6.7: Ethers
- Page ID
- 322681
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Learning Objectives
- Describe the structural difference between an alcohol and an ether that affects physical characteristics and reactivity of each.
- Name simple ethers.
- Describe the structure and uses of some ethers.
Structure and nomenclature of ethers
With the general formula ROR′, an ether may be considered a derivative of water in which both hydrogen atoms are replaced by alkyl or aryl groups. It may also be considered a derivative of an alcohol (ROH) in which the hydrogen atom of the OH group is been replaced by a second alkyl or aryl group:
\[\mathrm{HOH\underset{H\: atoms}{\xrightarrow{replace\: both}}ROR'\underset{of\: OH\: group}{\xleftarrow{replace\: H\: atom}}ROH} \]
Simple ethers have simple common names, formed from the names of the alkyl groups attached to oxygen atom listed alpagbetically, followed by the generic name ether. For example, CH3–O–CH2CH2CH3 is methyl propyl ether. If both groups are the same, the group name should be preceded by the prefix di-, as in dimethyl ether (CH3–O–CH3) and diethyl ether CH3CH2–O–CH2CH3.
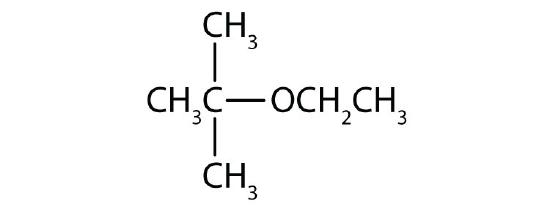
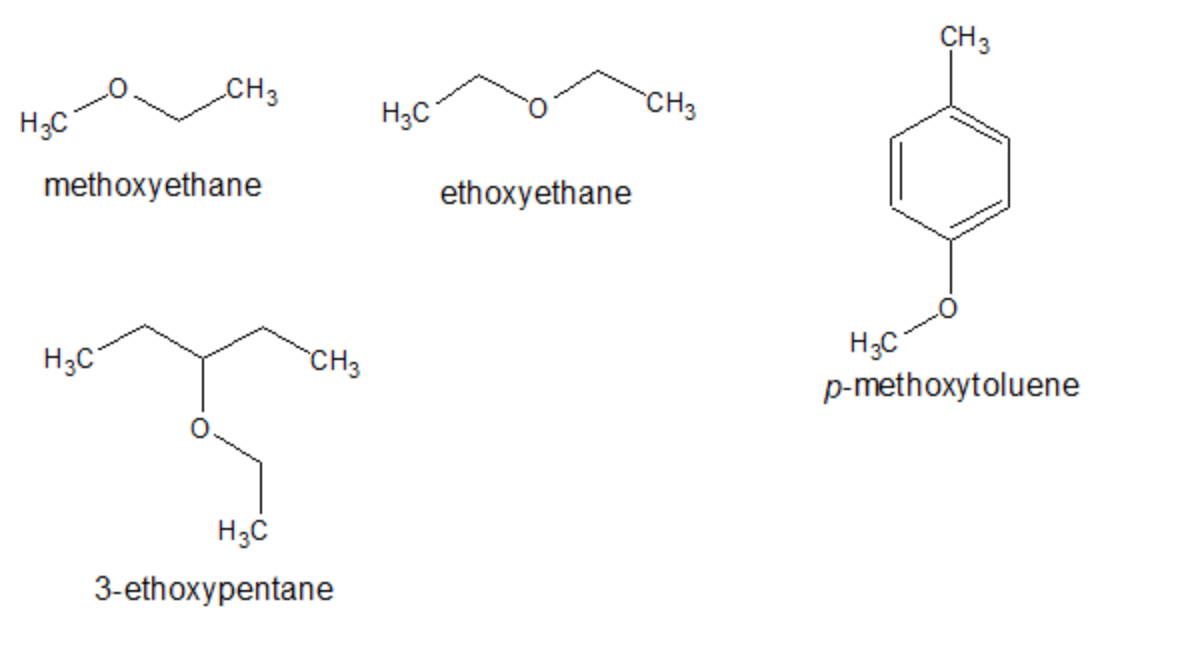
According to IUPAC rules, ethers are named as substituted alkanes bearing an -O-R (alkoxy) substituent. The name of the -O-R groups (which always contains the smaller -R) is obtained by replacing -yl ending of the R alkyl group by -oxy. For example, -OCH3 is named as a methoxy substituent. When necessary, the position of the -O-R group is indicated by numbers. Examples:
Physical properties
Ether molecules have no hydrogen atom on the oxygen atom (that is, no OH group). Therefore there is no intermolecular hydrogen bonding between ether molecules, and ethers therefore have quite low boiling points for a given molar mass. Indeed, ethers have boiling points about the same as those of alkanes of comparable molar mass and much lower than those of the corresponding alcohols (Table \(\PageIndex{1}\)). Therefore, ethers are quite flammable.
| Condensed Structural Formula | Name | Molar Mass | Boiling Point (°C) | Intermolecular Hydrogen Bonding in Pure Liquid? |
|---|---|---|---|---|
| CH3CH2CH3 | propane | 44 | –42 | no |
| CH3OCH3 | dimethyl ether | 46 | –25 | no |
| CH3CH2OH | ethyl alcohol | 46 | 78 | yes |
| CH3CH2CH2CH2CH3 | pentane | 72 | 36 | no |
| CH3CH2OCH2CH3 | diethyl ether | 74 | 35 | no |
| CH3CH2CH2CH2OH | butyl alcohol | 74 | 117 | yes |
Ether molecules do have an oxygen atom, however, and engage in hydrogen bonding with water molecules. Consequently, an ether has about the same solubility in water as the alcohol that is isomeric with it. For example, dimethyl ether and ethanol (both having the molecular formula C2H6O) are completely soluble in water, whereas diethyl ether and 1-butanol (both C4H10O) are barely soluble in water (8 g/100 mL of water). The solubility rapidly decreases with the number of atoms, and only ether up to three carbon atoms are soluble in water, that is, dimethyl ether and ethyl methyl ether
Example \(\PageIndex{1}\)
What is the common name and IUPAC for each ether?
- CH3CH2CH2OCH2CH2CH3
-
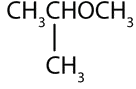
Solution
- The carbon groups on either side of the oxygen atom are propyl (CH3CH2CH2) groups, so the compound is dipropyl ether (common) or 1-propoxypropane (IUPAC)
- The three-carbon group is attached by the middle carbon atom, so it is an isopropyl group. The one-carbon group is a methyl group. The compound is isopropyl methyl ether (common) or 2-methoxypropane (IUPAC)
Exercise \(\PageIndex{1}\)
What is the common name for each ether?
-
CH3CH2CH2CH2OCH2CH2CH2CH3
-
Chemical and biological properties of ethers
Besides being flammable, ethers are very stable and non-reactive, similarly to alkanes. For this reason, they are mainly used as solvents in the industry.
Ethers have anesthetic properties. A general anesthetic acts on the brain to produce unconsciousness and a general insensitivity to feeling or pain. Diethyl ether (CH3CH2OCH2CH3) was the first general anesthetic to be used.
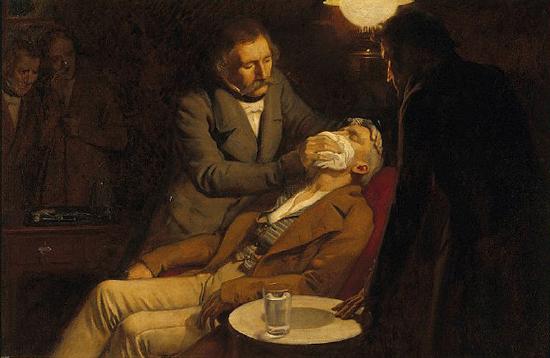
Diethyl ether is relatively safe because there is a fairly wide gap between the dose that produces an effective level of anesthesia and the lethal dose. However, because it is highly flammable and has the added disadvantage of causing nausea, it has been replaced by newer inhalant anesthetics, including the fluorine-containing compounds halothane, enflurane, and isoflurane. Unfortunately, the safety of these compounds for operating room personnel has been questioned. For example, female operating room workers exposed to halothane suffer a higher rate of miscarriages than women in the general population.
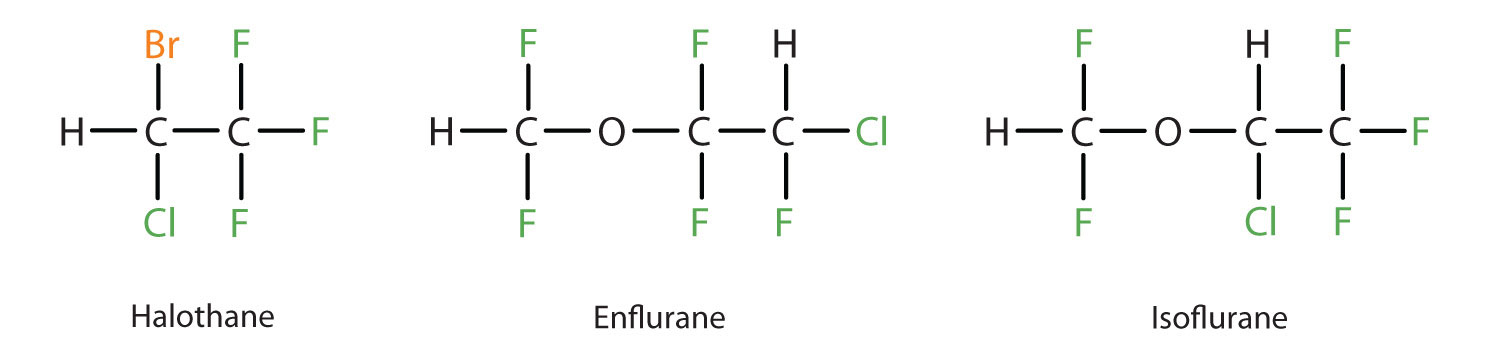
These three modern, inhalant, halogen-containing, anesthetic compounds are less flammable than diethyl ether.
Summary
To give ethers common names, simply name the groups attached to the oxygen atom, followed by the generic name ether. If both groups are the same, the group name should be preceded by the prefix di-. In the IUPAC system, ethers are named as substituted alkanes bearing an alkoxy (-O-R) substituent. Ether molecules have no OH group and thus no intermolecular hydrogen bonding. Ethers therefore have quite low boiling points for a given molar mass. Ether molecules have an oxygen atom and can engage in hydrogen bonding with water molecules. An ether molecule has about the same solubility in water as the alcohol that is isomeric with it.
Concept Review Exercises
-
Why does diethyl ether (CH3CH2OCH2CH3) have a much lower boiling point than 1-butanol (CH3CH2CH2CH2OH)?
-
Which is more soluble in water—ethyl methyl ether (CH3CH2OCH3) or 1-butanol (CH3CH2CH2CH2OH)? Explain.
Answers
-
Diethyl ether has no intermolecular hydrogen bonding because there is no OH group; 1-butanol has an OH and engages in intermolecular hydrogen bonding.
-
Ethyl methyl ether (three carbon atoms, one oxygen atom) is more soluble in water than 1-butanol (four carbon atoms, one oxygen atom), even though both can engage in hydrogen bonding with water.
Exercises
-
How can ethanol give two different products when heated with sulfuric acid? Name these products.
-
Which of these ethers is isomeric with ethanol—CH3CH2OCH2CH3, CH3OCH2CH3, or CH3OCH3?
-
Name each compound.
- CH3OCH2CH2CH3
-
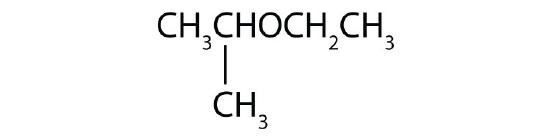
-
Name each compound.
- CH3CH2CH2CH2OCH3
- CH3CH2OCH2CH2CH3
-
Draw the structure for each compound.
- methyl ethyl ether
- tert-butyl ethyl ether
-
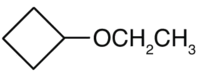
Draw the structure for each compound.
- diisopropyl ether
- cyclopropyl propyl ether
Answers
-
Intramolecular (both the H and the OH come from the same molecule) dehydration gives ethylene; intermolecular (the H comes from one molecule and the OH comes from another molecule) dehydration gives diethyl ether.
-
- methyl propyl ether
- ethyl isopropyl ether
-
- CH3OCH2CH3
-