6.4 Solution Concentration Units
- Page ID
- 218400
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Skills to Develop
- To describe the concentrations of solutions quantitatively
- To describe the concentration of a solution in the way that is most appropriate for a particular problem or application.
- To be familiar with the different units used to express the concentrations of a solution.
Many people have a qualitative idea of what is meant by concentration. Anyone who has made instant coffee or lemonade knows that too much powder gives a strongly flavored, highly concentrated drink, whereas too little results in a dilute solution that may be hard to distinguish from water. In chemistry, the concentration of a solution is the quantity of a solute that is contained in a particular quantity of solvent or solution. Knowing the concentration of solutes is important in controlling the stoichiometry of reactants for solution reactions. Chemists use many different methods to define concentrations, some of which are described in this section.
Molarity
The most common unit of concentration is molarity, which is also the most useful for calculations involving the stoichiometry of reactions in solution. The molarity (M) is defined as the number of moles of solute present in exactly 1 L of solution. It is, equivalently, the number of millimoles of solute present in exactly 1 mL of solution:
\[ molarity = \dfrac{moles\: of\: solute}{liters\: of\: solution} = \dfrac{mmoles\: of\: solute} {milliliters\: of\: solution} \label{4.5.1}\]
The units of molarity are therefore moles per liter of solution (mol/L), abbreviated as \(M\). An aqueous solution that contains 1 mol (342 g) of sucrose in enough water to give a final volume of 1.00 L has a sucrose concentration of 1.00 mol/L or 1.00 M. In chemical notation, square brackets around the name or formula of the solute represent the molar concentration of a solute. Therefore,
\[[\rm{sucrose}] = 1.00\: M\]
is read as “the concentration of sucrose is 1.00 molar.” The relationships between volume, molarity, and moles may be expressed as either
\[ V_L \, x \, M_{mol/L} = \cancel{L} \left( \dfrac{mol}{\cancel{L}} \right) = moles \label{4.5.2}\]
or
\[ V_{mL} \, x \, M_{mmol/mL} = \cancel{mL} \left( \dfrac{mmol} {\cancel{mL}} \right) = mmoles \label{4.5.3}\]
Figure \(\PageIndex{1}\) illustrates the use of Equations \(\ref{4.5.2}\) and \(\ref{4.5.3}\).

Figure \(\PageIndex{1}\): Preparation of a Solution of Known Concentration Using a Solid Solute
Example \(\PageIndex{1}\): Calculating Moles from Concentration of NaOH
Calculate the number of moles of sodium hydroxide (NaOH) in 2.50 L of 0.100 M NaOH.
Given: identity of solute and volume and molarity of solution
Asked for: amount of solute in moles
Strategy:
Use either Equation \ref{4.5.2} or Equation \ref{4.5.3}, depending on the units given in the problem.
Solution:
Because we are given the volume of the solution in liters and are asked for the number of moles of substance, Equation \ref{4.5.2} is more useful:
\( moles\: NaOH = V_L \, x \, M_{mol/L} = (2 .50\: \cancel{L} ) \left( \dfrac{0.100\: mol } {\cancel{L}} \right) = 0 .250\: mol\: NaOH \)
Exercise \(\PageIndex{1}\): Calculating Moles from Concentration of Alanine
Calculate the number of millimoles of alanine, a biologically important molecule, in 27.2 mL of 1.53 M alanine.
- Answer
-
41.6 mmol
The Preparation of Solutions
To prepare a solution that contains a specified concentration of a substance, it is necessary to dissolve the desired number of moles of solute in enough solvent to give the desired final volume of solution. Figure \(\PageIndex{1}\) illustrates this procedure for a solution of cobalt(II) chloride dihydrate in ethanol. Note that the volume of the solvent is not specified. Because the solute occupies space in the solution, the volume of the solvent needed is almost always less than the desired volume of solution. For example, if the desired volume were 1.00 L, it would be incorrect to add 1.00 L of water to 342 g of sucrose because that would produce more than 1.00 L of solution. As shown in Figure \(\PageIndex{2}\), for some substances this effect can be significant, especially for concentrated solutions.
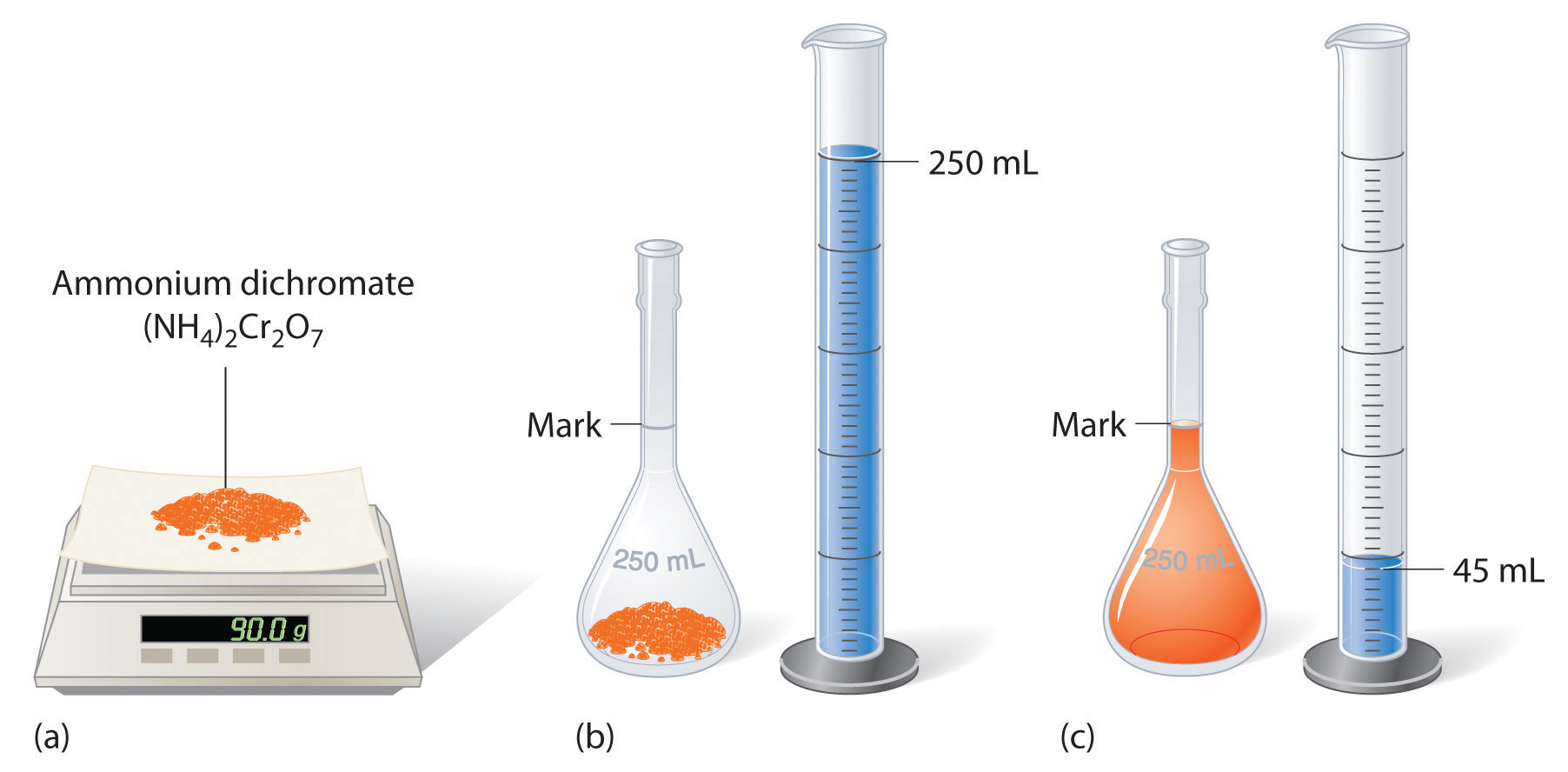
Figure \(\PageIndex{2}\): Preparation of 250 mL of a Solution of (NH4)2Cr2O7 in Water. The solute occupies space in the solution, so less than 250 mL of water are needed to make 250 mL of solution.
Example \(\PageIndex{2}\)
The solution contains 10.0 g of cobalt(II) chloride dihydrate, CoCl2•2H2O, in enough ethanol to make exactly 500 mL of solution. What is the molar concentration of \(\ce{CoCl2•2H2O}\)?
Given: mass of solute and volume of solution
Asked for: concentration (M)
Strategy:
To find the number of moles of \(\ce{CoCl2•2H2O}\), divide the mass of the compound by its molar mass. Calculate the molarity of the solution by dividing the number of moles of solute by the volume of the solution in liters.
Solution:
The molar mass of CoCl2•2H2O is 165.87 g/mol. Therefore,
\[ moles\: CoCl_2 \cdot 2H_2O = \left( \dfrac{10.0 \: \cancel{g}} {165 .87\: \cancel{g} /mol} \right) = 0 .0603\: mol \nonumber \]
The volume of the solution in liters is
\[ volume = 500\: \cancel{mL} \left( \dfrac{1\: L} {1000\: \cancel{mL}} \right) = 0 .500\: L \nonumber \]
Molarity is the number of moles of solute per liter of solution, so the molarity of the solution is
\[ molarity = \dfrac{0.0603\: mol} {0.500\: L} = 0.121\: M = CoCl_2 \cdot H_2O \nonumber \]
Exercise \(\PageIndex{2}\)
The solution shown in Figure \(\PageIndex{2}\) contains 90.0 g of (NH4)2Cr2O7 in enough water to give a final volume of exactly 250 mL. What is the molar concentration of ammonium dichromate?
- Answer
-
\[(NH_4)_2Cr_2O_7 = 1.43\: M \nonumber\]
To prepare a particular volume of a solution that contains a specified concentration of a solute, we first need to calculate the number of moles of solute in the desired volume of solution using the relationship shown in Equation \(\ref{4.5.2}\). We then convert the number of moles of solute to the corresponding mass of solute needed. This procedure is illustrated in Example \(\PageIndex{3}\).
Example \(\PageIndex{3}\): D5W Solution
The so-called D5W solution used for the intravenous replacement of body fluids contains 0.310 M glucose. (D5W is an approximately 5% solution of dextrose [the medical name for glucose] in water.) Calculate the mass of glucose necessary to prepare a 500 mL pouch of D5W. Glucose has a molar mass of 180.16 g/mol.
Given: molarity, volume, and molar mass of solute
Asked for: mass of solute
Strategy:
- Calculate the number of moles of glucose contained in the specified volume of solution by multiplying the volume of the solution by its molarity.
- Obtain the mass of glucose needed by multiplying the number of moles of the compound by its molar mass.
Solution:
A We must first calculate the number of moles of glucose contained in 500 mL of a 0.310 M solution:
\( V_L M_{mol/L} = moles \)
\( 500\: \cancel{mL} \left( \dfrac{1\: \cancel{L}} {1000\: \cancel{mL}} \right) \left( \dfrac{0 .310\: mol\: glucose} {1\: \cancel{L}} \right) = 0 .155\: mol\: glucose \)
B We then convert the number of moles of glucose to the required mass of glucose:
\( mass \: of \: glucose = 0.155 \: \cancel{mol\: glucose} \left( \dfrac{180.16 \: g\: glucose} {1\: \cancel{mol\: glucose}} \right) = 27.9 \: g \: glucose \)
Exercise \(\PageIndex{3}\)
Another solution commonly used for intravenous injections is normal saline, a 0.16 M solution of sodium chloride in water. Calculate the mass of sodium chloride needed to prepare 250 mL of normal saline solution.
- Answer
-
2.3 g NaCl
Dilution
A solution of a desired concentration can also be prepared by diluting a small volume of a more concentrated solution with additional solvent. A stock solution is a commercially prepared solution of known concentration and is often used for this purpose. Diluting a stock solution is preferred because the alternative method, weighing out tiny amounts of solute, is difficult to carry out with a high degree of accuracy. Dilution is also used to prepare solutions from substances that are sold as concentrated aqueous solutions, such as strong acids.
The procedure for preparing a solution of known concentration from a stock solution is shown in Figure \(\PageIndex{3}\). It requires calculating the number of moles of solute desired in the final volume of the more dilute solution and then calculating the volume of the stock solution that contains this amount of solute. Remember that diluting a given quantity of stock solution with solvent does not change the number of moles of solute present. The relationship between the volume and concentration of the stock solution and the volume and concentration of the desired diluted solution is therefore
\[(V_s)(M_s) = moles\: of\: solute = (V_d)(M_d)\label{4.5.4}\]
where the subscripts s and d indicate the concentrated stock solution and the desired dilute solution, respectively. Example \(\PageIndex{4}\) demonstrates the calculations involved in diluting a concentrated stock solution.
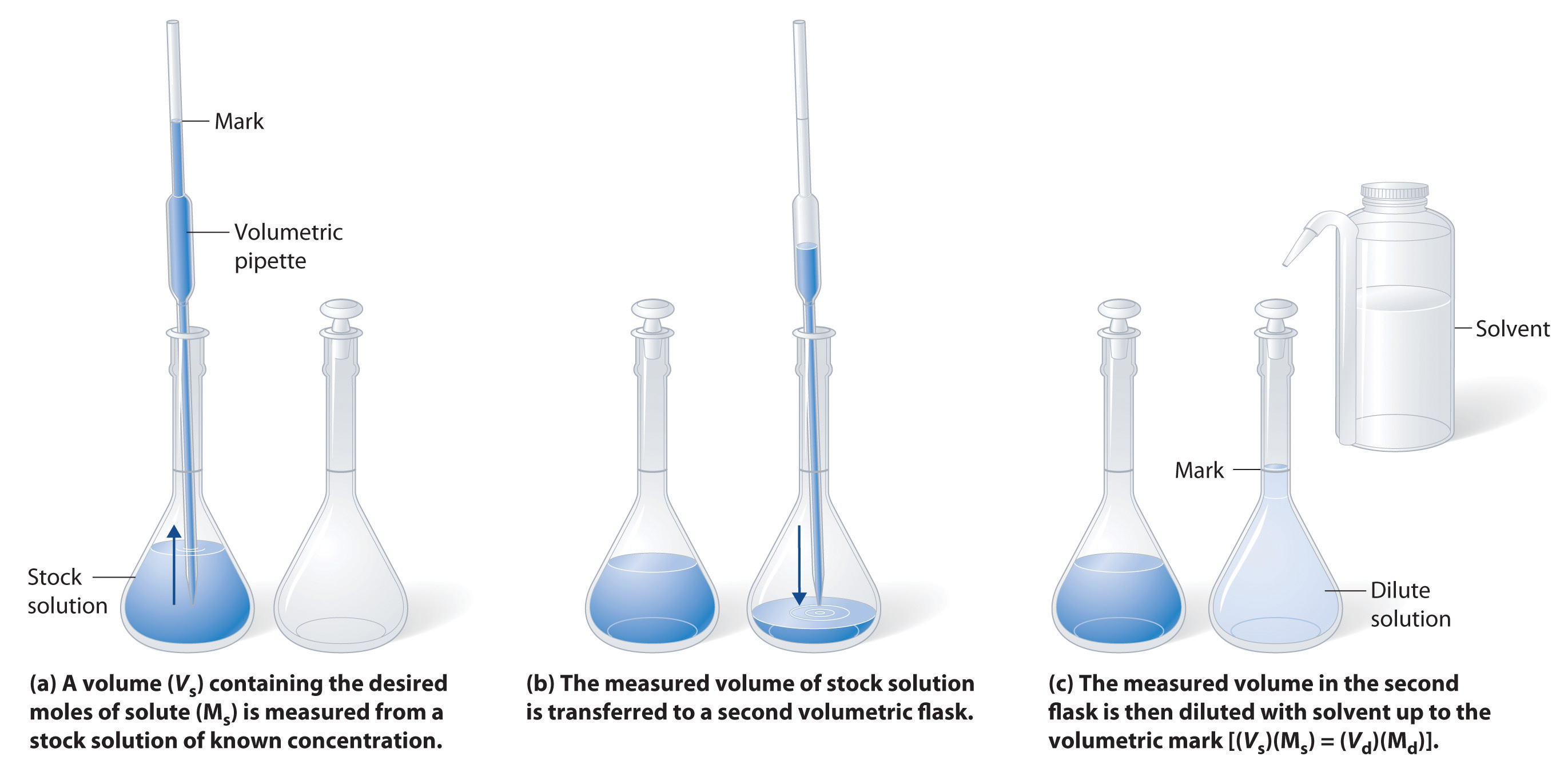
Figure \(\PageIndex{3}\): Preparation of a Solution of Known Concentration by Diluting a Stock Solution. (a) A volume (Vs) containing the desired moles of solute (Ms) is measured from a stock solution of known concentration. (b) The measured volume of stock solution is transferred to a second volumetric flask. (c) The measured volume in the second flask is then diluted with solvent up to the volumetric mark [(Vs)(Ms) = (Vd)(Md)].
Example \(\PageIndex{4}\)
What volume of a 3.00 M glucose stock solution is necessary to prepare 2500 mL of the D5W solution in Example \(\PageIndex{3}\)?
Given: volume and molarity of dilute solution
Asked for: volume of stock solution
Strategy:
- Calculate the number of moles of glucose contained in the indicated volume of dilute solution by multiplying the volume of the solution by its molarity.
- To determine the volume of stock solution needed, divide the number of moles of glucose by the molarity of the stock solution.
Solution:
A The D5W solution in Example 4.5.3 was 0.310 M glucose. We begin by using Equation 4.5.4 to calculate the number of moles of glucose contained in 2500 mL of the solution:
\[ moles\: glucose = 2500\: \cancel{mL} \left( \dfrac{1\: \cancel{L}} {1000\: \cancel{mL}} \right) \left( \dfrac{0 .310\: mol\: glucose} {1\: \cancel{L}} \right) = 0 .775\: mol\: glucose \]
B We must now determine the volume of the 3.00 M stock solution that contains this amount of glucose:
\[ volume\: of\: stock\: soln = 0 .775\: \cancel{mol\: glucose} \left( \dfrac{1\: L} {3 .00\: \cancel{mol\: glucose}} \right) = 0 .258\: L\: or\: 258\: mL \]
In determining the volume of stock solution that was needed, we had to divide the desired number of moles of glucose by the concentration of the stock solution to obtain the appropriate units. Also, the number of moles of solute in 258 mL of the stock solution is the same as the number of moles in 2500 mL of the more dilute solution; only the amount of solvent has changed. The answer we obtained makes sense: diluting the stock solution about tenfold increases its volume by about a factor of 10 (258 mL → 2500 mL). Consequently, the concentration of the solute must decrease by about a factor of 10, as it does (3.00 M → 0.310 M).
We could also have solved this problem in a single step by solving Equation 4.5.4 for Vs and substituting the appropriate values:
\[ V_s = \dfrac{( V_d )(M_d )}{M_s} = \dfrac{(2 .500\: L)(0 .310\: \cancel{M} )} {3 .00\: \cancel{M}} = 0 .258\: L \]
As we have noted, there is often more than one correct way to solve a problem.
Exercise \(\PageIndex{4}\)
What volume of a 5.0 M NaCl stock solution is necessary to prepare 500 mL of normal saline solution (0.16 M NaCl)?
- Answer
-
16 mL
Ion Concentrations in Solution
In Example \(\PageIndex{2}\), the concentration of a solution containing 90.00 g of ammonium dichromate in a final volume of 250 mL were calculated to be 1.43 M. Let’s consider in more detail exactly what that means. Ammonium dichromate is an ionic compound that contains two NH4+ ions and one Cr2O72− ion per formula unit. Like other many ionic compounds, it is a strong electrolyte that dissociates in aqueous solution to give hydrated NH4+ and Cr2O72− ions:
\[ (NH_4 )_2 Cr_2 O_7 (s) \xrightarrow {H_2 O(l)} 2NH_4^+ (aq) + Cr_2 O_7^{2-} (aq)\label{4.5.5} \]
Thus 1 mol of ammonium dichromate formula units dissolves in water to produce 1 mol of Cr2O72− anions and 2 mol of NH4+ cations (see Figure \(\PageIndex{4}\)).
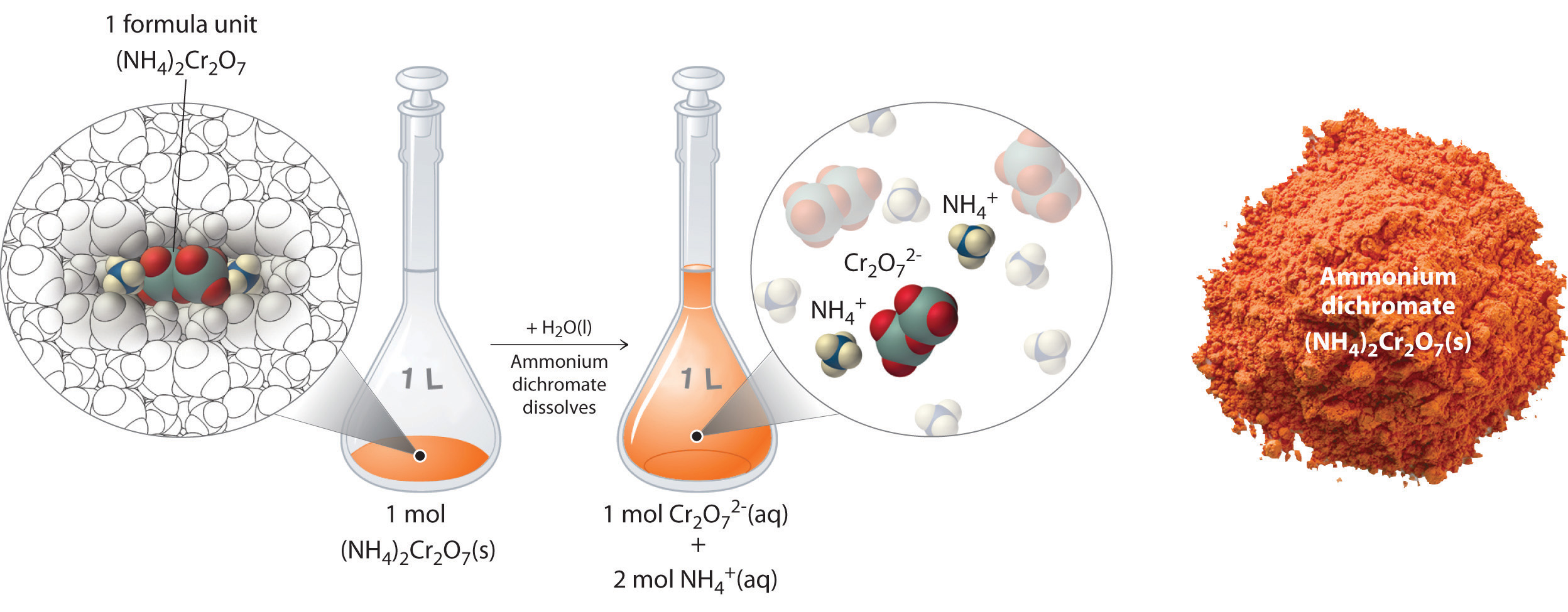
Figure \(\PageIndex{4}\): Dissolution of 1 mol of an Ionic Compound. In this case, dissolving 1 mol of (NH4)2Cr2O7 produces a solution that contains 1 mol of Cr2O72− ions and 2 mol of NH4+ ions. (Water molecules are omitted from a molecular view of the solution for clarity.)
When carrying out a chemical reaction using a solution of a salt such as ammonium dichromate, it is important to know the concentration of each ion present in the solution. If a solution contains 1.43 M (NH4)2Cr2O7, then the concentration of Cr2O72− must also be 1.43 M because there is one Cr2O72− ion per formula unit. However, there are two NH4+ ions per formula unit, so the concentration of NH4+ ions is 2 × 1.43 M = 2.86 M. Because each formula unit of (NH4)2Cr2O7 produces three ions when dissolved in water (2NH4+ + 1Cr2O72−), the total concentration of ions in the solution is 3 × 1.43 M = 4.29 M.
Example \(\PageIndex{5}\)
What are the concentrations of all species derived from the solutes in these aqueous solutions?
- 0.21 M NaOH
- 3.7 M (CH3)2CHOH
- 0.032 M In(NO3)3
Given: molarity
Asked for: concentrations
Strategy:
A Classify each compound as either a strong electrolyte or a nonelectrolyte.
B If the compound is a nonelectrolyte, its concentration is the same as the molarity of the solution. If the compound is a strong electrolyte, determine the number of each ion contained in one formula unit. Find the concentration of each species by multiplying the number of each ion by the molarity of the solution.
Solution:
-
Sodium hydroxide is an ionic compound that is a strong electrolyte (and a strong base) in aqueous solution:
\( NaOH(s) \xrightarrow {H_2 O(l)} Na^+ (aq) + OH^- (aq) \)B Because each formula unit of NaOH produces one Na+ ion and one OH− ion, the concentration of each ion is the same as the concentration of NaOH: [Na+] = 0.21 M and [OH−] = 0.21 M.
-
A The formula (CH3)2CHOH represents 2-propanol (isopropyl alcohol) and contains the –OH group, so it is an alcohol. Recall from Section 4.1 that alcohols are covalent compounds that dissolve in water to give solutions of neutral molecules. Thus alcohols are nonelectrolytes.
B The only solute species in solution is therefore (CH3)2CHOH molecules, so [(CH3)2CHOH] = 3.7 M.
-
A Indium nitrate is an ionic compound that contains In3+ ions and NO3− ions, so we expect it to behave like a strong electrolyte in aqueous solution:
\( In(NO _3 ) _3 (s) \xrightarrow {H_ 2 O(l)} In ^{3+} (aq) + 3NO _3^- (aq) \)
B One formula unit of In(NO3)3 produces one In3+ ion and three NO3− ions, so a 0.032 M In(NO3)3 solution contains 0.032 M In3+ and 3 × 0.032 M = 0.096 M NO3–—that is, [In3+] = 0.032 M and [NO3−] = 0.096 M.
Exercise \(\PageIndex{5}\)
What are the concentrations of all species derived from the solutes in these aqueous solutions?
- 0.0012 M Ba(OH)2
- 0.17 M Na2SO4
- 0.50 M (CH3)2CO, commonly known as acetone
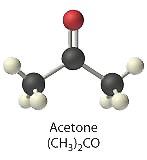
- Answer a
-
\([Ba^{2+}] = 0.0012\: M; \: [OH^-] = 0.0024\: M\)
- Answer b
-
\([Na^+] = 0.34\: M; \: [SO_4^{2-}] = 0.17\: M\)
- Answer c
-
\([(CH_3)_2CO] = 0.50\: M\)
Other Concentration Units
There are several different ways to quantitatively describe the concentration of a solution. For example, mole fraction, \(\dfrac{moles \, solute}{total \, moles \, of \, all \, components}\), is used not only to describe gas concentrations but also to determine the vapor pressures of mixtures of similar liquids. Example \(\PageIndex{6}\) shows the methods for calculating the molarity and mole fraction of a solution when the masses of its components are known.
Example \(\PageIndex{6}\): Molarity and Mole Fraction
Commercial vinegar is essentially a solution of acetic acid in water. A bottle of vinegar has 3.78 g of acetic acid per 100.0 g of solution. Assume that the density of the solution is 1.00 g/mL.
- What is its molarity?
- What is its mole fraction?
Given: mass of substance and mass and density of solution
Asked for: molarity and mole fraction
Strategy:
- Calculate the number of moles of acetic acid in the sample. Then calculate the number of liters of solution from its mass and density. Use these results to determine the molarity of the solution.
- Determine the mass of the water in the sample and calculate the number of moles of water. Then determine the mole fraction of acetic acid by dividing the number of moles of acetic acid by the total number of moles of substances in the sample.
Solution:
A The molarity is the number of moles of acetic acid per liter of solution. We can calculate the number of moles of acetic acid as its mass divided by its molar mass. The volume of the solution equals its mass divided by its density. The calculations follow:
\[\text{moles } \ce{CH_3CO_2H}=\dfrac{3.78\; \cancel{g}\; CH_3CO_2H}{60.05\; \cancel{g}/mol}=0.0629 \;mol \nonumber\]
\[\text{volume} =\dfrac{\text{mass}}{\text{density}}=\dfrac{100.0\; \cancel{g}\; \text{solution}}{1.00\; \cancel{g}/mL}=100\; mL\nonumber\]
\[ \begin{align*} \text{molarity of } \ce{CH_3CO_2H} &=\dfrac{\text{moles } \ce{CH3CO2H} }{\text{liter solution}} \\[5pt] &=\dfrac{0.0629\; mol\; \ce{CH_3CO_2H}}{(100\; \cancel{mL})(1\; L/1000\; \cancel{mL})}=0.629\; M \; \ce{CH_3CO_2H} \end{align*}\]
This result makes intuitive sense. If 100.0 g of aqueous solution (equal to 100 mL) contains 3.78 g of acetic acid, then 1 L of solution will contain 37.8 g of acetic acid, which is a little more than
B To calculate the mole fraction of acetic acid in the solution, we need to know the number of moles of both acetic acid and water. The number of moles of acetic acid is 0.0629 mol, as calculated in part (a). We know that 100.0 g of vinegar contains 3.78 g of acetic acid; hence the solution also contains (100.0 g − 3.78 g) = 96.2 g of water. We have
\[moles\; \ce{H_2O}=\dfrac{96.2\; \cancel{g}\; \ce{H_2O}}{18.02\; \cancel{g}/mol}=5.34\; mol\; \ce{H_2O}\nonumber\]
The mole fraction \(\chi\) of acetic acid is the ratio of the number of moles of acetic acid to the total number of moles of substances present:
\[ \begin{align*} \chi_{\ce{CH3CO2H}} &=\dfrac{moles\; \ce{CH_3CO_2H}}{moles \; \ce{CH_3CO_2H} + moles\; \ce{H_2O}} \\[5pt] &=\dfrac{0.0629\; mol}{0.0629 \;mol + 5.34\; mol} \\[5pt] &=0.0116=1.16 \times 10^{−2} \end{align*}\]
This answer makes sense, too. There are approximately 100 times as many moles of water as moles of acetic acid, so the ratio should be approximately 0.01.
Exercise \(\PageIndex{6}\): Molarity and Mole Fraction
A solution of \(\ce{HCl}\) gas dissolved in water (sold commercially as “muriatic acid,” a solution used to clean masonry surfaces) has 20.22 g of \(\ce{HCl}\) per 100.0 g of solution, and its density is 1.10 g/mL.
- What is its molarity?
- What is its mole fraction?
- Answer a
-
6.10 M HCl
- Answer b
-
\(\chi_{HCl} = 0.111\)
The concentration of a solution can also be described by its molality (m), the number of moles of solute per kilogram of solvent:
\[ \text{molality (m)} =\dfrac{\text{moles solute}}{\text{kilogram solvent}} \label{Eq10}\]
Molality, therefore, has the same numerator as molarity (the number of moles of solute) but a different denominator (kilogram of solvent rather than liter of solution). For dilute aqueous solutions, the molality and molarity are nearly the same because dilute solutions are mostly solvent. Thus because the density of water under standard conditions is very close to 1.0 g/mL, the volume of 1.0 kg of \(H_2O\) under these conditions is very close to 1.0 L, and a 0.50 M solution of \(KBr\) in water, for example, has approximately the same concentration as a 0.50 m solution.
Another common way of describing concentration is as the ratio of the mass of the solute to the total mass of the solution. The result can be expressed as percent by mass, parts per million (ppm), or parts per billion (ppb):
\[ \begin{align} \text{percent by mass}&=\dfrac{\text{mass of solute}}{\text{mass of solution}} \times 100 \label{Eq11} \\[5pt] \text{parts per million (ppm)} &=\dfrac{\text{mass of solute}}{\text{mass of solution}} \times 10^{6} \label{Eq12} \\[5pt] \text{parts per billion (ppb)}&=\dfrac{\text{mass of solute}}{\text{mass of solution}} \times 10^{9} \label{Eq13} \end{align}\]
In the health sciences, the concentration of a solution is often expressed as parts per thousand (ppt), indicated as a proportion. For example, adrenalin, the hormone produced in high-stress situations, is available in a 1:1000 solution, or one gram of adrenalin per 1000 g of solution.
The labels on bottles of commercial reagents often describe the contents in terms of mass percentage. Sulfuric acid, for example, is sold as a 95% aqueous solution, or 95 g of \(\ce{H_2SO_4}\) per 100 g of solution. Parts per million and parts per billion are used to describe concentrations of highly dilute solutions. These measurements correspond to milligrams and micrograms of solute per kilogram of solution, respectively. For dilute aqueous solutions, this is equal to milligrams and micrograms of solute per liter of solution (assuming a density of 1.0 g/mL).
Example \(\PageIndex{7}\): Molarity and Mass
Several years ago, millions of bottles of mineral water were contaminated with benzene at ppm levels. This incident received a great deal of attention because the lethal concentration of benzene in rats is 3.8 ppm. A 250 mL sample of mineral water has 12.7 ppm of benzene. Because the contaminated mineral water is a very dilute aqueous solution, we can assume that its density is approximately 1.00 g/mL.
- What is the molarity of the solution?
- What is the mass of benzene in the sample?
Given: volume of sample, solute concentration, and density of solution
Asked for: molarity of solute and mass of solute in 250 mL
Strategy:
- Use the concentration of the solute in parts per million to calculate the molarity.
- Use the concentration of the solute in parts per million to calculate the mass of the solute in the specified volume of solution.
Solution:
a. A To calculate the molarity of benzene, we need to determine the number of moles of benzene in 1 L of solution. We know that the solution contains 12.7 ppm of benzene. Because 12.7 ppm is equivalent to 12.7 mg/1000 g of solution and the density of the solution is 1.00 g/mL, the solution contains 12.7 mg of benzene per liter (1000 mL). The molarity is therefore
\[\begin{align*} \text{molarity}&=\dfrac{\text{moles}}{\text{liter solution}} \\[5pt] &=\dfrac{(12.7\; \cancel{mg}) \left(\frac{1\; \cancel{g}}{1000\; \cancel{mg}}\right)\left(\frac{1\; mol}{78.114\; \cancel{g}}\right)}{1.00\; L} \\[5pt] &=1.63 \times 10^{-4} M\end{align*}\]
b. B We are given that there are 12.7 mg of benzene per 1000 g of solution, which is equal to 12.7 mg/L of solution. Hence the mass of benzene in 250 mL (250 g) of solution is
\[\begin{align*} \text{mass of benzene} &=\dfrac{(12.7\; mg\; \text{benzene})(250\; \cancel{mL})}{1000\; \cancel{mL}} \\[5pt] &=3.18\; mg \\[5pt] &=3.18 \times 10^{-3}\; g\; \text{benzene} \end{align*}\]
Exercise \(\PageIndex{7}\): Molarity of Lead Solution
The maximum allowable concentration of lead in drinking water is 9.0 ppb.
- What is the molarity of \(\ce{Pb^{2+}}\) in a 9.0 ppb aqueous solution?
- Use your calculated concentration to determine how many grams of \(\ce{Pb^{2+}}\) are in an 8 oz glass of water.
- Answer a
-
4.3 × 10−8 M
- Answer b
-
2 × 10−6 g
Concentrations are also often reported on a mass-to-volume (m/v) basis, particularly in clinical laboratories and engineering applications. A concentration expressed on an m/v basis is the number of grams of solute per milliliter of solution. Each measurement can be expressed as a percentage by multiplying the ratio by 100; the result is reported as percent m/v. Even though reported on a mass-to-volume basis, the concentrations of very dilute solutions are often expressed in parts per million (ppm) or in parts per billion (ppb). For aqueous solutions at 20°C, 1 ppm corresponds to 1 μg per milliliter, and 1 ppb corresponds to 1 ng per milliliter.
How do chemists decide which units of concentration to use for a particular application? Although molarity is commonly used to express concentrations for reactions in solution or for titrations, it does have one drawback—molarity is the number of moles of solute divided by the volume of the solution, and the volume of a solution depends on its density, which is a function of temperature. Because volumetric glassware is calibrated at a particular temperature, typically 20°C, the molarity may differ from the original value by several percent if a solution is prepared or used at a significantly different temperature, such as 40°C or 0°C. For many applications this may not be a problem, but for precise work these errors can become important. In contrast, mole fraction, molality, and mass percentage depend on only the masses of the solute and solvent, which are independent of temperature.
Mole fraction is not very useful for experiments that involve quantitative reactions, but it is convenient for calculating the partial pressure of gases in mixtures, as discussed previously. Mole fractions are also useful for calculating the vapor pressures of certain types of solutions. Molality is particularly useful for determining how properties such as the freezing or boiling point of a solution vary with solute concentration. Because mass percentage and parts per million or billion are simply different ways of expressing the ratio of the mass of a solute to the mass of the solution, they enable us to express the concentration of a substance even when the molecular mass of the substance is unknown. Units of ppb or ppm are also used to express very low concentrations, such as those of residual impurities in foods or of pollutants in environmental studies.
Table \(\PageIndex{1}\) summarizes the different units of concentration and typical applications for each. When the molar mass of the solute and the density of the solution are known, it becomes relatively easy with practice to convert among the units of concentration we have discussed, as illustrated in Example \(\PageIndex{8}\).
| Unit | Definition | Application |
|---|---|---|
| *The molarity of a solution is temperature dependent, but the other units shown in this table are independent of temperature. | ||
| molarity (M) | moles of solute/liter of solution (mol/L) | Used for quantitative reactions in solution and titrations; mass and molecular mass of solute and volume of solution are known. |
| mole fraction (\(\chi\)) | moles of solute/total moles present (mol/mol) | Used for partial pressures of gases and vapor pressures of some solutions; mass and molecular mass of each component are known. |
| molality (m) | moles of solute/kg of solvent (mol/kg) | Used in determining how colligative properties vary with solute concentration; masses and molecular mass of solute are known. |
| percent by mass (%) | [mass of solute (g)/mass of solution (g)] × 100 | Useful when masses are known but molecular masses are unknown. |
| parts per thousand (ppt) | [mass of solute/mass of solution] × 103 (g solute/kg solution) | Used in the health sciences, ratio solutions are typically expressed as a proportion, such as 1:1000. |
| parts per million (ppm) | [mass of solute/mass of solution] × 106 (mg solute/kg solution) | Used for trace quantities; masses are known but molecular masses may be unknown. |
| parts per billion (ppb) | [mass of solute/mass of solution] × 109 (µg solute/kg solution) | Used for trace quantities; masses are known but molecular masses may be unknown. |
Example \(\PageIndex{8}\): Vodka
Vodka is essentially a solution of ethanol in water. Typical vodka is sold as “80 proof,” which means that it contains 40.0% ethanol by volume. The density of pure ethanol is 0.789 g/mL at 20°C. If we assume that the volume of the solution is the sum of the volumes of the components (which is not strictly correct), calculate the following for the ethanol in 80-proof vodka.
- the percent by mass
- the mole fraction
- the molarity
- the molality
Given: volume percent and density
Asked for: percent by mass, mole fraction, molarity, and molality
Strategy:
- Use the density of the solute to calculate the mass of the solute in 100.0 mL of solution. Calculate the mass of water in 100.0 mL of solution.
- Determine the percent by mass of solute by dividing the mass of ethanol by the mass of the solution and multiplying by 100.
- Convert grams of solute and solvent to moles of solute and solvent. Calculate the mole fraction of solute by dividing the moles of solute by the total number of moles of substances present in solution.
- Calculate the molarity of the solution: moles of solute per liter of solution. Determine the molality of the solution by dividing the number of moles of solute by the kilograms of solvent.
Solution:
The key to this problem is to use the density of pure ethanol to determine the mass of ethanol (\(CH_3CH_2OH\)), abbreviated as EtOH, in a given volume of solution. We can then calculate the number of moles of ethanol and the concentration of ethanol in any of the required units. A Because we are given a percentage by volume, we assume that we have 100.0 mL of solution. The volume of ethanol will thus be 40.0% of 100.0 mL, or 40.0 mL of ethanol, and the volume of water will be 60.0% of 100.0 mL, or 60.0 mL of water. The mass of ethanol is obtained from its density:
\[mass\; of\; EtOH=(40.0\; \cancel{mL})\left(\dfrac{0.789\; g}{\cancel{mL}}\right)=31.6\; g\; EtOH\nonumber\]
If we assume the density of water is 1.00 g/mL, the mass of water is 60.0 g. We now have all the information we need to calculate the concentration of ethanol in the solution.
B The percent by mass of ethanol is the ratio of the mass of ethanol to the total mass of the solution, expressed as a percentage:
\[ \begin{align*} \%EtOH &=\left(\dfrac{mass\; of\; EtOH}{mass\; of\; solution}\right)(100) \\[5pt] &=\left(\dfrac{31.6\; \cancel{g}\; EtOH}{31.6\; \cancel{g} \;EtOH +60.0\; \cancel{g} \; H_2O} \right)(100) \\[5pt]&= 34.5\%\end{align*}\]
C The mole fraction of ethanol is the ratio of the number of moles of ethanol to the total number of moles of substances in the solution. Because 40.0 mL of ethanol has a mass of 31.6 g, we can use the molar mass of ethanol (46.07 g/mol) to determine the number of moles of ethanol in 40.0 mL:
\[ \begin{align*} moles\; \ce{CH_3CH_2OH}&=(31.6\; \cancel{g\; \ce{CH_3CH_2OH}}) \left(\dfrac{1\; mol}{46.07\; \cancel{g\; \ce{CH_3CH_2OH}}}\right) \\[5pt] &=0.686 \;mol\; \ce{CH_3CH_2OH} \end{align*}\]
Similarly, the number of moles of water is
The mole fraction of ethanol is thus
\[ \chi_{\ce{CH_3CH_2OH}}=\dfrac{0.686\; \cancel{mol}}{0.686\; \cancel{mol} + 3.33\;\cancel{ mol}}=0.171\nonumber\]
D The molarity of the solution is the number of moles of ethanol per liter of solution. We already know the number of moles of ethanol per 100.0 mL of solution, so the molarity is
The molality of the solution is the number of moles of ethanol per kilogram of solvent. Because we know the number of moles of ethanol in 60.0 g of water, the calculation is again straightforward:
\[ m_{\ce{CH_3CH_2OH}}=\left(\dfrac{0.686\; mol\; EtOH}{60.0\; \cancel{g}\; H_2O } \right) \left(\dfrac{1000\; \cancel{g}}{kg}\right)=\dfrac{11.4\; mol\; EtOH}{kg\; H_2O}=11.4 \;m \nonumber\]
Exercise \(\PageIndex{8}\): Toluene/Benzene Solution
A solution is prepared by mixing 100.0 mL of toluene with 300.0 mL of benzene. The densities of toluene and benzene are 0.867 g/mL and 0.874 g/mL, respectively. Assume that the volume of the solution is the sum of the volumes of the components. Calculate the following for toluene.
- percent by mass
- mole fraction
- molarity
- molality
- Answer a
-
percent by mass toluene = 24.8%
- Answer b
-
\(\chi_{toluene} = 0.219\)
- Answer c
-
2.35 M toluene
- Answer d
-
3.59 m toluene
Summary
The concentration of a substance is the quantity of solute present in a given quantity of solution.
Different units are used to express the concentrations of a solution depending on the application. The concentration of a solution is the quantity of solute in a given quantity of solution. It can be expressed in several ways: molarity, moles of solute per liter of solution; mole fraction, the ratio of the number of moles of solute to the total number of moles of substances present; percent by mass, the ratio of the mass of the solute to the mass of the solution times 100; parts per thousand (ppt), grams of solute per kilogram of solution; parts per million (ppm), milligrams of solute per kilogram of solution; parts per billion (ppb), micrograms of solute per kilogram of solution; and molality (m), the number of moles of solute per kilogram of solvent.
Solution concentrations are typically expressed in terms of molarity and can be prepared by dissolving a known mass of solute in a solvent or diluting a stock solution.
- definition of molarity: \[ molarity = \dfrac{moles\: of\: solute}{liters\: of\: solution} = \dfrac{mmoles\: of\: solute} {milliliters\: of\: solution} \]
- relationship among volume, molarity, and moles: \[ V_L \, x \, M_{mol/L} = \cancel{L} \left( \dfrac{mol}{\cancel{L}} \right) = moles \]
- relationship between volume and concentration of stock and dilute solutions: \[(V_s)(M_s) = moles\: of\: solute = (V_d)(M_d)\]
Contributors
Modified by Joshua Halpern (Howard University)
- Modified by Tom Neils (Grand Rapids Community College)

