10.5: Organic Structural Representations: Condensed Structural Notation
- Page ID
- 237750
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Define condensed structural notation.
- Represent an organic molecule using condensed structural notation.
- Identify the compositional characteristics of a molecule that can be determined by analyzing a structure that is represented using condensed structural notation.
As stated previously, organic chemists use several different notations to represent organic molecules. While a molecular formula only indicates the types and quantities of elements that are present in a molecule, nearly all of the compositional characteristics of an organic substance can be determined by analyzing a structure that is symbolized using expanded structural notation. The types of elements that are present are indicated by the elemental symbols that are shown in the corresponding structural representation. The quantity of each element, as well as the total number of bonds and lone pairs that are present in a molecule, can be established by counting the number of times that each symbol is written in a molecular image. Furthermore, the connectivity, or relative bonding arrangement, of each atom, bond, and lone pair can be explained by describing the placement of each of these components, relative to one another, in an organic structural representation. The types of bonds that are found in a molecule can be determined by counting the number of lines that are drawn between any two atoms in the corresponding image. Because structures that are drawn using Valence Shell Electron Pair Repulsion notation reflect the three-dimensional spatial orientations of organic molecules and their constituent atoms, in addition to all of the compositional characteristics that are listed above above, VSEPR notation is the most comprehensive of the organic structural representations. However, since organic molecules can contain thousands of individual carbon atoms, the expanded and VSEPR images of these substances can become prohibitively-large in size. Furthermore, representing large molecules using these notations is incredibly time-consuming, due to the amount of detail that must be incorporated into the pictures that are generated. Therefore, in order to visually-represent organic molecules using smaller and, therefore, more manageable, images, organic chemists have developed two additional drawing conventions, called condensed structural notation and bond-line notation, which will be discussed in the current and following section of this chapter, respectively.
Images that are symbolized using condensed structural notation, which uses a "formula-style" format to indicate the number of hydrogens that are bonded to a particular non-metal, are generated by eliminating specific structural components from a provided expanded or VSEPR picture. Therefore, condensed structures are substantially smaller in scale, compared to the structures from which they are derived, and are relatively simple to draw, since fewer details must be incorporated into this type of structural representation. In particular,
- all lone pairs,
- all carbon/hydrogen bonds, and
- all single bonds that are written in a horizontal, or left-to-right, direction
should be removed when generating a condensed structure from an image that is drawn using a more detailed notation. The locations and relative orientations of all double and triple bonds, as well as all vertical and angled single bonds, should be maintained and, therefore, are directly translated from a given picture to the corresponding positions in a condensed structure. Finally, all hydrogen groupings should be represented using a "JHQ" format, in which the generic placeholder letter "J" should be replaced with the elemental symbol of a non-metal, and the "Q" subscript corresponds to the number of hydrogens that are bonded to that atom. If only a single hydrogen is associated with a particular atom, no subscript should be written, since values of "1" are usually implicitly-understood in chemistry.
For example, represent each of the following organic molecular structures using condensed structural notation.
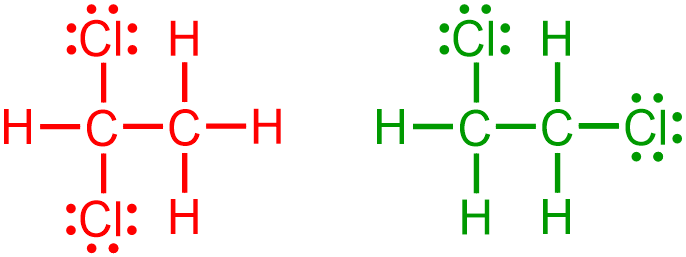
A condensed structure is generated by eliminating specific structural components from a provided expanded or VSEPR picture. In particular, when generating a condensed structure from an image that is represented in a more detailed notation, all lone pairs,
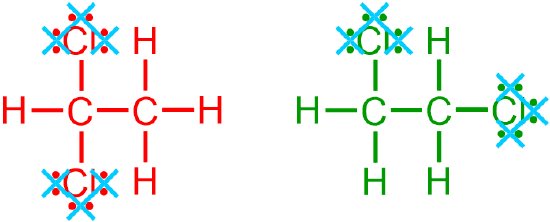
all carbon/hydrogen bonds,
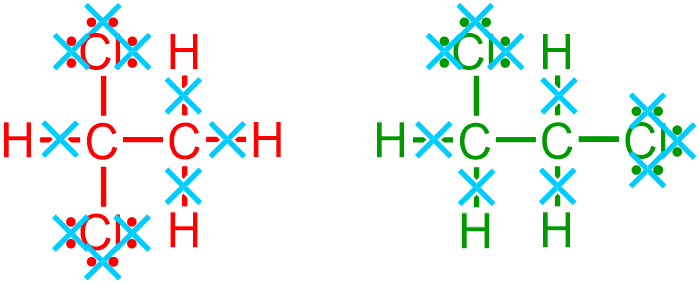
and all single bonds that are written in a horizontal, or left-to-right, direction,
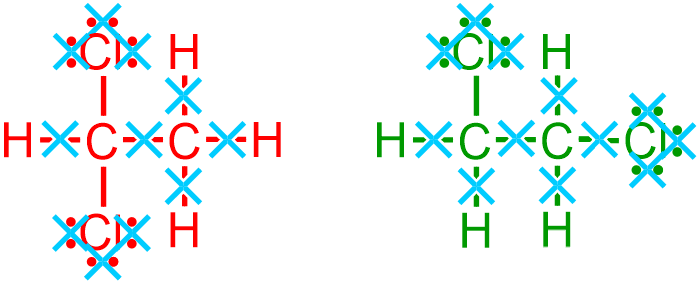
are removed, as indicated by the light blue " " symbols in the images that are shown above. The locations and relative orientations of the remaining vertical single bonds should be maintained and, therefore, are directly translated from the given picture to the corresponding positions in the condensed structure that is being developed, as shown below.
" symbols in the images that are shown above. The locations and relative orientations of the remaining vertical single bonds should be maintained and, therefore, are directly translated from the given picture to the corresponding positions in the condensed structure that is being developed, as shown below.

Finally, all hydrogen groupings are represented using a "JHQ" format, in which the generic placeholder letter "J" is replaced with the elemental symbol of a non-metal, and the "Q" subscript corresponds to the number of hydrogens that are bonded to that atom. The first and second carbon atoms in the first given structure are bonded to one hydrogen and three hydrogens, respectively. Because values of "1" are usually implicitly-understood in chemistry, these groupings are represented as "CH" and "CH3," respectively, in the condensed structure that is being developed. Furthermore, since both of the carbon atoms in the second given structure are bonded to two hydrogens, these groupings are both condensed as "CH2," as shown below. The following pictures, which are substantially smaller in scale, relative to the expanded structures from which they were derived, are both chemically-correct condensed structural representations of the given images.
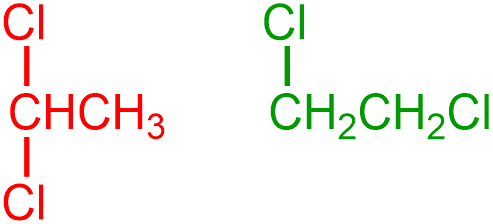
Nearly all of the compositional characteristics of a molecule can be determined by analyzing a structure that is symbolized using condensed structural notation. The identities of the elements that are present in a molecule are indicated by the elemental symbols that are shown in a condensed structure, and the quantity of each element is established by adding the subscripts that are associated with the corresponding symbols. The total number of bonds and lone pairs that are present in a molecule, as well as the connectivity of each atom, bond, and lone pair, can be inferred by applying an understanding of standard non-metal bonding configurations. Finally, the types of bonds that are found in a molecule can be determined by explicitly observing the presence of double and triple bonds and by recognizing that all other atoms are connected using single bonds. However, because condensed structural notation is a two-dimensional drawing convention, images that are drawn using this type of representation do not correctly depict the spatial orientations of organic molecules or their constituent atoms.

