4.7: Characteristics of Ionic Compounds
- Page ID
- 221351
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Learning Objectives
- To describe the physical properties of ionic compounds.
The figure below shows just a few examples of the color and brilliance of naturally occurring ionic crystals. The regular and orderly arrangement of ions in the crystal lattice is responsible for the various shapes of these crystals, while transition metal ions give rise to the colors.

Physical Properties of Ionic Compounds
Melting Points
Because of the many simultaneous attractions between cations and anions that occur, ionic crystal lattices are very strong. The process of melting an ionic compound requires the addition of large amounts of energy in order to break all of the ionic bonds in the crystal. For example, sodium chloride has a melting temperature of about 800oC. As a comparison, the molecular compound water melts at 0 °C.
Shattering
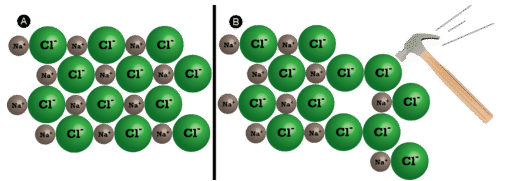
Ionic compounds are generally hard, but brittle. Why? It takes a large amount of mechanical force, such as striking a crystal with a hammer, to force one layer of ions to shift relative to its neighbor. However, when that happens, it brings ions of the same charge next to each other (see below). The repulsive forces between like-charged ions cause the crystal to shatter. When an ionic crystal breaks, it tends to do so along smooth planes because of the regular arrangement of the ions.
Solubility in water
A considerable high number of ionic compounds are soluble in water. Table salt, or sodium chloride (NaCl), the most common ionic compound, is soluble in water (360 g/L). Recall that NaCl is a salt crystal composed not of discrete NaCl molecules, but rather of an extended array of Na+ and Cl- ions bound together in three dimensions through electrostatic interactions. When a crystal of NaCl comes into contact with water, the water molecules interact with the Na+ and Cl- ions on the crystal’s surface, as shown in the figure \(\PageIndex{3}\). As a consequence of the interaction between the Na+ and Cl- ions and the water molecules, the electrostatic interactions within the crystal are broken.
Figure \(\PageIndex{3}\): Dosilution of sodium chloride in water. Image by Ahazard.sciencewriter, CC BY-SA 4.0, via Wikimedia Commons
Conductivity
Another characteristic property of ionic compounds is their electrical conductivity. The figure below shows three experiments in which two electrodes that are connected to a light bulb are placed in beakers containing three different substances.
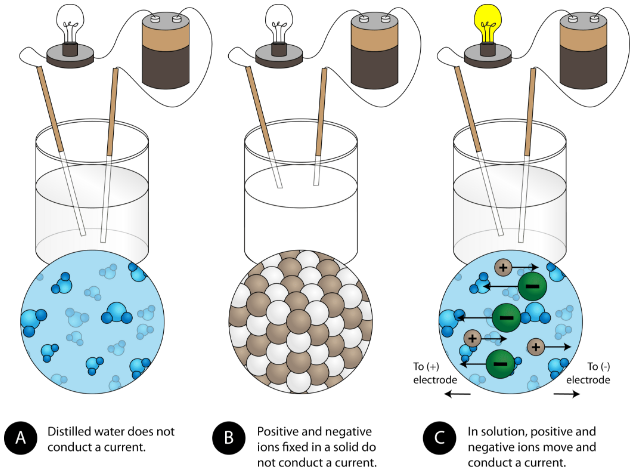
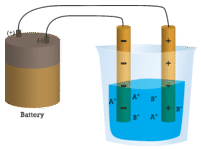
In the first beaker, distilled water does not conduct a current because water is a molecular compound. In the second beaker, solid sodium chloride also does not conduct a current. Despite being ionic and thus composed of charged particles, the solid crystal lattice does not allow the ions to move between the electrodes. Mobile charged particles are required for the circuit to be complete and the light bulb to light up. In the third beaker, the NaCl has been dissolved into the distilled water. Now the crystal lattice has been broken apart and the individual positive and negative ions can move. Cations move to one electrode, while anions move to the other, allowing electricity to flow (see figure below). Melting an ionic compound also frees the ions to conduct a current. Ionic compounds conduct an electric current when melted or dissolved in water.
Example \(\PageIndex{1}\)
Write the dissociation equation of solid NaCl in water.
Solution
NaCl(s) → Na+(aq) + Cl–(aq)
Exercise \(\PageIndex{1}\)
Write the dissociation equation of solid NH4NO3 in water.
- Answer
-
NH4NO3(s) → NH4+(aq) + NO3–(aq)
Key Takeaways
- Ionic compounds have high melting points.
- Ionic compounds are hard and brittle.
- Ionic compounds dissociate into ions when dissolved in water.
- Solutions of ionic compounds and melted ionic compounds conduct electricity, but solid materials do not.
- An ionic compound can be identified by its chemical formula: metal + nonmetal or polyatomic ions.
Contributors and Attributions
- CK-12 Foundation by Sharon Bewick, Richard Parsons, Therese Forsythe, Shonna Robinson, and Jean Dupon.
Exercises
1. Explain how the bonding in an ionic solid explains some of the properties of these solids.
2. Which type(s) of solid conduct(s) electricity in their liquid state but not in their solid state?
3. Based on chemical formula, identify which of the following is an ionic solid?
a. Hg
b. PH3
c. Ba(NO3)2
4. Based on chemical formula, identify which of the following will conduct electric current in the melted state but not in the solid state?
a. H2S
b. Si
c. KBr
5. Based on chemical formula, identify which of the following will dissociate into ions when dissolved in water?
a. Co
b. CO
c. CaCl2
6. Write dissociation equations for the following ionic compounds.
- LiF(s)
- CuSO4(s)
- Na2S(s)
- MgBr2(s)
Answers
1. The oppositely charged ions are very strongly held together, so ionic crystals have high melting points. Ionic crystals are also brittle because any distortion of the crystal moves same-charged ions closer to each other, so they repel. When melted and dissolved in solution, the ions become free to conduct electric current.
2. Ionic compounds
3. Ba(NO3)2
4. KBr
5. CaCl2
6.
- LiF(s) → Li+(aq) + F–(aq)
- CuSO4(s) → Cu2+(aq) + SO42–(aq)
- Na2S(s) → 2Na+(aq) + S2–(aq)
- MgBr2(s) → Mg2+(aq) + 2Br–(aq)
Citations
Solubility of Ionic Compounds: Salts. (2019, June 5). Retrieved May 18, 2021, from https://chem.libretexts.org/@go/page/52399
Characteristics of Ionic Compounds. (2020, August 10). Retrieved May 18, 2021, from https://chem.libretexts.org/@go/page/207049


