ref{1}. The colligative molality, \(m_{c}\), is related to the molality of the solution, \(m\), by the expression:
\[m_{c} =i \times m \label{2}\]
where \(i\) is the number of solute particles produced per formula unit of dissolved solute, and \(m\) is the number of moles of solute per kilogram of solvent. Since only non-dissociating solutes will be used in this experiment, the value of \(i\) for your unknown solute can be considered to be 1. Thus, you may assume that \(m_{c} = m\). From the experimentally determined value of \(m\) and the mass of solute added, you can determine the molar mass of the unknown solute.
The solvent that will be used in this experiment is water. It has a freezing point of 0 °C.
In order to determine the freezing point of this pure solvent you must cool it using an ice bath, and then measure the temperature as a function of time as the liquid cools. At first the temperature will fall quite rapidly. When the freezing point is reached, solid will begin to form, and the temperature will tend to hold steady until the sample is all solid. This behavior is shown in Figure 2 below. The freezing point of the pure liquid is the constant temperature observed while the liquid is freezing to a solid.
The cooling behavior of a solution is somewhat different from that of a pure liquid, also shown in Figure 2. As discussed earlier, the temperature at which a solution freezes is lower than that for the pure solvent. In addition, there is a slow gradual fall in temperature as freezing proceeds. The best value for the freezing point of the solution is obtained by drawing two straight lines connecting the points on the temperature-time graph. The first line connects points where the solution is all liquid. The second line connects points where solid and liquid coexist. The point where the two lines intersect is the freezing point of the solution.
Note that when the solid first appears the temperature may fall below the freezing point, but then it comes back up as more of the solid forms. This effect is called supercooling, and is a phenomenon that may occur with both the pure liquid and the solution (see Figure 2). When drawing the straight line in the solid-liquid region of the graph, ignore points where supercooling is observed. To establish the proper straight line in the solid-liquid region it is necessary to record the temperature until the trend with time is smooth and clearly established.
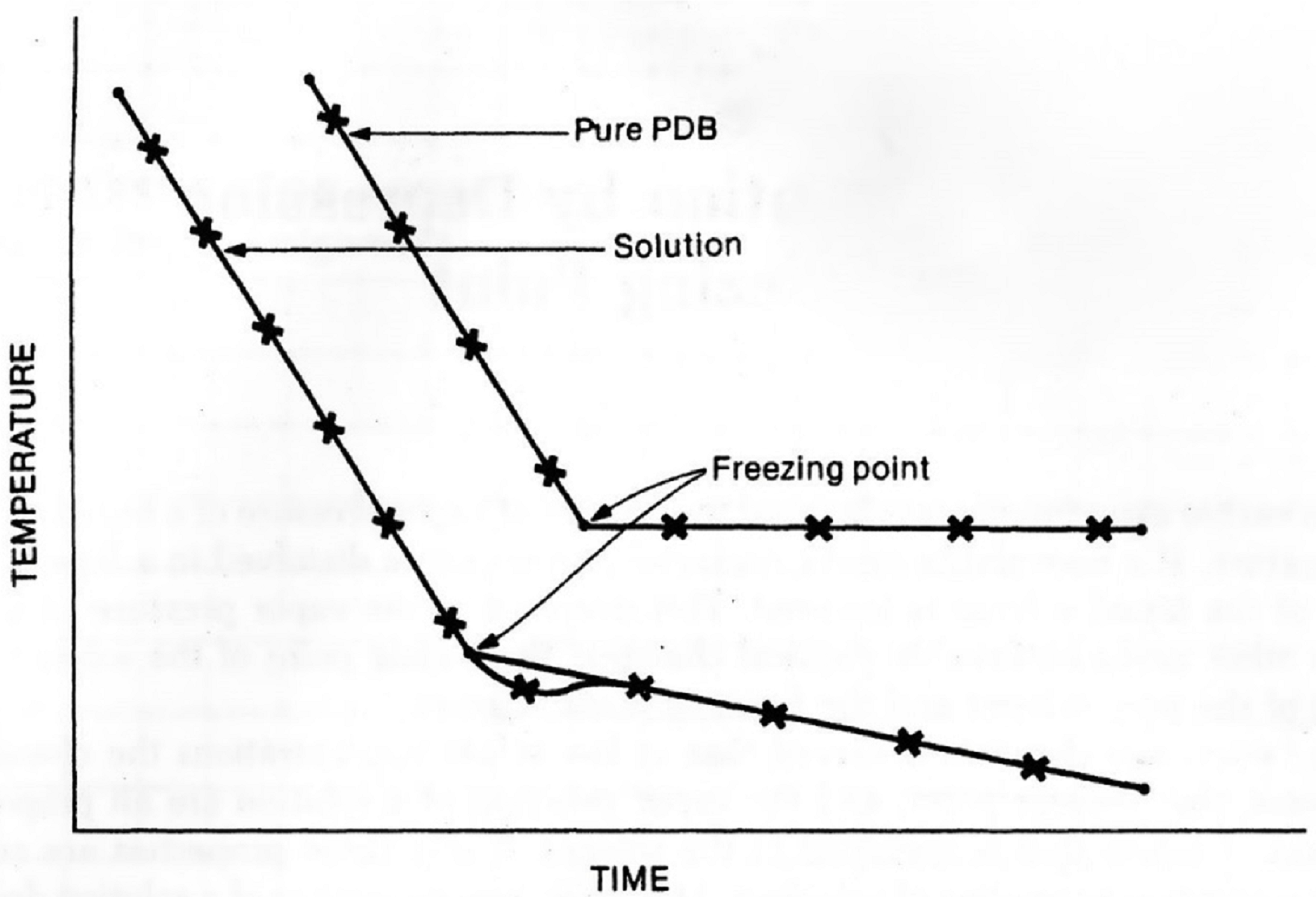
Figure 2
Procedure
Materials Required
Water, thermometer, sugar, 100 ml beaker or a test tube/small glass tumbler/glass bottle to hold the solutions for cooling, scale, spoon, Table salt/ice cream salt, a bigger and wider beaker/container to make ice-bath, ice
Since we are using non-hazardous chemicals, no safety precautions are advised except for glass safety
Watch the following video for a demonstration of a similar experiment.
Part 1: Determining the Freezing Point of Pure Water (not so pure if it is not distilled!)
- Take a bigger beaker (1 L) or container. Fill 3/4th of this container with ice cubes with 10-20 grams of salt added in between to make an ice-bath.
- Weigh a clean dry large test tube or a small 100 ml beaker using an electronic balance, and record its mass. Add about 10 grams of water to the test tube. Reweigh and record the mass of the test tube and the water. Calculate the mass of water in the test tube by difference.
- Insert your thermometer into the water.
- Place the container in the ice-bath.
- Monitor the temperature of the water as it cools. Stir the liquid slowly but continuously to help minimize supercooling. When the temperature of the water reaches 0 °C begin recording its temperature to the nearest tenth of a degree every 30 seconds. Continue for at least 4 minutes after the first solid starts to appear, or until the liquid has solidified to a point that you are no longer able to stir it. Near the melting point you will observe crystals of water in the liquid, and these will increase in amount as the cooling proceeds. Note the temperature at which these crystals first start to appear.
Part 2: Determining the Freezing Point of Water with about 3 g Unknown Solute (but we know it is sugar!)
- Weigh out and transfer about 3 grams of the unknown solid sample sugar into the test tube, taking great care that none of the unknown sample is spilled during this process. If you do spill some you will need to start this step over with a fresh sample of weighed water. Add 10 grams of water into this and dissolve well.
- Place the well dissolved sugar solution in the test with a thermometer inserted into in the ice-bath to cool.
- Monitor the temperature of the water-unknown sugar mixture as it cools. Stir the liquid slowly but continuously to help minimize supercooling. When the temperature of the mixture reaches 0 °C begin recording its temperature to the nearest tenth of a degree every 30 seconds. Continue for at least 4 minutes after the first solid starts to appear, or until the mixture has solidified to a point that you are no longer able to stir it. Note the temperature at which crystals first start to appear.
- The dependence of temperature on time for the mixture should be similar to that observed for the pure water, except that the first crystals should appear at a lower temperature for the mixture. There may be some supercooling, as evidenced by a rise in temperature shortly after the first appearance of crystals.
Part 3: Determining the Freezing Point of Water with about 5 g Unknown Solute (but we know it is sugar!)
- Weigh out and transfer 5 grams of the unknown solid sample sugar into the test tube, taking great care that none of the unknown sample is spilled during this process. If you do spill some you will need to start this step over with a fresh sample of weighed water. Add 10 grams of water into this and dissolve well.
- Place the well dissolved sugar solution in the test with a thermometer inserted into in the ice-bath to cool.
- Monitor the temperature of the water-unknown sugar mixture as it cools. Stir the liquid slowly but continuously to help minimize supercooling. When the temperature of the mixture reaches 0 °C begin recording its temperature to the nearest tenth of a degree every 30 seconds. Continue for at least 4 minutes after the first solid starts to appear, or until the mixture has solidified to a point that you are no longer able to stir it. Note the temperature at which crystals first start to appear.
- The dependence of temperature on time for the mixture should be similar to that observed for the pure water, except that the first crystals should appear at a lower temperature for the mixture. There may be some supercooling, as evidenced by a rise in temperature shortly after the first appearance of crystals.
Lab Report: Determination of Molar Mass by Freezing Point Depression
Experimental Data
| Part 1: Freezing Point of Pure Water | |
|---|---|
| Mass of large empty test tube | |
| Mass of test tube & water | |
| Mass of water (by difference) | |
| Part 2: Freezing Point of PDB-Unknown Mixture (~3 g) | |
|---|---|
| Mass of unknown added , Sugar Sample I | |
| Part 3: Freezing Point of PDB-Unknown Mixture (~5 g) | |
|---|---|
| Mass of unknown added, Sugar sample II | |
Temperature Measurements: Record the temperature every 30 seconds as the pure solvent and two solutions are cooled. Note the temperature at which any solid first appears.
| Time Elapsed (minutes) | Temperature (°C) | ||
|---|---|---|---|
| --- | Pure Solvent (water only) |
Solution I (water + 3 g sugar) |
Solution II (water + 5 g sugar) |
| 0 | |||
| 0.5 | |||
| 1.5 | |||
| 2 | |||
| 2.5 | |||
| 3 | |||
| 3.5 | |||
| 4 | |||
| 4.5 | |||
| 5 | |||
| 5.5 | |||
| 6 | |||
| 6.5 | |||
| 7 | |||
| 7.5 | |||
| 8 | |||
| 8.5 | |||
| 9 | |||
| 9.5 | |||
| 10 | |||
| 10.5 | |||
| 11 | |||
| 11.5 | |||
| 12 | |||
| 12.5 | |||
| 13 | |||
| 13.5 | |||
Graphical Analysis of Data
Use Excel to create three separate graphs of “Temperature versus Time” for the pure solvent and the two solutions studied. Each graph should have an appropriate title and labeled axes with an appropriate scale. Add two trendlines to the data points of each graph. You can do this by hand with a ruler or by using Excel. The first line is applied to data points that correspond to the cooling of the liquid stat: these are the points on the steep part of the graph. The second line is applied to data points that correspond to the co-existence of both the solid and liquid (freezing): these are the points on the part of the graph where the temperature levels out. Extrapolate the two trendlines towards each other until they intersect. The temperature at the point of intersection is the solvent freezing point and should be clearly shown on each graph. Attach your three graphs to this report.
Record the freezing point temperatures obtained from the graphs below:
- Pure water________________°C
- Solution I _________________°C
- Solution II ________________°C
Calculation of Molar Mass
Complete the table below with the results of your calculations. Be sure to include all units. Note that \(K_{f}\) PDB = 7.10 °C·kg·mol-1.
|
Solution I |
Solution II |
|
|---|---|---|
|
Mass of water added |
||
|
Total mass of unknown added |
||
|
Freezing point of pure water |
||
|
Freezing point of solution |
||
|
Total Freezing point depression, \(\Delta T_{f}\) |
||
|
Molality of solution |
||
|
Moles of unknown in solution |
||
|
Molar Mass of unknown |
- Unknown number ______________________ has an average molar mass of _______________________g/mol.
- On a separate sheet of paper, show the equations used and calculations performed for molality of the solutions, moles of unknown in the solutions, and molar mass of the unknown. Attach your sheet to this report.
Questions
- Look up the freezing point of pure water This may be found in the CRC Handbook of Chemistry and Physics (library) or from various online sources.
- Freezing Point of water =_________________________
- Reference source used:
- Using the freezing point from your reference source, determine the percentage error in your experimentally measured freezing point. Show your calculation below, and be sure to report your answer to the correct number of significant figures.
- Suppose you find out that the actual molar mass of your unknown solid is exactly three times larger than the value you determined experimentally. What could you conclude about the nature of your unknown solid and the assumptions you made in your calculations in such a case? Explain.
Pre-Laboratory Assignment: Determination of Molar Mass by Freezing Point Depression
- Camphor melts at 179.8oC and has a freezing point depression constant, \(K_{f}\) = 40 oC/molal. When 0.186 g of an unknown organic solid is dissolved in 22.01 g of liquid camphor, the freezing point of the mixture is found to be 176.7oC. What is the molar mass of the solute?
- A particular unknown solid depresses the freezing point of PDB (\(K_{f}\) = 7.1oC/molal) by 3.5oC. If you dissolve the same solid in \(\ce{H2O}\) (\(K_{f}\) = 1.86oC/molal) to the same final molality as you had it in the PDB, what would the change in freezing point be?
- You are instructed to add about 2 g of the unknown solid when determining the freezing point of the solution. Suppose you accidentally add 5 g of solid. Will the measured freezing point of the solution be higher or lower as a result of this mistake? Explain.
- Cleaning PDB from the test tube after the experiment could be a long and arduous process. Describe the procedure that you will use to clean PDB (and the unknown solid) from your test tube after your experiments are complete.
Contributors and Attributions
The experiment is from Chem 12 Experiments shared under a CC BY-NC license and was authored, remixed, and/or curated by Santa Monica College. Manjusha Saraswathiamma, Minnesota State Community and Technical College, Moorhead, has modified this to fit to a homeschool environment with less hazardous and cost-effective lab supplies.Manjusha like to ackwoledge the creators of the YouTube video embedded on this page.