7.01: Types of Chemical Reactions - Double Displacement Reactions
- Page ID
- 178145
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)
Learning Objectives
- Recognize chemical reactions as double-replacement reactions.
- Use the periodic table and solubility rules to predict whether double-replacement reactions will occur.
Up to now, we have presented chemical reactions as a topic, but we have not discussed how the products of a chemical reaction can be predicted. Here we will begin our study of certain types of chemical reactions that allow us to predict what the products of the reaction will be.
A double-replacement reaction occurs when parts of two ionic compounds are exchanged, making two new compounds. A characteristic of a double-replacement equation is that there are two compounds as reactants and two different compounds as products. An example is
\[\ce{CuCl2(aq) + 2AgNO3(aq) → Cu(NO3)2(aq) + 2AgCl(s)}\]
There are two equivalent ways of considering a double-replacement equation: either the cations are swapped, or the anions are swapped. (You cannot swap both; you would end up with the same substances you started with.) Either perspective should allow you to predict the proper products, as long as you pair a cation with an anion and not a cation with a cation or an anion with an anion.
Example \(\PageIndex{1}\)
What cations and anions are participating in this chemical reaction?
\[\ce{BaCl2 + Na2SO4 → }\]
Solution
Looking at a periodic table we can determine the charges for the barium ion, the chloride ion and the sodium ion:
Ba2+ (barium is in group 2A)
Na+ (sodium is in group 1A)
Cl- (chloride is in group 7A)
Our final anion is a polyatomic ion, so we can't use the periodic table if we don't have the charge on sulfate memorized, but we can use the charge on sodium to figure it out.
We need two Na+ to cancel out the charge on our SO4 so the charge on our sulfate has to be 2-:
2(+1) + x = 0 --> x = -2
All together we have Ba2+, Cl-, Na+ and SO42- participating in our reaction.
Example \(\PageIndex{2}\)
Predict the products of this double-replacement equation:
\[\ce{BaCl2 + Na2SO4 → }\]
Solution
We saw in example \(\PageIndex{1}\) that the ions participating in the reaction were Ba2+, Cl-, Na+ and SO42-. We know that like charges repel each other, so our anions (Ba2+ and Na+) are not going to come together, same thing goes for our cations (Cl- and SO42-). For a chemical reaction to have taken place something has to have changed so we can't put the barium back with the chloride and the sodium back with the sulfate. This leaves pairing the barium ion with our sulfate and the sodium ion with our chloride as our only option.
\[\ce{Ba^2+ + SO4^2- → BaSO4}\]
\[\ce{Na+ + Cl- → NaCl}\]
Putting this all together and balancing our chemical equation we get
\[\ce{BaCl2 + Na2SO4 → BaSO4 + 2 NaCl}\]
Exercise \(\PageIndex{3}\)
Predict the products of this double-replacement equation:
\[\ce{KBr + AgNO3 → }\]
Answer
KNO3 and AgBr
Predicting whether a double-replacement reaction occurs is somewhat more difficult than predicting a single-replacement reaction. However, there is one type of double-replacement reaction that we can predict: the precipitation reaction. A precipitation reaction occurs when two ionic compounds are dissolved in water and form a new ionic compound that does not dissolve; this new compound falls out of solution as a solid precipitate. The formation of a solid precipitate is the driving force that makes the reaction proceed.
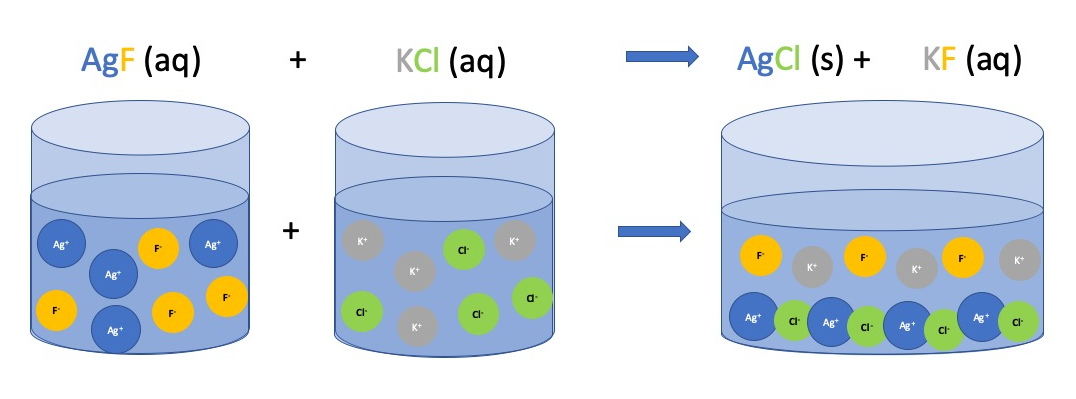
Figure \(\PageIndex{1}\): Illustration of the double-replacement reaction between aqueous silver fluoride (AgF) and aqueous potassium chloride (KCl).
As illustrated in Figure \(PageIndex{1}\) upon mixing the AgF solution and the KCl solution the silver ion (Ag+) and the chloride ion (Cl-) combine to form a insoluble salt, the precipitate. As the salt is insoluble it will not dissolve in water and we use the (s) symbol for solid. If no precipitate formed we would essentially have given the silver, fluoride, potassium and chloride ions a change of scenery, but they would still be floating around as ions. In this case no reaction occurs.
.jpg?revision=2)
Figure \(\PageIndex{1}\): The formation of a silver chloride precipitate. The precipitate is the white solid we see appearing in the solution
By Luisbrudna - Own work, CC BY-SA 4.0, https://commons.wikimedia.org/w/inde...curid=40139259
To judge whether double-replacement reactions will occur, we need to know what kinds of ionic compounds form precipitates. For this, we use solubility rules, which are general statements that predict which ionic compounds dissolve (are soluble) and which do not (are not soluble or insoluble). Table 4.3.1 - Some Useful Solubility Rules, lists some general solubility rules. We need to consider each ionic compound (both the reactants and the possible products) in light of the solubility rules. If a compound is soluble, we use the (aq) label with it, indicating it dissolves. If a compound is not soluble, we use the (s) label with it and assume that it will precipitate out of solution. If everything is soluble, then no reaction will be expected.
| These compounds generally dissolve in water (are soluble): | Exceptions: |
| All compounds of Li+, Na+, K+, Rb+, Cs+, and NH4+ | None |
| All compounds of NO3− and C2H3O2− | None |
| Compounds of Cl−, Br−, I− | Ag+, Hg22+, Pb2+ |
| Compounds of SO42- | Hg22+, Pb2+, Sr2+, Ba2+ |
| These compounds generally do not dissolve in water (are insoluble): | Exceptions: |
| Compounds of CO32− and PO43− | Compounds of Li+, Na+, K+, Rb+, Cs+, and NH4+ |
| Compounds of OH− | Compounds of Li+, Na+, K+, Rb+, Cs+, NH4+, Sr2+, and Ba2+ |
For example, consider the possible double-replacement reaction between Na2SO4 and SrCl2. The solubility rules say that all ionic sodium compounds are soluble and all ionic chloride compounds are soluble except for Ag+, Hg22+, and Pb2+, which are not being considered here. Therefore, Na2SO4 and SrCl2 are both soluble. The possible double-replacement reaction products are NaCl and SrSO4. Are these soluble? NaCl is (by the same rule we just quoted), but what about SrSO4? Compounds of the sulfate ion are generally soluble, but Sr2+ is an exception: we expect it to be insoluble-a precipitate. Therefore, we expect a reaction to occur, and the balanced chemical equation would be
\[\ce{Na2SO4(aq) + SrCl2(aq) → 2NaCl(aq) + SrSO4(s)}\]
You would expect to see a visual change corresponding to SrSO4 precipitating out of solution (Figure \(\PageIndex{2}\)).

Example \(\PageIndex{4}\):
Will a double-replacement reaction occur? If so, identify the products.
- Ca(NO3)2 + KBr → ?
- NaOH + FeCl2 → ?
Solution
- According to the solubility rules, both Ca(NO3)2 and KBr are soluble. Now we consider what the double-replacement products would be by switching the cations (or the anions)-namely, CaBr2 and KNO3. However, the solubility rules predict that these two substances would also be soluble, so no precipitate would form. Thus, we predict no reaction in this case.
-
According to the solubility rules, both NaOH and FeCl2 are expected to be soluble. If we assume that a double-replacement reaction may occur, we need to consider the possible products, which would be NaCl and Fe(OH)2. NaCl is soluble, but, according to the solubility rules, Fe(OH)2 is not. Therefore, a reaction would occur, and Fe(OH)2(s) would precipitate out of solution. The balanced chemical equation is \[\ce{2NaOH(aq) + FeCl2(aq) → 2NaCl(aq) + Fe(OH)2(s)}\]
Exercise \(\PageIndex{4}\)
\[\ce{Sr(NO3)2 + KCl → }\]
Answer
No reaction; all possible products are soluble.
Key Takeaways
- A double-replacement reaction exchanges the cations (or the anions) of two ionic compounds.
- A precipitation reaction is a double-replacement reaction in which one product is a solid precipitate.
- Solubility rules are used to predict whether some double-replacement reactions will occur.

