21.1 Naming Carboxylic Acid Derivatives
- Page ID
- 90992
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Objectives
After completing this section, you should be able to
- provide a satisfactory name for an acid halide of given structure.
- draw the condensed or shorthand structure of an acid halide, given either its commonly used or systematic name.
- provide a satisfactory name for an acid anhydride of given structure.
- draw the condensed or shorthand structure of an acid anhydride, given either its trivial or IUPAC name.
- provide an acceptable name for an amide of given structure.
- draw the condensed or shorthand structure of an amide, given its trivial or IUPAC name.
- provide an acceptable name for an ester of given structure.
- draw the condensed or shorthand structure of an ester, given either its trivial or IUPAC name.
- provide an acceptable name for a thioester of given structure.
- draw the condensed or shorthand structure of a thioester, given its trivial or IUPAC name.
- provide an acceptable name for an acyl phosphate.
- draw the condensed or shorthand structure of an acyl phosphate, given either its trivial or IUPAC name.
Nomenclature of acid halides
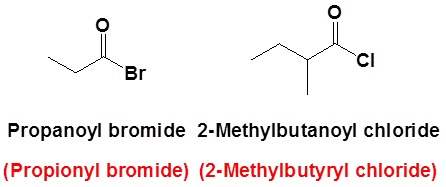
The nomenclature of acid halides starts with the name of the corresponding carboxylic acid. The –ic acid ending is removed and replaced with the ending -yl followed by the name of the halogen with an –ide ending. This is true for both common and IUPAC nomenclature. The carbonyl carbon is given the #1 location number. It is not necessary to include the location number in the name because it is assumed that the functional group will be on the end of the parent chain.
The acid anhydride functional group results when two carboxylic acids combine and lose water (anhydride = without water). Symmetrical acid anhydrides are named like carboxylic acids except the ending -acid is replaced with -anhydride. This is true for both the IUPAC and Common nomenclature.
Nomenclature of Anhydrides
Symmetrical anhydrides
A symmetrical anhydride is a carboxylic acid anhydride that has the following general structural formula.
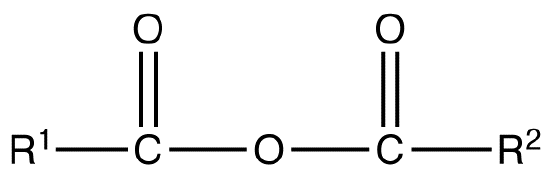
where R1=R2= hydrogen atoms, alkyl groups, aryl groups
Unsymmetrical Anhydrides
A mixed or unsymmetrical anhydride is a carboxylic acid anhydride that has the following general structural formula.
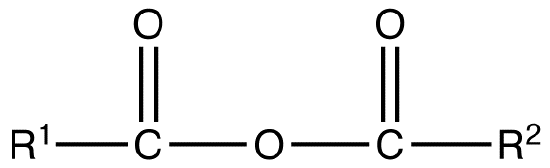
where R1≠R2, but are hydrogen atoms, alkyl groups, aryl groups. When naming unsymmetrical acid anhydrides, name both using alkanoic general method and then put the two names alphabetically. Hence, first name each component and alphabetically arranged them followed by spaces and then the word anhydride.
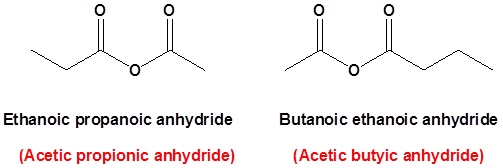
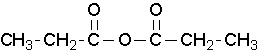
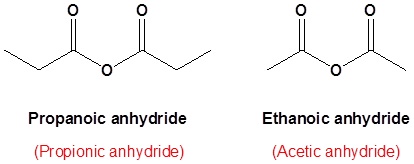
propanoic anhydride
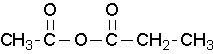
ethanoic propanoic anhydride
Common names that you should know
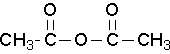
acetic anhydride (Try to name this anhydride by the proper name. J )
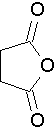
succinic anhydride (Try to name this anhydride by the proper name. J )
Nomenclature of Esters
Esters are made from a carboxylic acid and an alcohol.
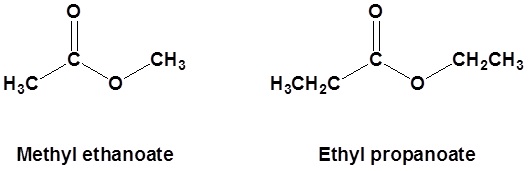
Esters are named as if the alkyl chain from the alcohol is a substituent. No number is assigned to this alkyl chain. This is followed by the name of the parent chain form the carboxylic acid part of the ester with an –e remove and replaced with the ending –oate.
Nomenclature of Thioesters
Thiosters are made from a carboxylic acid and an thiol.
Thioesters are named as if the alkyl chain from the alcohol is a substituent. No number is assigned to this alkyl chain. This is followed by the name of the parent chain from the carboxylic acid part of the thioester named as an alkane with the ending –thiooate.
Nomenclature of amides
Primary amides
Primary amides are named by changing the name of the acid by dropping the -oic acid or -ic acid endings and adding -amide. The carbonyl carbon is given the #1 location number. It is not necessary to include the location number in the name because it is assumed that the functional group will be on the end of the parent chain.
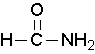
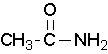
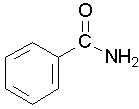
methanamide or formamide (left), ethanamide or acetamide (center) , benzamide (right)
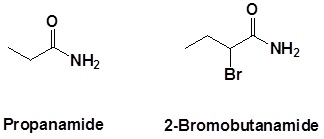
Secondary amides
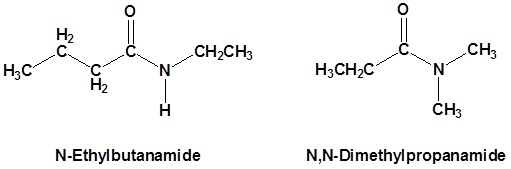
Secondary amides are named by using an upper case N to designate that the alkyl group is on the nitrogen atom. Alkyl groups attached to the nitrogen are named as substituents. The letter N is used to indicate they are attached to the nitrogen. Tertiary amides are named in the same way.
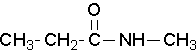
N-methylpropanamide
Name the parent alkane (include the carbon atom of the nitrile as part of the parent) followed with the word -nitrile. The carbon in the nitrile is given the #1 location position. It is not necessary to include the location number in the name because it is assumed that the functional group will be on the end of the parent chain.
Naming Phosphates
Phosphoryl groups are derivatives of phosphoric acid, a strong acid that is commonly used in the laboratory. The fully deprotonated conjugate base of phosphoric acid is called a phosphate ion, or inorganic phosphate (often abbreviated 'Pi'). When two phosphate groups are linked to each other, the linkage is referred to as a 'phosphate anhydride', and the ion is called 'inorganic pyrophosphate' (abbreviation PPi).
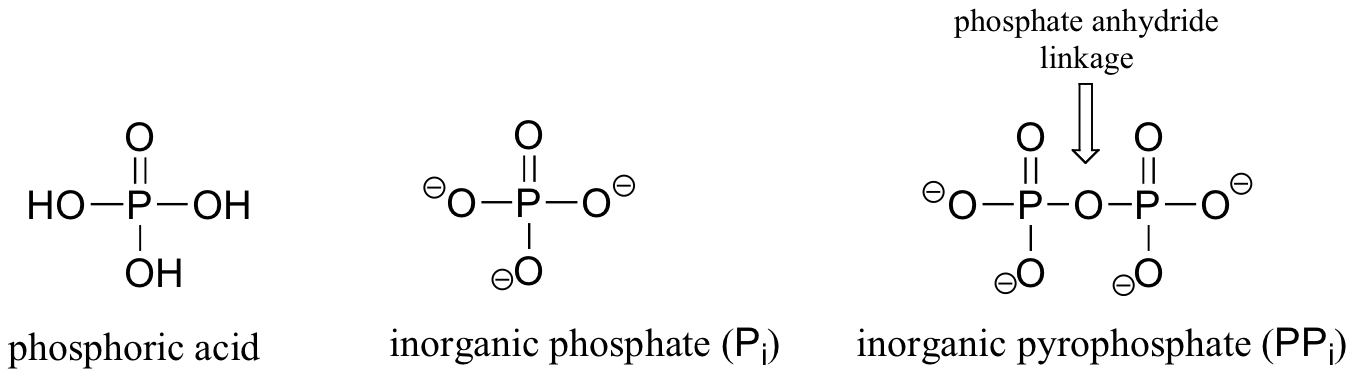
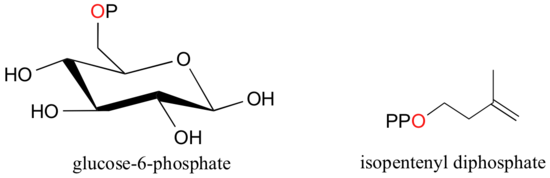
When a phosphate ion is attached to a carbon atom on an organic molecule, the chemical linkage is referred to as a phosphate ester, and the whole species is called an organic monophosphate. Glucose-6-phosphate is an example.

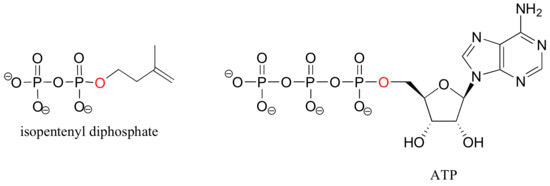
If an organic molecule is linked to two or three phosphate groups, the resulting species are called organic diphosphates and organic triphosphates.

Isopententyl diphosphate and adenosine triphosphate (ATP) are good examples:
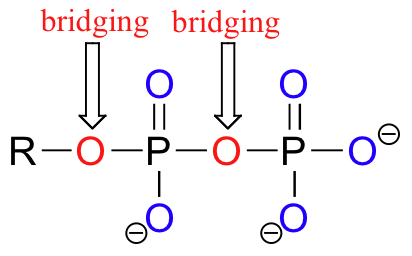
Oxygen atoms in phosphate groups are referred to either 'bridging' and 'non-bridging', depending on their position. An organic diphosphate has two bridging and five non-bridging oxygens.
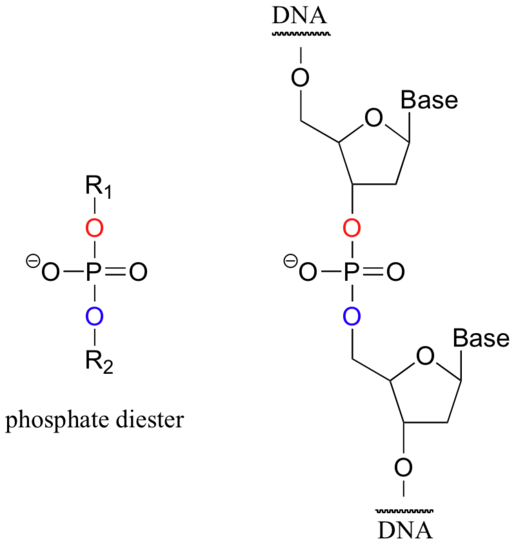
When a single phosphate is linked to two organic groups, the term 'phosphate diester' is used. The backbone of DNA is composed of phosphate diesters.
The term 'phosphoryl group' is a general way to refer to all of the phosphate-based groups mentioned in the paragraphs above.
Recall (section 1.4A) that phosphate groups on organic structures are sometimes abbreviated simply as 'P', a convention that we will use throughout this text. For example, glucose-6-phosphate and isopentenyl diphosphate are often depicted as shown below. Notice that the 'P' abbreviation includes the oxygen atoms and negative charges associated with the phosphate groups.
Exercises
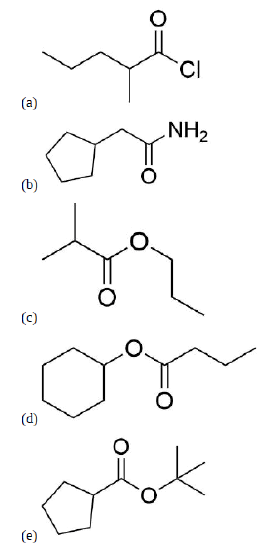
Name the following compounds using IUPAC conventions.
- Answer
-
a) 2-methylpentanoyl chloride
b) 2-
cyclopentylacetamide c) propyl 2-methylpropanoat
d) cyclohexylbutanoate
e) sec-butyl cyclopentanecarboxylate
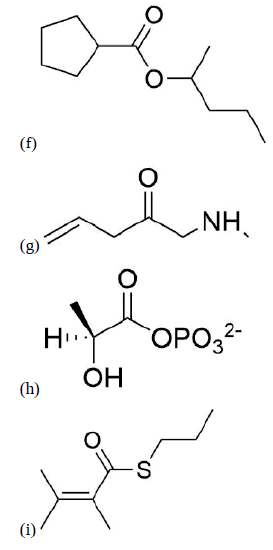
Name the following compounds using IUPAC conventions.
- Answer
-
f) 1-methylbutylcyclopentane carboxylate
g) N-methyl-3-butenamide
h) (S)-2-hydroxypropanoyl phosphate
i) propyl 2,3-dimethyl-2-butenethioate
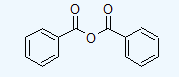
Draw structures corresponding to the following names:
- benzoic anhydride
- phenyl hexanoate
- methyl benzoate
- 3-chloro-N-ethylbenzamide
- pentanamide
- Answer
-
a.
b.
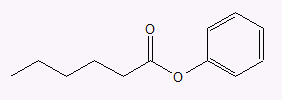
c.

d.
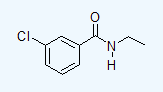
e.
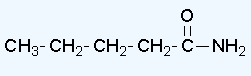
Draw structures corresponding to the following names:
- N-phenylethanamide
- Ethyl 3-oxopentanoate
- Methyl p-bromobenzenethioate
- Formic propanoic anhydride
- cis-2-Methylcyclopentanecarbonyl bromide
- Answer
-
a.

b.

c.
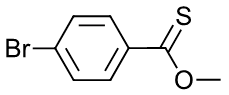
d.
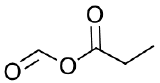
e.
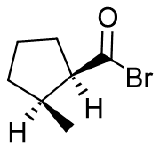
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)
Organic Chemistry With a Biological Emphasis by Tim Soderberg (University of Minnesota, Morris)

