Predicting the Bond-Order of Diatomic Species
- Page ID
- 35764
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Bond-order usually predicted from the Molecular Orbital Theory1,2. Molecular Orbital Theory (MOT) was first proposed by Friedrich Hund and Robert Mulliken in 1933. They developed an approach to covalent bond formation which is based upon the effects of the various electron fields upon each other and which employs molecular orbital rather than atomic orbital. Each such orbital characterizing the molecule as a whole is described by a definite combination of quantum numbers and possesses relative energy value.
In this article text based learning approaches have been highlighted by innovative and time economic way3-5 to enhance interest of students’ who belong to paranoia zone in chemical bonding. In this pedagogical survey, I have tried to hub one time economic pedagogy by including four (04) new formulae in the field of chemical education. This article explores the results and gives implications for context based teaching, learning and assessment in a time economic way.
Classification
First of all we classify the molecules or ions into the following four sets based on total number of electrons present in them.
SET 1: Molecules and ions having total no of electrons within the range (1-2):
In such case Bond order = n/2; [Where n = Total no of electrons]
Eg. H2 (Total electrons = 2), Therefore B.O. = n/2 = 2/2 = 1
SET 2: Molecules and ions having total no of electrons within the range (2-6):
In such case Bond order = I 4- n I / 2 ;
where n = Total no of electrons, ‘I I’ indicates Mod function i.e. the value of bond order is always positive]
Eg. Li2+(5electrons) Therefore B.O. = I 4-5 I / 2 = 1/2 = 0.5.
SET 3: Molecules and ions having total no of electrons within the range (6-14):
In such case Bond order = I 8-n I / 2
Eg: CO (Total electrons = 6+8=14), Therefore B.O.= I 8-14 I / 2 = 3
SET 4: Molecules and ions having total no of electrons within the range (14-20):
In such case Bond order = (20-n) / 2 ; [Where n = Total no of electrons]
Eg. NO (Total electrons = 15), Therefore B.O. = 20-15/2 = 2.5
|
Species (Molecules or ions) |
Total Number of electrons (n) |
Bond-Order (B.O.) |
|---|---|---|
|
Bond-Order Values for the species having (1-2) electrons ; Bond order = n/2 |
||
|
H2+ H2, He22+ |
1 2 |
0.5 1 |
|
Bond-Order Values for the species having (2-6) electrons ; Bond order = I 4- n I / 2 |
||
|
H2-,He2+ He2, Li2+,He2- Li2, He22-, Be22+ |
3 4 5 6 |
0.5 0 0.5 1 |
|
Bond-Order Values for the species having (6-14) electrons ; Bond order = I 8- n I / 2 |
||
|
Be2+, Li2- Be2, Li22- Be2-, B2+ B2, Be22-, HF B2-, C2+ C2, B22-, N22+, CN+ C2-, N2+ N2, CO, NO+, C22-, CN-,O22+ |
7 8 9 10 11 12 13 14 |
0.5 0 0.5 1 1.5 2 2.5 3 |
|
Bond-Order Values for the species having (14-20) electrons; Bond order = (20-n) / 2 |
||
|
N2-, NO, O2+ NO-, O2 O2- F2, O22-, HCl F2- Ne2 |
15 16 17 18 19 20 |
2.5 2 1.5 1 0.5 0 |
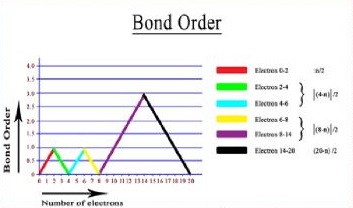
Graphical Presentation of Bond-Order
The graphical representation presented in Fig. 1 shows that bond-order gradually increases to 1 in the range (0-2) electrons then falls to zero in the range (2-4) electrons then it further rises to 1 for (4-6) electrons and once again falls to zero for (6-8) electrons then again rises to 3 in the range (8-14) electrons and then finally falls to zero for (14-20) electrons. For total no of electrons 2, 6 and 14, we may use multiple formulae, because they fall in the overlapping region in which they intersect with each other.
References
- “Spectroscopy. Molecular Orbitals and Chemical Bonding”, Nobel Lectures, Chemistry 1963-1970,Elsevier Publishing Company, 1972-1966.
- Hall, George G. Lennard-Jones Paper of “Foundations of Molecular Orbital Theory”, Advances in Quantum Chemistry,1929, 22. Bibcode:1991AdQC…22…1H. doi:10.1016/S0065-3276(08)60361-5, ISBN-978-0-12-034822-0, ISSN 0065-3276.
- Arijit Das, ‘New Methods for Prediction of Bond Order of Mono and Diatomic Homo and Hetero Nuclear Molecules or Ions Having (1-20)electrons and Oxide Based Acid Radicals Without MOT – a Rapid Innovative Approach’,IJAR, 2013, 3(11), 41-43, ISSN-2249-555X.
- Arijit Das, ‘Simple Thinking Makes Chemistry Metabolic And Interesting- A Review Article’, IOSR-JAC, 2013, 6(4), 8-15, e-ISSN: 2278-5736, doi:10.9790/5736-0640815.
- Arijit Das, R.Sanjeev and V.Jagannadham, “Innovative And Time Economic Pedagogical Views In Chemical Education – A Review Article”, World Journal of Chemical Education, 2014, 2(3), 29-38, Science and Education Publishing , USA, DOI:10.12691/wjce-2-3-1.
External Links
- communities.acs.org/docs/DOC-46667
- communities.acs.org/docs/<wbr/>DOC-45853
- www.drarijitdaschem.in/Innova...Views%20in.pdf
Contributor
- Dr. Arijit Das, Ph.D. (Inorganic Chemistry), MACS ( Invited,USA ), SFICS, MISC, MIAFS (India), Assistant Professor, Department of Chemistry, Ramthakur College, Agartala, Tripura(W), Tripura, India, Pin-799003.

