Ionization Constants of Weak Acids
- Page ID
- 221297
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
| Ionization Constants of Weak Acids | |||
|---|---|---|---|
| Acid | Formula | Ka at 25 °C | Lewis Structure |
| acetic | CH3CO2H | 1.8 × 10−5 | 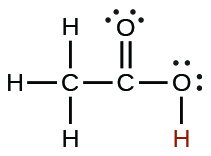 |
| arsenic | H3AsO4 | 5.5 × 10−3 | 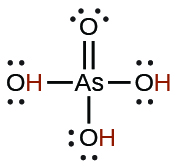 |
 | 1.7 × 10−7 | ||
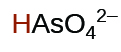 | 5.1 × 10−12 | ||
| arsenous | H3AsO3 | 5.1 × 10−10 | 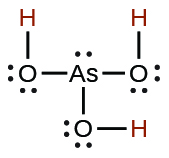 |
| boric | H3BO3 | 5.4 × 10−10 | 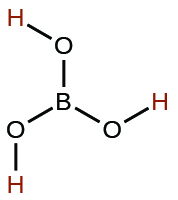 |
| carbonic | H2CO3 | 4.3 × 10−7 | 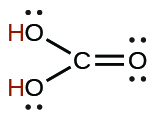 |
 | 5.6 × 10−11 | ||
| cyanic | HCNO | 2 × 10−4 | 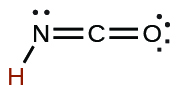 |
| formic | HCO2H | 1.8 × 10−4 | 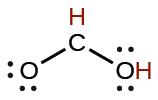 |
| hydrazoic | HN3 | 2.5 × 10−5 |  |
| hydrocyanic | HCN | 4.9 × 10−10 | |
| hydrofluoric | HF | 3.5 × 10−4 | |
| hydrogen peroxide | H2O2 | 2.4 × 10−12 | 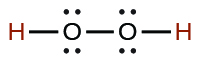 |
| hydrogen selenide | H2Se | 1.29 × 10−4 | |
| HSe– | 1 × 10−12 | ||
| hydrogen sulfate ion |  | 1.2 × 10−2 | 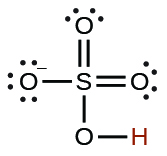 |
| hydrogen sulfide | H2S | 8.9 × 10−8 | |
| HS– | 1.0 × 10−19 | ||
| hydrogen telluride | H2Te | 2.3 × 10−3 | |
| HTe– | 1.6 × 10−11 | ||
| hypobromous | HBrO | 2.8 × 10−9 | |
| hypochlorous | HClO | 2.9 × 10−8 | |
| nitrous | HNO2 | 4.6 × 10−4 | 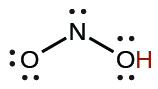 |
| oxalic | H2C2O4 | 6.0 × 10−2 | 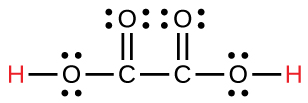 |
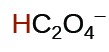 | 6.1 × 10−5 | ||
| phosphoric | H3PO4 | 7.5 × 10−3 | 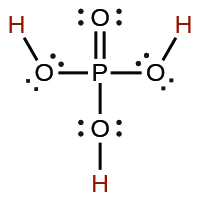 |
 | 6.2 × 10−8 | ||
 | 4.2 × 10−13 | ||
| phosphorous | H3PO3 | 5 × 10−2 | 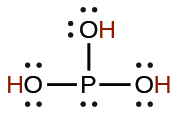 |
 | 2.0 × 10−7 | ||
| sulfurous | H2SO3 | 1.6 × 10−2 | 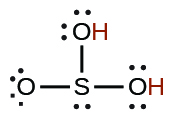 |
 | 6.4 × 10−8 | ||


