9.4: Oxidation of Fatty Acids
- Page ID
- 234043
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Learning Objectives
- To describe the reactions needed to completely oxidize a fatty acid to carbon dioxide and water.
Like glucose, the fatty acids released in the digestion of triglycerides and other lipids are broken down in a series of sequential reactions accompanied by the gradual release of usable energy. Some of these reactions are oxidative and require nicotinamide adenine dinucleotide (NAD+) and flavin adenine dinucleotide (FAD). The enzymes that participate in fatty acid catabolism are located in the mitochondria, along with the enzymes of the citric acid cycle, the electron transport chain, and oxidative phosphorylation. This localization of enzymes in the mitochondria is of the utmost importance because it facilitates efficient utilization of energy stored in fatty acids and other molecules.
Fatty acid oxidation is initiated on the outer mitochondrial membrane. There the fatty acids, which like carbohydrates are relatively inert, must first be activated by conversion to an energy-rich fatty acid derivative of coenzyme A called fatty acyl-coenzyme A (CoA). The activation is catalyzed by acyl-CoA synthetase. For each molecule of fatty acid activated, one molecule of coenzyme A and one molecule of adenosine triphosphate (ATP) are used, equaling a net utilization of the two high-energy bonds in one ATP molecule (which is therefore converted to adenosine monophosphate [AMP] rather than adenosine diphosphate [ADP]):
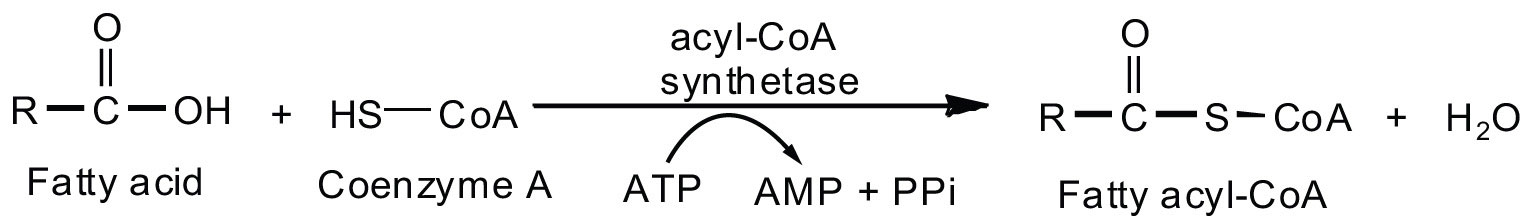
The fatty acyl-CoA diffuses to the inner mitochondrial membrane, where it combines with a carrier molecule known as carnitine in a reaction catalyzed by carnitine acyltransferase. The acyl-carnitine derivative is transported into the mitochondrial matrix and converted back to the fatty acyl-CoA.
Carnitine is an amino acid derivative synthesized from methionine and lysine.
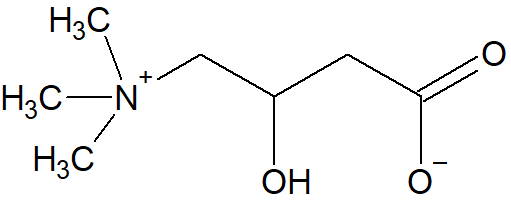
Figure \(\PageIndex{1}\): Chemical structure of the non-proteinogenic amino acid carnitine
Figure \(\PageIndex{2}\): Role of carnitine in the transport of Acyl-CoA from cytosol to the mitochondrial matrix. Image adapted from Original image: Slagtvectorization: Own work, CC0, via Wikimedia Commons
Steps in the β-Oxidation of Fatty Acids
Further oxidation of the fatty acyl-CoA occurs in the mitochondrial matrix via a sequence of four reactions known collectively as β-oxidation because the β-carbon undergoes successive oxidations in the progressive removal of two carbon atoms from the carboxyl end of the fatty acyl-CoA (Figure \(\PageIndex{1}\)).
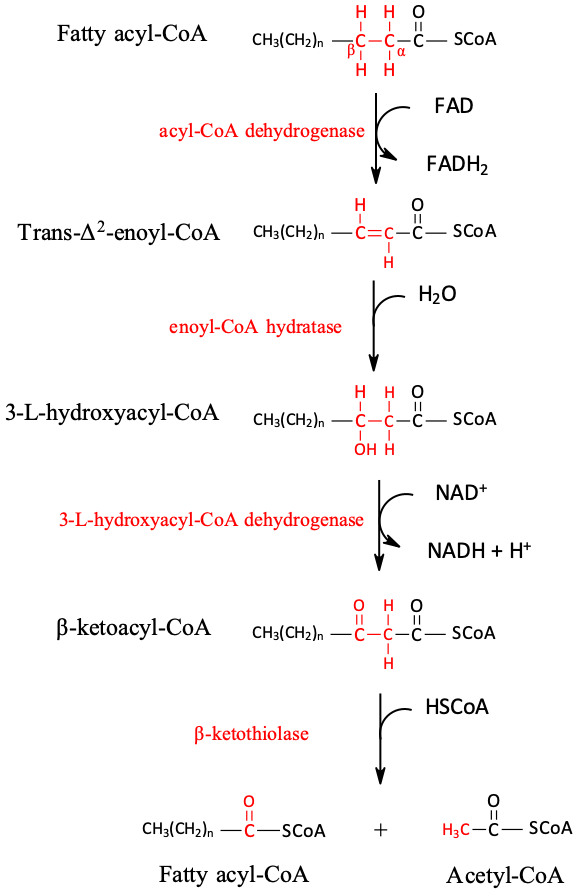
Figure \(\PageIndex{3}\): Fatty Acid Oxidation. The fatty acyl-CoA formed in the final step becomes the substrate for the first step in the next round of β-oxidation. β-oxidation continues until two acetyl-CoA molecules are produced in the final step.
The first step in the catabolism of fatty acids is the formation of an alkene in an oxidation reaction catalyzed by acyl-CoA dehydrogenase. In this reaction, the coenzyme FAD accepts two hydrogen atoms from the acyl-CoA, one from the α-carbon and one from the β-carbon, forming reduced flavin adenine dinucleotide (FADH2).
The FADH2 is reoxidized back to FAD via the electron transport chain that supplies energy to form 1.5–2 molecules of ATP.
Next, the trans-alkene is hydrated to form a secondary alcohol in a reaction catalyzed by enoyl-CoA hydratase. The enzyme forms only the L-isomer.
The secondary alcohol is then oxidized to a ketone by β-hydroxyacyl-CoA dehydrogenase, with NAD+ acting as the oxidizing agent. The reoxidation of each molecule of NADH to NAD+ by the electron transport chain furnishes 2.5–3 molecules of ATP.
The final reaction is cleavage of the β-ketoacyl-CoA by a molecule of coenzyme A. The products are acetyl-CoA and a fatty acyl-CoA that has been shortened by two carbon atoms. The reaction is catalyzed by thiolase.
The shortened fatty acyl-CoA is then degraded by repetitions of these four steps, each time releasing a molecule of acetyl-CoA. The overall equation for the β-oxidation of palmitoyl-CoA (16 carbon atoms) is as follows:

Because each shortened fatty acyl-CoA cycles back to the beginning of the pathway, β-oxidation is sometimes referred to as the fatty acid spiral.
The fate of the acetyl-CoA obtained from fatty acid oxidation depends on the needs of an organism. It may enter the citric acid cycle and be oxidized to produce energy, it may be used for the formation of water-soluble derivatives known as ketone bodies, or it may serve as the starting material for the synthesis of fatty acids.
ATP Yield from Fatty Acid Oxidation. Comparison with Glucose Oxidation.
The amount of ATP obtained from fatty acid oxidation depends on the size of the fatty acid being oxidized. For our purposes here. we’ll study palmitic acid, a saturated fatty acid with 16 carbon atoms, as a typical fatty acid in the human diet. Calculating its energy yield provides a model for determining the ATP yield of all other fatty acids.
The breakdown by an organism of 1 mol of palmitic acid requires 1 mol of ATP (for activation) and forms 8 mol of acetyl-CoA. Recall from the metabolism of carbohydrates that each mole of acetyl-CoA metabolized by the citric acid cycle yields 12 mol of ATP. The complete degradation of 1 mol of palmitic acid requires the β-oxidation reactions to be repeated seven times. Thus, 7 mol of NADH and 7 mol of FADH2 are produced. Reoxidation of these compounds through respiration yields 3 and 2 mol of ATP, respectively. The energy calculations can be summarized as follows:
| 1 mol of ATP is split to AMP and 2Pi | −2 ATP |
| 8 mol of acetyl-CoA formed (8 × 12 ATP) | 96 ATP |
| 7 mol of FADH2 formed (7 × 1.5) | 10.5 ATP |
| 7 mol of NADH formed (7 × 2.5) | 17.5 ATP |
| Total | 122 ATP |
The number of times β-oxidation is repeated for a fatty acid containing n carbon atoms is n/2 – 1 because the final turn yields two acetyl-CoA molecules.
The combustion of 1 mol of palmitic acid releases a considerable amount of energy:
\[C_{16}H_{32}O_2 + 23O_2 → 16CO_2 + 16H_2O + 2,340\; kcal\]
The percentage of this energy that is conserved by the cell in the form of ATP is as follows:
\[\mathrm{\dfrac{energy\: conserved}{total\: energy\: available}\times100=\dfrac{(122\: ATP)(7.4\: kcal/ATP)}{2,340\: kcal}\times100=39\%}\]
In terms of moles of reactant, the efficiency of fatty acid metabolism is comparable to that of carbohydrate metabolism, which we calculated to be 39%. However, in terms of grams, there is a big difference between the energy that can be generated per gram of glucose and per gram of fatty acid. In the carbohydrate metabolism module, we determine that the oxidation of 1 mol of glucose produces 32 ATP moles, that is, 38 x 7.4 kcal /mol ATP = 236.8 kcal. That is the amount of energy produced by 1 mol, or 180 g of glucose. In other words, 1 gram of glucose produces 1.32 kcal of energy (1.32/g glucose). For a fatty acid, such as palmitic acid, we are able to produce 122 ATP molees per mol of palmitic acid, that is, 122 x 7.4 kcal/mol ATP = 902.8 kal. One mole of palmitic acid equals 256 grams. Therefore, the complete oxidation of palmitic acid produces 3.52 kcal/g of palmitic acid, which is more than twice the amount of energy obtained per mole of glucose. The fact that carbons atoms in fatty acids are more reduced than the carbon atoms in glucose explains the difference in the amount of energy produced by their oxidation.
Interesting fact: the oxidation of fatty acids also produces large quantities of water. This water, which sustains migratory birds and animals (such as the camel) for long periods of time.
Ketone Bodies
In the liver, most of the acetyl-CoA obtained from fatty acid oxidation is oxidized by the citric acid cycle. However, under certain metabolic conditions such as starvation or diabetes mellitus, the rate of fatty acid oxidation increases to provide energy. This leads to an increase in the concentration of acetyl-CoA. The increased acetyl-CoA cannot be oxidized by the citric acid cycle because of a decrease in the concentration of oxaloacetate, which is diverted to glucose synthesis. Therefore, some of the acetyl-CoA is used to synthesize a group of compounds known as ketone bodies: acetoacetate, β-hydroxybutyrate, and acetone. Two acetyl-CoA molecules combine, in a reversal of the final step of β-oxidation, to produce acetoacetyl-CoA. The acetoacetyl-CoA reacts with another molecule of acetyl-CoA and water to form β-hydroxy-β-methylglutaryl-CoA, which is then cleaved to acetoacetate and acetyl-CoA. Most of the acetoacetate is reduced to β-hydroxybutyrate, while a small amount is decarboxylated to carbon dioxide and acetone.
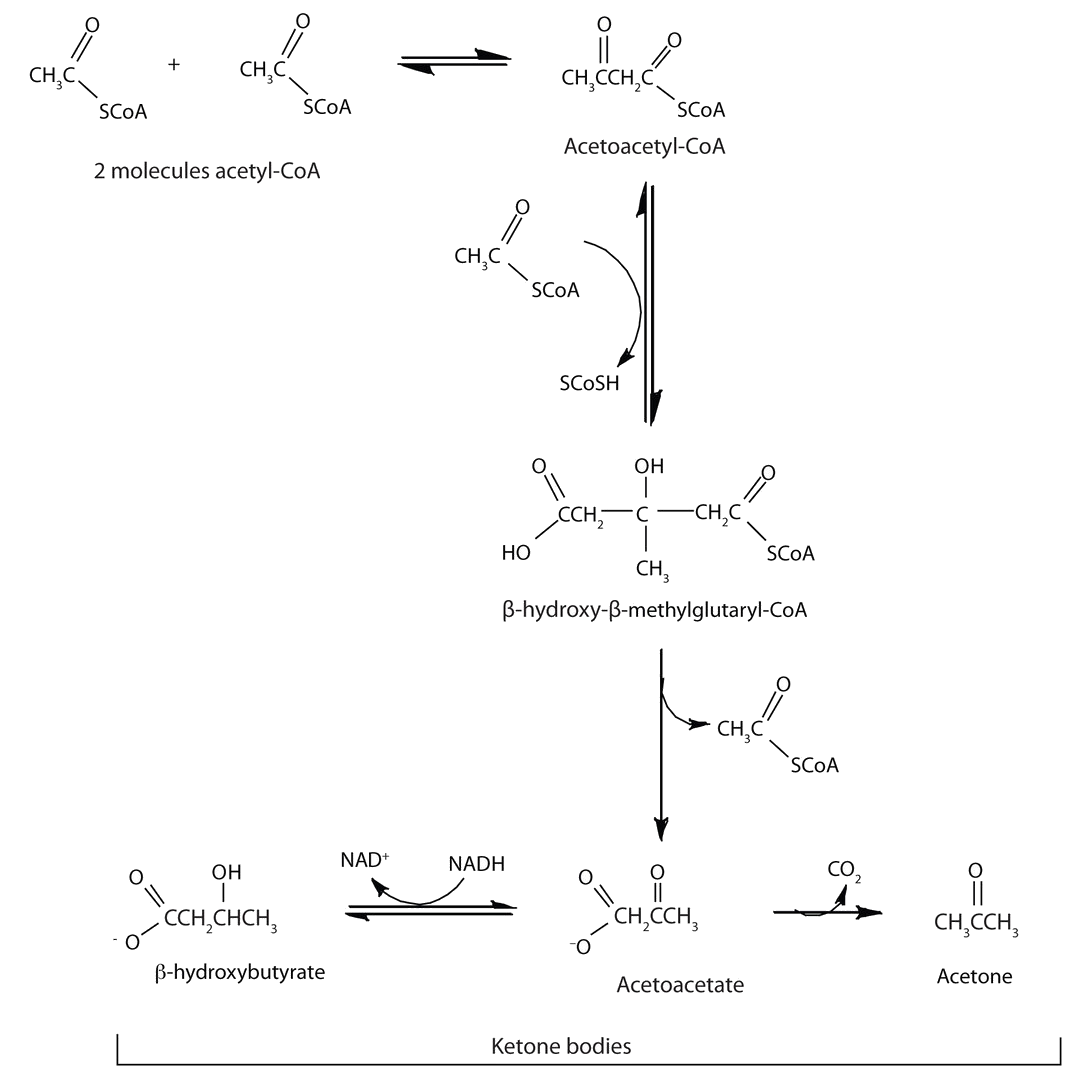
Figure \(\PageIndex{4}\): Formation og ketone bodies
The acetoacetate and β-hydroxybutyrate synthesized by the liver are released into the blood for use as a metabolic fuel (to be converted back to acetyl-CoA) by other tissues, particularly the kidney and the heart. Thus, during prolonged starvation, ketone bodies provide about 70% of the energy requirements of the brain. Under normal conditions, the kidneys excrete about 20 mg of ketone bodies each day, and the blood levels are maintained at about 1 mg of ketone bodies per 100 mL of blood.
In starvation, diabetes mellitus, and certain other physiological conditions in which cells do not receive sufficient amounts of carbohydrate, the rate of ketone body formation in the liver increases further, to a level much higher than can be used by other tissues. The excess ketone bodies accumulate in the blood and the urine, a condition referred to as ketosis. When the acetone in the blood reaches the lungs, its volatility causes it to be expelled in the breath. The sweet smell of acetone, a characteristic of ketosis, is frequently noticed on the breath of severely diabetic patients.
Because two of the three kinds of ketone bodies are weak acids, their presence in the blood in excessive amounts overwhelms the blood buffers and causes a marked decrease in blood pH (to 6.9 from a normal value of 7.4). This decrease in pH leads to a serious condition known as acidosis. One of the effects of acidosis is a decrease in the ability of hemoglobin to transport oxygen in the blood. In moderate to severe acidosis, breathing becomes labored and very painful. The body also loses fluids and becomes dehydrated as the kidneys attempt to get rid of the acids by eliminating large quantities of water. The lowered oxygen supply and dehydration lead to depression; even mild acidosis leads to lethargy, loss of appetite, and a generally run-down feeling. Untreated patients may go into a coma. At that point, prompt treatment is necessary if the person’s life is to be saved.
Summary
- Fatty acids, obtained from the breakdown of triglycerides and other lipids, are oxidized through a series of reactions known as β-oxidation.
- In each round of β-oxidation, 1 molecule of acetyl-CoA, 1 molecule of NADH, and 1 molecule of FADH2 are produced.
- The acetyl-CoA, NADH, and FADH2 are used in the citric acid cycle, the electron transport chain, and oxidative phosphorylation to produce ATP


