19.2 Preparing Aldehydes and Ketones
- Page ID
- 90965
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Objectives
After completing this section, you should be able to
- describe in detail the methods for preparing aldehydes discussed in earlier units (i.e., the oxidation of primary alcohols and the cleavage of alkenes).
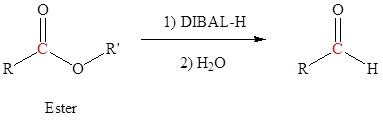
- write an equation to describe the reduction of an ester to an aldehyde.
- identify the product formed when a given ester is reduced with diisobutylaluminum hydride.
- identify the reagents and conditions used in the reduction of an ester to an aldehyde.
- identify the disadvantages of using diisobutylaluminum hydride to reduce an ester to an aldehyde.
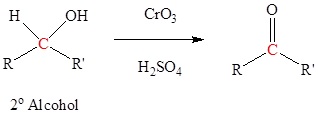
- describe in detail the methods for preparing ketones discussed in earlier units (i.e., the oxidation of secondary alcohols, the ozonolysis of alkenes, Friedel‑Crafts acylation, and the hydration of terminal alkynes).
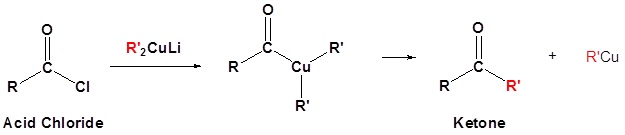
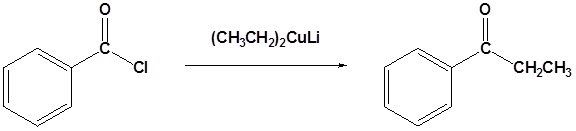
- write an equation to illustrate the formation of a ketone through the reaction of an acid chloride with a dialkylcopper lithium reagent.
- identify the ketone produced from the reaction of a given acid chloride with a specified dialkylcopper lithium reagent.
- identify the acid chloride, the dialkylcopper lithium reagent, or both, needed to prepare a specific ketone.
You may wish to review the sections in which we discuss the oxidation of alcohols (17.7) and the cleavage of alkenes (8.8). A third method of preparing aldehydes is to reduce a carboxylic acid derivative; for example, to reduce an ester with diisobutylaluminum hydride (DIBAL‑H).
There are essentially five methods of preparing ketones in the laboratory. Four of them have been discussed in earlier sections:
- the oxidation of a secondary alcohol—Section 17.7.
- the ozonolysis of an alkene—Section 8.8.
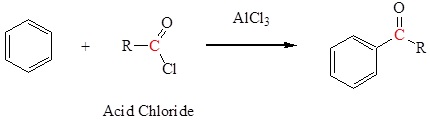
- Friedel‑Crafts acylation—Section 16.3.
- the hydration of a terminal alkyne—Section 9.4.
The “new” method we introduce in this section involves the reaction of an acid chloride with a diorganocopper reagent. The latter substances were discussed in Section 10.7, which you might now wish to review.
Aldehydes and ketones can be prepared using a wide variety of reactions. Although these reactions are discussed in greater detail in other sections, they are listed here as a summary and to help with planning multistep synthetic pathways. Please use the appropriate links to see more details about the reactions.
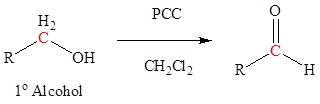
Oxidation of 1o alcohols with PCC to form aldehydes
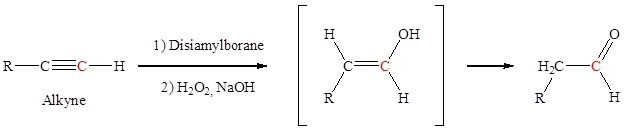
Hydration of an alkyne to form aldehydes
Anti-Markovnikov addition of a hydroxyl group to an alkyne forms an aldehyde. The addition of a hydroxyl group to an alkyne causes tautomerization which subsequently forms a carbonyl.
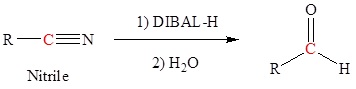
Reduction of an ester, acid chloride or nitrile to form aldehydes
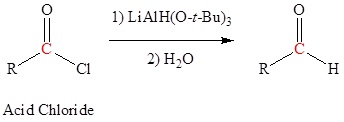
Oxidation of 2o alcohols to form ketones
Typically uses Jones reagent (CrO3 in H2SO4) but many other reagents can be used
Hydration of an alkyne to form ketones
The addition of a hydroxyl group to an alkyne causes tautomerization which subsequently forms a carbonyl. Markovnikov addition of a hydroxyl group to an alkyne forms a ketone.
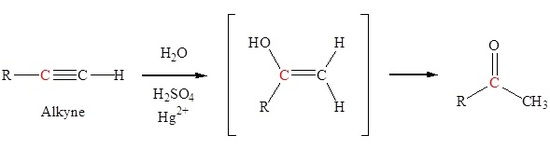
Friedel-Crafts acylation to form a ketone
Reaction of Grignard reagents with nitriles to form ketones
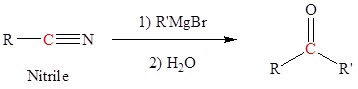
Alkenes can be cleaved using ozone (O3) to form aldehydes and/or ketones
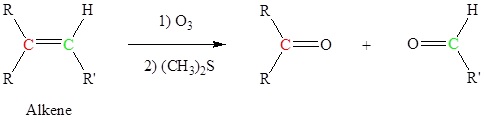
This is an example of a Ozonolysis reaction.
Organocuprate reagents convert acid chlorides to ketones
Exercise
What reagents would be required to prepare hexanal from the following starting materials?
(a) CH3CH2CH2CH2CH2CH2OH
(b) CH3CH2CH2CH2CH2CH=CH2
(c) CH3CH2CH2CH2CO2CH3
(d) CH3CH2CH2CH=CH2
- Answer
-
(a) Dess–Martin periodinane or PCC in CH2Cl2
(b) 1. O3, 2. Zn/H3O+
(c) 1. DIBAH 2. H2O
(d) 1. BH3 2. H2O2/NaOH; 2. Dess–Martin periodinane or PCC
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)