Extra Credit 8
- Page ID
- 418941
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)↵
Introduction
Vitamins help your body grow and work the way it should. Vitamins have different jobs to help keep the body working properly. Some vitamins help you resist infections and keep your nerves healthy, while others may help your body get energy from food or help your blood clot properly. Vitamin C, also known as ascorbic acid, is an essential water-soluble vitamin that is necessary for the growth and repair of the human body. Unfortunately, our bodies do not create these vitamins, such as Vitamin C, so we must look to fruits or fortified foods to receive our daily intake. What are fortified foods? Fortified foods are foods containing additional minerals, micronutrients, and vitamins such as vitamin C. Eating fortified foods can help maintain healthy nutrient levels to keep our bones strong, help your digestion, and prevent vitamin deficiency. However vitamin C is easily degraded during storage. Studies have shown that temperature is a factor that affects the stability of ascorbic acid in these foods. At higher temperatures, the rate constant of degradation increases as well, leading to a loss of vitamin C. The objective of this research is to study the rate constant of ascorbic acid degradation at high temperatures.
Rate of Degradation
One basic and commonly known rule of chemistry is that chemical reactions occur more rapidly at higher temperatures. Milk turns sour more readily if stored at room temperature than a refrigerator; cookies bake faster in the oven at higher temperatures. Thermal energy relates direction to motion at the molecular level. As the temperature rises, molecules move faster and collide more frequently, increasing the probability of reactions. Whether or not, it’s common knowledge that higher temperatures speed up reactions, the reason was not clear to early chemists, until Svante Arrhenius (1859-1927).

Figure 1: Swedish chemist, Svante Arrhenius (1859-1927)
Arrhenius developed a function of the temperature and activation energy that models the dependence of the rate constant of a chemical reaction on absolute temperature (K)
Figure 2: Arrhenius equation
- k : Rate Constant
- A: The pre-exponential factor or frequency factor
- Ea: The activation energy is the minimum amount of energy needed to acquire
- R: The gas constant
- T: The absolute temperature at the which the reaction takes place
Let’s break down this formula. A is the frequency factor constant, or pre-exponential factor. This is the rate of collisions. Ea is the activation energy, which is the minimum quantity of energy needed to undergo a reaction. This is divided by the gas constant R multiplied by temperature in Kelvin. This is an exponential decay model, where what is decaying is not the concentration of a reaction as a function of time, but the magnitude of the rate constant as the function of the exponent activation energy (Ea) over the average kinetic energy (RT). This means at higher temperatures and lower activation energies, the rate constant will increase. Bringing the application of chemistry into the real world, the degradation rate is the rate of how quickly a substance/polymer will break down. By using the arrhenius function, we can study the degradation rate of organic polymers such as ascorbic acid. Studies have shown that ascorbic acid degradation has been modeled as a first order rate reaction. A first order is a reaction in which the rate is directly proportional to the concentration of the reactant as a function of time.
Ascorbic Acid 
Ascorbic Acid is a natural water-soluble vitamin (Vitamin C). Ascorbic acid is a potent reducing and antioxidant agent that functions in detoxifying reaction, the formation of collagen in various tissues such as your connective tissue and your epithelial tissue, fighting bacterial infection, and more. Found naturally in fruits and vegetables, L-ascorbic acid, an isomer, cannot be produced or stored by humans.
Ascorbic acid is a monosaccharide and has the chemical formula, C6H8O6 It contains 6 carbon atoms, 8 hydrogen atoms, and 6 oxygen atoms linked by double and single covalent bonds.
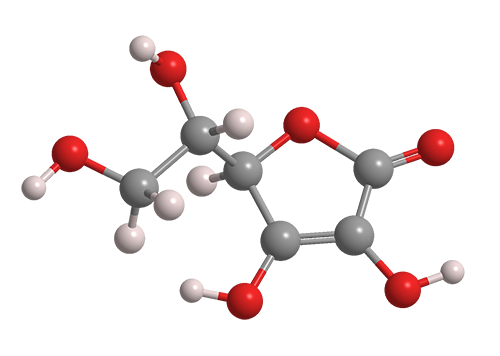
Figure 3: 3D Model of Ascorbic Acid
The degradation of ascorbic acid involves complex oxidation and intermolecular rearrangement reactions, and is considered to be one of the main reasons for quality and color changes during food processing and storage. The oxidation of ascorbic acid leads to the formation of the intermediate dehydroascorbic acid (In figure above) then to the formation of products such as furfural, 2-furoic acid, and 3-hydroxy-2-pyrone. By studying the oxidation of ascorbic acid to dehydroascorbic acid, we can determine the degradation rate constant of Vitamin C.

In a high school experiment studying the degradation rate of Vitamin C in fresh strawberry juice, Sarah determined the activation energy (Ea) to be 4.954 kcal/mol and the pre-exponential factor (A) to be 1.372 M-1s-1. The average temperature of a refrigerator is 5 C. Use this information to estimate the rate constant of vitamin C at room temperature. Estimate the new rate constant at refrigerator temperature.
4.954 kcal/mol * 4.184kJ/1mol
- Answers:
- 1)

