3.8: Binary Molecules - Formulae and Names
- Page ID
- 430558
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Identify covalent and ionic compounds.
- Determine the chemical formula of a simple covalent compound from its name.
- Determine the name of a simple covalent compound from its chemical formula.
COVALENT AND IONIC COMPOUNDS
What elements make covalent bonds? Covalent bonds form when two or more nonmetals combine. For example, both hydrogen and oxygen are nonmetals, and when they combine to make water, they do so by forming covalent bonds. Compounds that are composed of only non-metals or semi-metals with non-metals will display covalent bonding and will be classified as molecular compounds.
As a general rule of thumb, compounds that involve a metal binding with either a non-metal or a semi-metal will display ionic bonding. Thus, the compound formed from sodium and chlorine will be ionic (a metal and a non-metal). Nitrogen monoxide (NO) will be a covalently bound molecule (two non-metals), silicon dioxide (SiO2) will be a covalently bound molecule (a semi-metal and a non-metal) and MgCl2 will be ionic (a metal and a non-metal).
A polyatomic ion is an ion composed of two or more atoms that have a charge as a group (poly = many). The ammonium ion (see figure below) consists of one nitrogen atom and four hydrogen atoms. Together, they comprise a single ion with a 1+ charge and a formula of NH4+. The carbonate ion (see figure below) consists of one carbon atom and three oxygen atoms and carries an overall charge of 2−. The formula of the carbonate ion is CO32−.
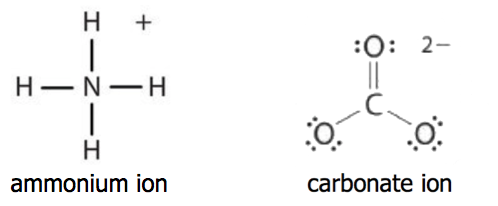
The atoms of a polyatomic ion are tightly bonded together and so the entire ion behaves as a single unit. Nonmetal atoms in polyatomic ions are joined by covalent bonds, but the ion as a whole participates in ionic bonding. For example, ammonium chloride (NH4Cl) has ionic bonding between a polyatomic ion, \(\ce{NH_4^{+}}\), and \(\ce{Cl^{−}}\) ions, but within the ammonium ion (NH4+), the nitrogen and hydrogen atoms are connected by covalent bonds (shown above).
Both ionic and covalent bonding are also found in calcium carbonate. Calcium carbonate (CaCO3) has ionic bonding between calcium ion \(\ce{Ca^{2+}}\) and a polyatomic ion, \(\ce{CO_3^{2-}}\), but within the carbonate ion (CO32-), the carbon and oxygen atoms are connected by covalent bonds (shown above).
Characteristics of Covalent (Molecular) Compounds
Compounds that contain covalent bonds (also called molecular compounds) exhibit different physical properties than ionic compounds. Because the attraction between molecules, which are electrically neutral, is weaker than that between electrically charged ions, covalent compounds generally have much lower melting and boiling points than ionic compounds. For example, water (molecular compound) boils at 100 °C while sodium chloride (ionic compound) boils at 1413 °C. In fact, many covalent compounds are liquids or gases at room temperature, and, in their solid states, they are typically much softer than ionic solids. Furthermore, whereas ionic compounds are good conductors of electricity when dissolved in water, most covalent compounds, being electrically neutral, are poor conductors of electricity in any state.
Is each compound formed from ionic bonds, covalent bonds, or both?
- \(\ce{Na_2O}\)
- \(\ce{Na_3PO_4}\)
- \(\ce{N_2O_4}\)
- Answer a
-
The elements in \(\ce{Na_2O}\) are a metal and a nonmetal, which form ionic bonds.
- Answer b
-
Because sodium is a metal and we recognize the formula for the phosphate ion, we know that this compound is ionic. However, within the polyatomic phosphate ion, the atoms are held together by covalent bonds, so this compound contains both ionic and covalent bonds.
- Answer c
-
The elements in \(\ce{N_2O_4}\) are both nonmetals, rather than a metal and a nonmetal. Therefore, the atoms form covalent bonds.
Is each compound are formed from ionic bonds, covalent bonds, or both?
- \(\ce{Ba(OH)_2}\)
- \(\ce{F_2}\)
- \(\ce{PCl_3}\)
- Answer a:
-
both
- Answer b:
-
covalent
- Answer c:
-
covalent
MOLECULAR FORMULAS
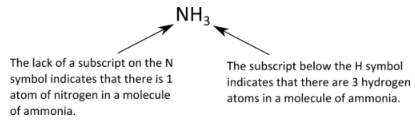
The chemical formulas for covalent compounds are referred to as molecular formulas because these compounds exist as separate, discrete molecules. Typically, a molecular formula begins with the nonmetal that is closest to the lower left corner of the periodic table, except that hydrogen is almost never written first (H2O is the prominent exception). Then the other nonmetal symbols are listed. Numerical subscripts are used if there is more than one of a particular atom. For example, we have already seen CH4, the molecular formula for methane. Below is the molecular formula of ammonia, NH3.
NAMING COVALENT COMPOUNDS
Naming binary (two-element) covalent compounds is similar to naming simple ionic compounds. The first element in the formula is simply listed using the name of the element. The second element is named by taking the stem of the element name and adding the suffix -ide. A system of numerical prefixes is used to specify the number of atoms in a molecule. Table \(\PageIndex{1}\) lists these numerical prefixes. Normally, no prefix is added to the first element’s name if there is only one atom of the first element in a molecule. If the second element is oxygen, the trailing vowel is usually omitted from the end of a polysyllabic prefix but not a monosyllabic one (that is, we would say “monoxide” rather than “monooxide” and “trioxide” rather than “troxide”).
| Number of Atoms in Compound | Prefix on the Name of the Element |
|---|---|
| 1 | mono-* |
| 2 | di- |
| 3 | tri- |
| 4 | tetra- |
| 5 | penta- |
| 6 | hexa- |
| 7 | hepta- |
| 8 | octa- |
| 9 | nona- |
| 10 | deca- |
| *This prefix is not used for the first element’s name. | |
Let us practice by naming the compound whose molecular formula is CCl4. The name begins with the name of the first element—carbon. The second element, chlorine, becomes chloride, and we attach the correct numerical prefix (“tetra-”) to indicate that the molecule contains four chlorine atoms. Putting these pieces together gives the name carbon tetrachloride for this compound.
Write the molecular formula for each compound.
- chlorine trifluoride
- phosphorus pentachloride
- sulfur dioxide
- dinitrogen pentoxide
Solution
If there is no numerical prefix on the first element’s name, we can assume that there is only one atom of that element in a molecule.
- ClF3
- PCl5
- SO2
- N2O5 (The di- prefix on nitrogen indicates that two nitrogen atoms are present.)
Write the molecular formula for each compound.
- nitrogen dioxide
- dioxygen difluoride
- sulfur hexafluoride
- selenium monoxide
- Answer a:
-
a. NO2
- Answer b:
-
O2F2
- Answer c:
-
SF6
- Answer d:
-
SeO
Because it is so unreactive, sulfur hexafluoride is used as a spark suppressant in electrical devices such as transformers.
Write the name for each compound.
- BrF5
- S2F2
- CO
Solution
- bromine pentafluoride
- disulfur difluoride
- carbon monoxide
Write the name for each compound.
- CF4
- SeCl2
- SO3
- Answer a:
-
carbon tetrafluoride
- Answer b:
-
selenium dichloride
- Answer c:
-
sulfur trioxide
For some simple covalent compounds, we use common names rather than systematic names. We have already encountered these compounds, but we list them here explicitly:
- H2O: water
- NH3: ammonia
- CH4: methane
Methane is the simplest organic compound. Organic compounds are compounds with carbon atoms and are named by a separate nomenclature system.
The prefix of mono are not used for the two molecules below:
HCl: hydrogen chloride
HF: hydrogen fluoride
Oxides of Nitrogen-Applications in Medicine
Nitric Oxide
Nitrogen monoxide (NO) is known more commonly as nitric oxide. It is a colorless gas formed when nitrogen and oxygen in the air combine when gasoline is burned in the engines of cars and trucks. So this compound has a bad reputation regarding atmospheric pollution. However, NO has many biological functions. It regulates blood pressure, dilates arteries and blood vessels, and is used by the immune system to help fight infections.
These effects of NO on the human body were noticed in the mid 19 th century soon after the discovery of the explosive nitroglycerin that was used to make dynamite. It was noticed that people who worked with nitroglycerin experienced splitting headaches which was the result of vasodilation (relaxation) of the blood vessels in the brain. This led to the use of nitroglycerin being used as a medication for the treatment of angina which is caused by constriction of the arteries that carry blood to the heart. A century later scientists discovered that the body produces NO from nitroglycerin and it is NO that causes coronary arteries to dilate reducing the symptoms of angina. In 1998 Nobel Prize in Medicine was awarded to three scientists who worked on the nitric oxide research.
Nitrous oxide
Nitrous oxide (N2O) is prepared by the careful thermal decomposition of ammonium nitrate (NH4NO3). Nitrous oxide is known as "laughing gas" due to the euphoric effects of inhaling it, a property that has led to its recreational use as a hallucinogen. However, it is as an anesthetic that it has a legitimate application. The first use of nitrous oxide as anesthetic drug was when dentist Horace Wells with assistance by Gardner Quincy Colton and John Mankey Riggs, demonstrated insensitivity to pain from a dental extraction in December 1844. Wells subsequently treated 12-15 patients, and according to his own record it only failed as an anesthetic in two cases.
The method did not come into general use until 1863, when Colton successfully used it for more than 25,000 patients. As such, the usage of nitrous oxide rapidly became the preferred anesthetic method in dentistry. The gas is mild enough to keep a patient in a conscious and conversational state, and yet in most cases strong enough to suppress the pain caused by dental work. It remains the preferred gas anesthetic in today's dentistry.
Key Takeaways
- The chemical formula of a simple covalent compound can be determined from its name.
- The name of a simple covalent compound can be determined from its chemical formula.


