15.2: Carboxylic Acids - Structures and Names
- Page ID
- 492385
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Name carboxylic acids with common names.
- Name carboxylic acids according to IUPAC nomenclature.
Carboxylic acids occur widely in nature, often combined with alcohols or other functional groups, as in fats, oils, and waxes. They are components of many foods, medicines, and household products (Figure \(\PageIndex{1}\)). Not surprisingly, many of them are best known by common names based on Latin and Greek words that describe their source.

The simplest carboxylic acid, formic acid (HCOOH), was first obtained by the distillation of ants (Latin formica, meaning “ant”). The bites of some ants inject formic acid, and the stings of wasps and bees contain formic acid (as well as other poisonous materials).

The next higher homolog is acetic acid, which is made by fermenting cider and honey in the presence of oxygen. This fermentation produces vinegar, a solution containing 4%–10% acetic acid, plus a number of other compounds that add to its flavor. Acetic acid is probably the most familiar weak acid used in educational and industrial chemistry laboratories.
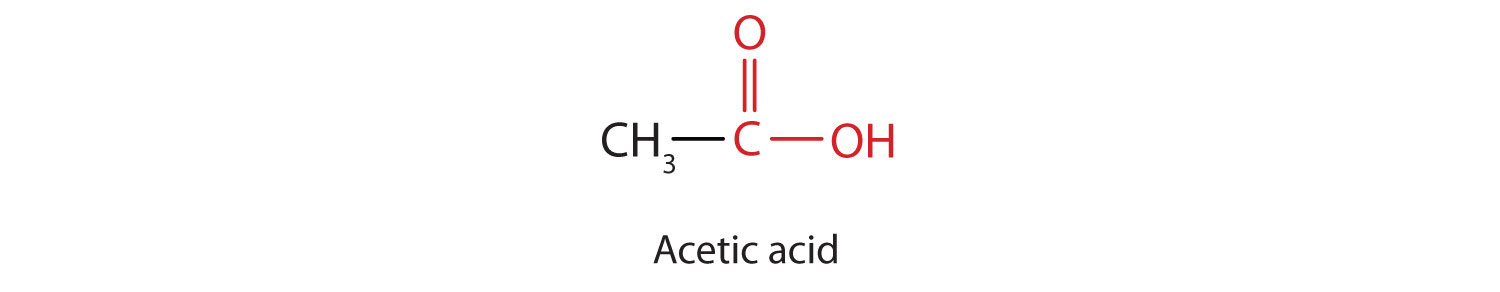
Pure acetic acid solidifies at 16.6°C, only slightly below normal room temperature. In the poorly heated laboratories of the late 19th and early 20th centuries in northern North America and Europe, acetic acid often “froze” on the storage shelf. For that reason, pure acetic acid (sometimes called concentrated acetic acid) came to be known as glacial acetic acid, a name that survives to this day.
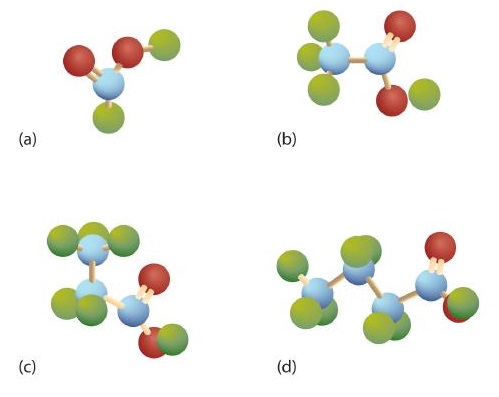
The third homolog, propionic acid (CH3CH2COOH), is seldom encountered in everyday life. The fourth homolog, butyric acid (CH3CH2CH2COOH), is one of the most foul-smelling substances imaginable. It is found in rancid butter and is one of the ingredients of body odor. By recognizing extremely small amounts of this and other chemicals, bloodhounds are able to track fugitives. Models of the first four carboxylic acids are shown in Figure \(\PageIndex{2}\).
The acid with the carboxyl group attached directly to a benzene ring is called benzoic acid (C6H5COOH).

The common names of carboxylic acids use Greek letters (α, β, γ, δ, and so forth), not numbers, to designate the position of substituent groups in acids. These letters refer to the position of the carbon atom in relation to the carboxyl carbon atom.
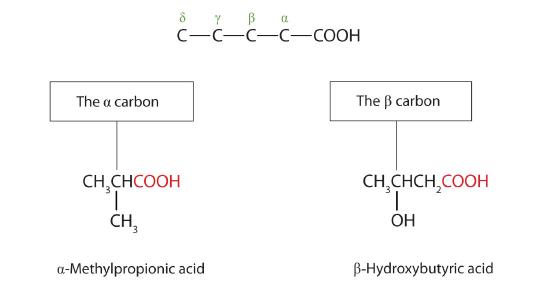
In the nomenclature system of the International Union of Pure and Applied Chemistry (IUPAC), the parent hydrocarbon is the one that corresponds to the longest continuous chain (LCC) containing the carboxyl group. The -e ending of the parent alkane is replaced by the suffix -oic and the word acid. For example, the carboxylic acid derived from pentane is pentanoic acid (CH3CH2CH2CH2COOH). As with aldehydes, the carboxyl carbon atom is counted first; numbers are used to indicate any substituted carbon atoms in the parent chain.
Greek letters are used with common names; numbers are used with IUPAC names.
Table of the First 10 Carboxylic Acids
| IUPAC Name | Common Name | Structural Formula |
|---|---|---|
| Methanoic Acid | Formic Acid | HCOOH |
| Ethanoic Acid | Acetic Acid | CH₃COOH |
| Propanoic Acid | Propionic Acid | CH₃CH₂COOH |
| Butanoic Acid | Butyric Acid | CH₃(CH₂)₂COOH |
| Pentanoic Acid | Valeric Acid | CH₃(CH₂)₃COOH |
| Hexanoic Acid | Caproic Acid | CH₃(CH₂)₄COOH |
| Heptanoic Acid | Enanthic Acid | CH₃(CH₂)₅COOH |
| Octanoic Acid | Caprylic Acid | CH₃(CH₂)₆COOH |
| Nonanoic Acid | Pelargonic Acid | CH₃(CH₂)₇COOH |
| Decanoic Acid | Capric Acid | CH₃(CH₂)₈COOH |
Give the common and IUPAC names for each compound.
- ClCH2CH2CH2COOH
Solution
- The LCC contains four carbon atoms; the compound is therefore named as a substituted butyric (or butanoic) acid.
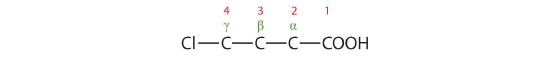
The chlorine atom is attached to the γ-carbon in the common system or C4 in the IUPAC system. The compound is γ-chlorobutyric acid or 4-chlorobutanoic acid.
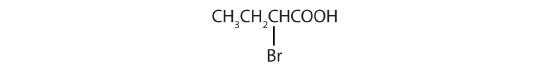
- The LCC contains four carbon atoms; the compound is therefore named as a substituted butyric (or butanoic) acid.
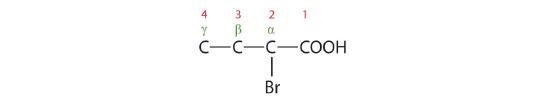
The bromine (Br) atom is at the α-carbon in the common system or C2 in the IUPAC system. The compound is α-bromobutyric acid or 2-bromobutanoic acid.
Give the IUPAC name for each compound.
- ClCH2CH2CH2CH2COOH
- (CH3)2CHCH2CHBrCOOH
Write the condensed structural formula for β-chloropropionic acid.
Solution
Propionic acid has three carbon atoms: C–C–COOH. Attach a chlorine (Cl) atom to the parent chain at the beta carbon atom, the second one from the carboxyl group: Cl–C–C–COOH. Then add enough hydrogen atoms to give each carbon atom four bonds: ClCH2CH2COOH.
Write the condensed structural formula for 4-bromo-5-methylhexanoic acid.
Key Takeaways
- Simple carboxylic acids are best known by common names based on Latin and Greek words that describe their source (e.g., formic acid, Latin formica, meaning “ant”).
- Greek letters, not numbers, designate the position of substituted acids in the common naming convention.
- IUPAC names are derived from the LCC of the parent hydrocarbon with the -e ending of the parent alkane replaced by the suffix -oic and the word acid.
IUPAC and Common Nomenclature for Carboxylic Acids
IUPAC Naming Rules
- Identify the Longest Carbon Chain: The chain containing the carboxyl group (-COOH) is identified.
- Replace the Alkane Suffix: The suffix “-e” of the parent alkane is replaced with “-oic acid.”
- Number the Chain: The carboxyl carbon is always numbered as 1.
- Name Substituents: Identify and name any substituents attached to the main chain.
Examples:
- Methanoic Acid: HCOOH (Formic Acid)
- Ethanoic Acid: CH₃COOH (Acetic Acid)
- Propanoic Acid: CH₃CH₂COOH (Propionic Acid)
Carboxylic Acids with Alkyl Group Substituents
- Identify and Number the Longest Chain: The chain containing the carboxyl group is numbered starting from the carboxyl carbon.
- Name the Alkyl Substituents: Name and number the alkyl groups attached to the main chain.
- Combine the Names: Combine the names of the substituents with the base name of the carboxylic acid.
Examples:
- 2-Methylpropanoic Acid: (CH₃)₂CHCOOH (Isobutyric Acid)
- 3-Methylbutanoic Acid: CH₃CH₂CH(CH₃)COOH (Isovaleric Acid)
- 2,2-Dimethylpropanoic Acid: (CH₃)₃CCOOH (Pivalic Acid)
Common Naming Rules
- Historical Names: Many carboxylic acids have common names based on their historical sources or properties.
- Prefixes and Suffixes: Common names often use prefixes like “form-” for one carbon, “acet-” for two carbons, etc.
Examples:
- Formic Acid: HCOOH (Methanoic Acid)
- Acetic Acid: CH₃COOH (Ethanoic Acid)
- Propionic Acid: CH₃CH₂COOH (Propanoic Acid)
These guidelines help in systematically naming carboxylic acids, ensuring clarity and consistency in chemical communication.


