Gas Laws - Overview
- Page ID
- 1517
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Created in the early 17th century, the gas laws have been around to assist scientists in finding volumes, amount, pressures and temperature when coming to matters of gas. The gas laws consist of three primary laws: Charles' Law, Boyle's Law and Avogadro's Law (all of which will later combine into the General Gas Equation and Ideal Gas Law).
Introduction
The three fundamental gas laws discover the relationship of pressure, temperature, volume and amount of gas. Boyle's Law tells us that the volume of gas increases as the pressure decreases. Charles' Law tells us that the volume of gas increases as the temperature increases. And Avogadro's Law tell us that the volume of gas increases as the amount of gas increases. The ideal gas law is the combination of the three simple gas laws.
Ideal Gases
Ideal gas, or perfect gas, is the theoretical substance that helps establish the relationship of four gas variables, pressure (P), volume (V), the amount of gas (n) and temperature(T). It has characters described as follow:
- The particles in the gas are extremely small, so the gas does not occupy any spaces.
- The ideal gas has constant, random and straight-line motion.
- No forces between the particles of the gas. Particles only collide elastically with each other and with the walls of container.
Real Gases
Real gas, in contrast, has real volume and the collision of the particles is not elastic, because there are attractive forces between particles. As a result, the volume of real gas is much larger than of the ideal gas, and the pressure of real gas is lower than of ideal gas. All real gases tend to perform ideal gas behavior at low pressure and relatively high temperature.
The compressibility factor (Z) tells us how much the real gases differ from ideal gas behavior.
\[ Z = \dfrac{PV}{nRT} \nonumber \]
For ideal gases, \( Z = 1 \). For real gases, \( Z\neq 1 \).
Boyle's Law
In 1662, Robert Boyle discovered the correlation between Pressure (P)and Volume (V) (assuming Temperature(T) and Amount of Gas(n) remain constant):
\[ P\propto \dfrac{1}{V} \rightarrow PV=x \nonumber \]
where x is a constant depending on amount of gas at a given temperature.
- Pressure is inversely proportional to Volume
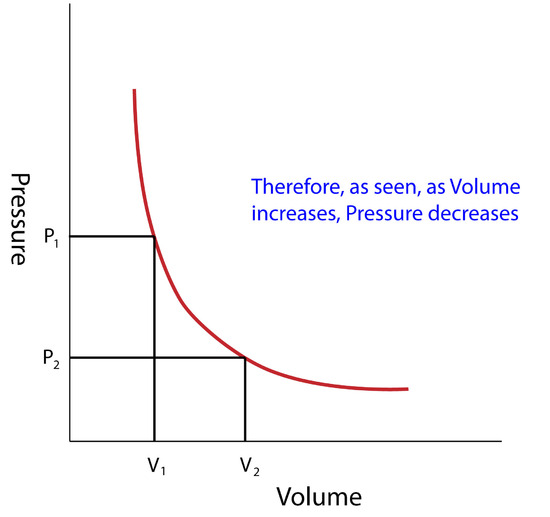
Another form of the equation (assuming there are 2 sets of conditions, and setting both constants to eachother) that might help solve problems is:
\[ P_1V_1 = x = P_2V_2 \nonumber \]
A 17.50mL sample of gas is at 4.500 atm. What will be the volume if the pressure becomes 1.500 atm, with a fixed amount of gas and temperature?
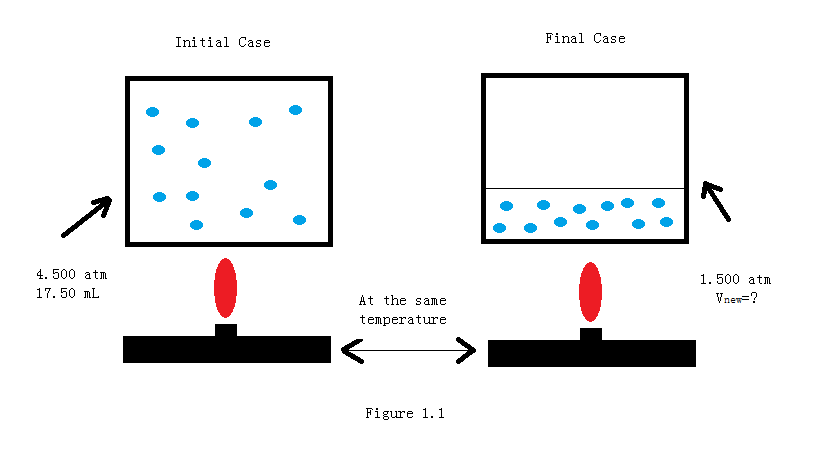
Solution
\[ \begin{align*} V_2 &= \dfrac {P_1 V_1}{P_2} \\[4pt] &=\dfrac{4.500~ atm \cdot 17.50~mL}{1.500 ~atm} \\[4pt] &= 52.50~mL \end{align*} \]
Charles' Law
In 1787, French physicists Jacques Charles, discovered the correlation between Temperature(T) and Volume(V) (assuming Pressure (P) and Amount of Gas(n) remain constant):
\[ V \propto T \rightarrow V=yT \nonumber \]
where y is a constant depending on amount of gas and pressure. Volume is directly proportional to Temperature
.jpg?revision=1)
Another form of the equation (assuming there are 2 sets of conditions, and setting both constants to each other) that might help solve problems is:
\[ \dfrac{V_1}{T_1} = y = \dfrac{V_2}{T_2} \nonumber \]
A sample of Carbon dioxide in a pump has volume of 20.5 mL and it is at 40.0 oC. When the amount of gas and pressure remain constant, find the new volume of Carbon dioxide in the pump if temperature is increased to 65.0 oC.
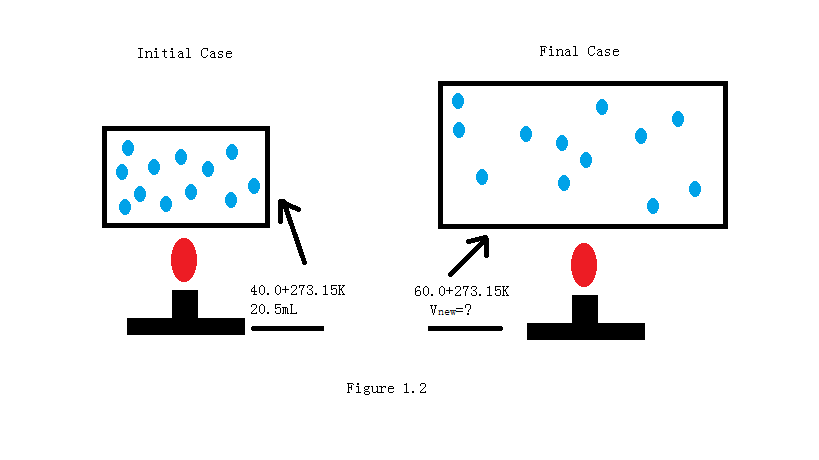
Solution
\[ \begin{align*} V_2&=\dfrac{V_1 \,T_2}{T_1} \\[4pt] &=\dfrac{20.5~mL (60+273.15~K)}{40+273.15~K} \\[4pt] &= 22.1~mL \end{align*} \]
Avogadro's Law
In 1811, Amedeo Avogadro fixed Gay-Lussac's issue in finding the correlation between the Amount of gas(n) and Volume(V) (assuming Temperature(T) and Pressure(P) remain constant):
\[ V \propto n \rightarrow V = zn \nonumber \]
where \(z\) is a constant depending on Pressure and Temperature.
- Volume (V) is directly proportional to the Amount of gas (n)
Another form of the equation (assuming there are 2 sets of conditions, and setting both constants to each other) that might help solve problems is:
\[ \dfrac{P_1}{n_1} = z= \dfrac{P_2}{n_2} \nonumber \]
A 3.80 g of oxygen gas in a pump has volume of 150 mL. constant temperature and pressure. If 1.20g of oxygen gas is added into the pump. What will be the new volume of oxygen gas in the pump if temperature and pressure held constant?
Solution
V1=150 mL
\[ n_1= \dfrac{m_1}{M_{\text{oxygen gas}}} \nonumber \]
\[ n_2= \dfrac{m_2}{M_{\text{oxygen gas}}} \nonumber \]
\[ \begin{align*} V_2 &=\dfrac{V_1 \cdot n_2}{n_1} \\[4pt] &= \dfrac{150~mL \cdot \dfrac{5.00~g}{32.0~g \cdot mol^{-1}}} {\dfrac{3.80~g}{32.0~g \cdot mol^{-1}}} \\[4pt] &= 197~mL\end{align*} \]
Ideal Gas Law
The ideal gas law is the combination of the three simple gas laws. By setting all three laws directly or inversely proportional to Volume, you get:
\[ V \propto \dfrac{nT}{P} \nonumber \]
Next replacing the directly proportional to sign with a constant(R) you get:
\[ V = \dfrac{RnT}{P} \nonumber \]
And finally get the equation:
\[ PV = nRT \nonumber \]
where \(P\) is the absolute pressure of ideal gas
- \(V\) is the volume of ideal gas
- \(n\) is the amount of gas
- \(T\) is the absolute temperature
- \(R\) is the gas constant
Here, \(R\) is the called the gas constant. The value of \(R\) is determined by experimental results and its numerical value changes with units.
\[\begin{align} R &= 8.3145 ~J \cdot mol^{-1} \cdot K^{-1}~ \tag{in SI Units}
\\[4pt] &= 0.082057 ~L \cdot atm \cdot K^{-1} \cdot mol^{-1} \nonumber \end{align}\]
At 655 mmHg and 25.0 oC, a sample of Chlorine gas has volume of 750 mL. How many moles of chlorine gas at this condition?
- P=655 mmHg
- T=25+273.15 K
- V=750 mL=0.75L
- n=?
Solution
\[\begin{align*} n &=\frac{PV}{RT} \\[4pt] &=\frac{655~mmHg \cdot \frac{1 ~atm}{760~mmHg} \cdot 0.75~L}{0.082057~L \cdot atm \cdot mol^{-1} \cdot K^{-1} \cdot (25+273.15K) } \\[4pt] &=0.026~ mol \end{align*} \]
Evaluation of the Gas Constant, R
You can get the numerical value of gas constant, R, from the ideal gas equation, \(PV=nRT\). At standard temperature and pressure, where temperature is 0 oC, or 273.15 K, pressure is at 1 atm, and with a volume of \(22.4140~L\),
\[ \begin{align*} R &= \frac{PV}{nT} \\[4pt] &= \frac{1 ~atm \cdot 22.4140~L}{1 ~mol \cdot 273.15~K} \\[4pt] &=0.082057 \; L \cdot atm \cdot mol^{-1} K^{-1} \end{align*}\]
\[ \begin{align*} R &= \frac{PV}{nT} \\[4pt] &= \frac{1~ atm \cdot 2.24140 \times 10^{-2}~m^3}{1 ~mol \cdot 273.15~K} \\[4pt] &= 8.3145\; m^3\; Pa \cdot mol^{-1} \cdot K^{-1} \end{align*}\]
General Gas Equation
In an Ideal Gas situation, \( \frac{PV}{nRT} = 1 \) (assuming all gases are "ideal" or perfect). In cases where \( \frac{PV}{nRT} \neq 1 \) or if there are multiple sets of conditions (Pressure(P), Volume(V), number of gas(n), and Temperature(T)), use the General Gas Equation:
Assuming 2 set of conditions:
Initial Case: Final Case:
\[ P_iV_i = n_iRT_i \; \; \; \; \; \; P_fV_f = n_fRT_f \nonumber \]
Setting both sides to R (which is a constant with the same value in each case), one gets:
\[ R= \dfrac{P_iV_i}{n_iT_i} \; \; \; \; \; \; R= \dfrac{P_fV_f}{n_fT_f} \nonumber \]
If one substitutes one R for the other, one will get the final equation and the General Gas Equation:
\[ \dfrac{P_iV_i}{n_iT_i} = \dfrac{P_fV_f}{n_fT_f} \nonumber \]
Standard Conditions
If in any of the laws, a variable is not give, assume that it is given. For constant temperature, pressure and amount:
- Absolute Zero (Kelvin): 0 K = -273.15 oC
T(K) = T(oC) + 273.15 (unit of the temperature must be Kelvin)
2. Pressure: 1 Atmosphere (760 mmHg)
3. Amount: 1 mol = 22.4 Liter of gas
4. In the Ideal Gas Law, the gas constant R = 8.3145 Joules · mol-1 · K-1
= 0.082057 L · atm·K-1 · mol-1
The Van der Waals Equation For Real Gases
Dutch physicist Johannes Van Der Waals developed an equation for describing the deviation of real gases from the ideal gas. There are two correction terms added into the ideal gas equation. They are \( 1 +a\frac{n^2}{V^2}\), and \( 1/(V-nb) \).
Since the attractive forces between molecules do exist in real gases, the pressure of real gases is actually lower than of the ideal gas equation. This condition is considered in the van der Waals equation. Therefore, the correction term \( 1 +a\frac{n^2}{V^2} \) corrects the pressure of real gas for the effect of attractive forces between gas molecules.
Similarly, because gas molecules have volume, the volume of real gas is much larger than of the ideal gas, the correction term \(1 -nb \) is used for correcting the volume filled by gas molecules.
If 4L of H2 gas at 1.43 atm is at standard temperature, and the pressure were to increase by a factor of 2/3, what is the final volume of the H2 gas? (Hint: Boyle's Law)
- Answer
-
To solve this question you need to use Boyle's Law:
\[ P_1V_1 = P_2V_2 \nonumber \]
Keeping the key variables in mind, temperature and the amount of gas is constant and therefore can be put aside, the only ones necessary are:
Initial Pressure: 1.43 atm Initial Volume: 4 L Final Pressure: 1.43x1.67 = 2.39Plugging these values into the equation you get:
V2=(1.43atm x 4 L)/(2.39atm) = 2.38 L
- Final Volume(unknown): V2
If 1.25L of gas exists at 35 oC with a constant pressure of .70 atm in a cylindrical block and the volume were to be multiplied by a factor of 3/5, what is the new temperature of the gas? (Hint: Charles's Law)
- Answer
-
To solve this question you need to use Charles's Law:
\[\frac{V_1}{T_1}=\frac{V_2}{T_2} \nonumber \]
Once again keep the key variables in mind. The pressure remained constant and since the amount of gas is not mentioned, we assume it remains constant. Otherwise the key variables are:
Initial Volume: 1.25 L Initial Temperature: 35oC + 273.15 = 308.15K Final Volume: 1.25L*3/5 = .75 LSince we need to solve for the final temperature you can rearrange Charles's:
\[T_2=\frac{T_1 V_2}{V_1} \nonumber \]
Once you plug in the numbers, you get: T2=(308.15 K x .75 L)/(1.25 L) = 184.89 K- Final Temperature: T2
A ballon with 4.00g of Helium gas has a volume of 500mL. When the temperature and pressure remain constant. What will be the new volume of Helium in the ballon if another 4.00g of Helium is added into the ballon? (Hint: Avogadro's Law)
- Answer
-
Using Avogadro's Law to solve this problem, you can switch the equation into \( V_2=\frac{n_1\centerdot V_2}{n_2} \). However, you need to convert grams of Helium gas into moles.
\[ n_1 = \frac{4.00g}{4.00g/mol} = \text{1 mol} \nonumber \]
Similarly, n2=2 mol
\[ V_2=\frac{n_2 \centerdot V_2}{n_1} \nonumber \]
\[ =\frac{2 mol \centerdot 500mL}{1 mol} \nonumber \]
\[ = \text{1000 mL or 1L } \nonumber \]
References
- Petrucci, Ralph H. General Chemistry: Principles and Modern Applications. 9th Ed. Upper Saddle River, NJ: Pearson Prentice Hall, 2007.
- Staley, Dennis. Prentice Hall Chemistry. Boston, MA: Pearson Prentice Hall, 2007.
-
Olander, Donald R. "Chapter2 Equation of State." General Thermodynamics. Boca Raton, NW: CRC, 2008. Print
-
O'Connell, John P., and J. M. Haile. "Properties Relative to Ideal Gases." Thermodynamics: Fundamentals for Applications. Cambridge: Cambridge UP, 2005. Print.
-
Ghare, Shakuntala. "Ideal Gas Laws for One Component." Ideal Gas Law, Enthalpy, Heat Capacity, Heats of Solution and Mixing. Vol. 4. New York, NY, 1984. Print. F.

