Nucleophilic Reactions of Benzene Derivatives
- Page ID
- 1205
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)An early method of preparing phenol (the Dow process) involved the reaction of chlorobenzene with a concentrated sodium hydroxide solution at temperatures above 350 ºC. The chief products are phenol and diphenyl ether (see below). This apparent nucleophilic substitution reaction is surprising, since aryl halides are generally incapable of reacting by either an SN1 or SN2 pathway.
C6H5–Cl + NaOH solution |
350 ºC |
C6H5–OH + C6H5–O–C6H5 + NaCl |
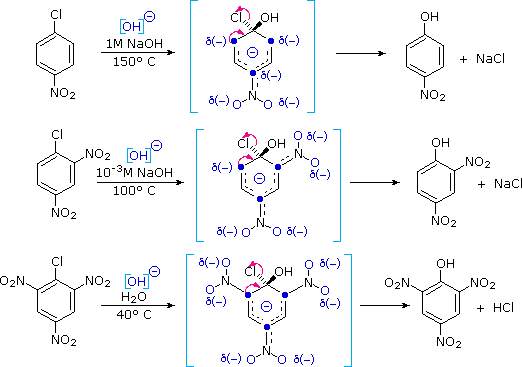
The presence of electron-withdrawing groups (such as nitro) ortho and para to the chlorine substantially enhance the rate of substitution, as shown in the set of equations presented below.
To explain this, a third mechanism for nucleophilic substitution has been proposed. This two-step mechanism is characterized by initial addition of the nucleophile (hydroxide ion or water) to the aromatic ring, followed by loss of a halide anion from the negatively charged intermediate as illustrated below. The sites over which the negative charge is delocalized are colored blue, and the ability of nitro, and other electron withdrawing, groups to stabilize adjacent negative charge accounts for their rate enhancing influence at the ortho and para locations.
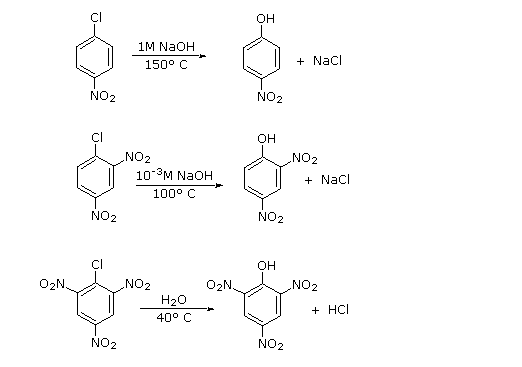
Three additional examples of aryl halide nucleophilic substitution are presented below. Only the 2- and 4-chloropyridine isomers undergo rapid substitution, the 3-chloro isomer is relatively unreactive. Nitrogen nucleophiles will also react, as evidenced by the use of Sanger's reagent for the derivatization of amino acids. The resulting N-2,4-dinitrophenyl derivatives are bright yellow crystalline compounds that facilitated analysis of peptides and proteins, a subject for which Frederick Sanger received one of his two Nobel Prizes in chemistry.
Additional Examples
Such addition-elimination processes generally occur at sp2 or sp hybridized carbon atoms, in contrast to SN1 and SN2 reactions. When applied to aromatic halides, as in the present discussion, this mechanism is called SNAr. Some distinguishing features of the three common nucleophilic substitution mechanisms are summarized in the following table.
|
Mechanism |
Number of Steps |
Bond Formation Timing |
Carbon Hybridization |
|---|---|---|---|
|
SN1 |
Two |
After Bond Breaking |
Usually sp3 |
|
SN2 |
One |
Simultaneous with |
Usually sp3 |
|
SNAr |
Two |
Prior to Bond Breaking |
Usually sp2 |
Nucleophilic Elimination Reactions
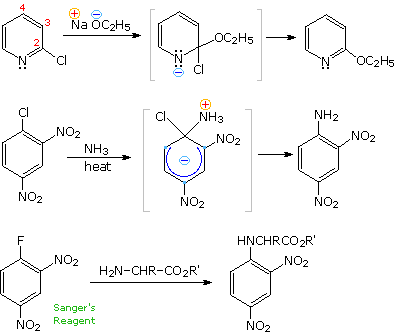
There is good evidence that the synthesis of phenol from chlorobenzene does not proceed by the addition-elimination mechanism (SNAr) as previously described. For example, treatment of para-chlorotoluene with sodium hydroxide solution at temperatures above 350 ºC gave an equimolar mixture of meta- and para-cresols (hydroxytoluenes). Chloro and bromobenzene reacted with the very strong base sodium amide (NaNH2 at low temperature (-33 ºC in liquid ammonia) to give good yields of aniline (aminobenzene). However, ortho-chloroanisole gave exclusively meta-methoxyaniline under the same conditions. These reactions are described by the following equations.
The mechanism for which is as follows:
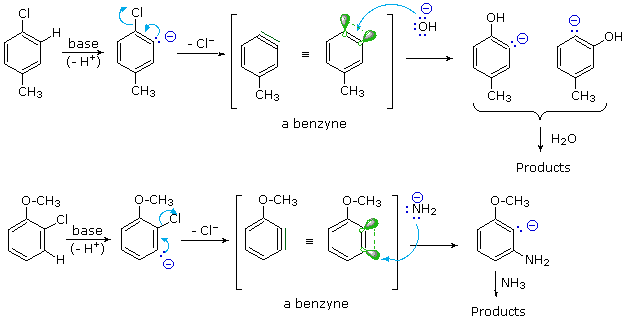
The explanation for this curious repositioning of the substituent group lies in a different two-step mechanism we can refer to as an elimination-addition process. The intermediate in this mechanism is an unstable benzyne species, as displayed in the above illustration by clicking the "Show Mechanism" button. In contrast to the parallel overlap of p-orbitals in a stable alkyne triple bond, the p-orbitals of a benzyne are tilted ca.120º apart, so the reactivity of this incipient triple bond to addition reactions is greatly enhanced. In the absence of steric hindrance (top example) equal amounts of meta- and para-cresols are obtained. The steric bulk of the methoxy group and the ability of its ether oxygen to stabilize an adjacent anion result in a substantial bias in the addition of amide anion or ammonia.
Nucleophilic Addition Reactions
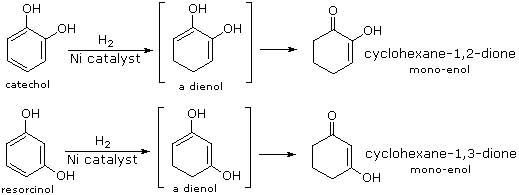
Although it does so less readily than simple alkenes or dienes, benzene adds hydrogen at high pressure in the presence of Pt, Pd or Ni catalysts. The product is cyclohexane and the heat of reaction provides evidence of benzene's thermodynamic stability. Substituted benzene rings may also be reduced in this fashion, and hydroxy-substituted compounds, such as phenol, catechol and resorcinol, give carbonyl products resulting from the fast ketonization of intermediate enols. Nickel catalysts are often used for this purpose, as noted in the following equations.
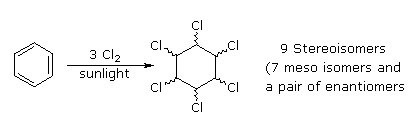
Benzene is more susceptible to radical addition reactions than to electrophilic addition. We have already noted that benzene does not react with chlorine or bromine in the absence of a catalyst and heat. In strong sunlight or with radical initiators benzene adds these halogens to give hexahalocyclohexanes. It is worth noting that these same conditions effect radical substitution of cyclohexane, the key factors in this change of behavior are the pi-bonds array in benzene, which permit addition, and the weaker C-H bonds in cyclohexane. The addition of chlorine is shown below on the left; two of the seven meso-stereoisomers are displayed to the right.
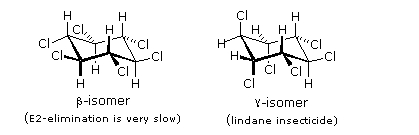
The Birch Reduction
Another way of adding hydrogen to the benzene ring is by treatment with the electron rich solution of alkali metals, usually lithium or sodium, in liquid ammonia. See examples of this reaction, which is called the Birch Reduction.
Contributors
- William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry