13.1: Intermolecular Interactions
- Page ID
- 41388
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Learning Objectives
- Classify intermolecular forces as ionic, covalent, London dispersion, dipole-dipole, or hydrogen bonding.
- Explain properties of material in terms of type of intermolecular forces.
- Predict the properties of a substance based on the dominant intermolecular force.
Forces binding atoms in a molecule are due to chemical bonding. The energy required to break a bond is called the bond-energy. For example, the average bond-energy for \(\ce{O-H}\) bonds in water is 463 kJ/mol. On average, 463 kJ is required to break 6.023x1023 \(\ce{O-H}\) bonds, or 926 kJ to convert 1.0 mole of water into 1.0 mol of \(\ce{O}\) and 2.0 mol of \(\ce{H}\) atoms. The forces holding molecules together are generally called intermolecular forces. The energy required to break molecules apart is much smaller than a typical bond-energy, but intermolecular forces play important roles in determining the properties of a substance. Intermolecular forces are particularly important in terms of how molecules interact and form biological organisms or even life. This link gives an excellent introduction to the interactions between molecules.
Classifying Intermolecular Forces
In general, intermolecular forces can be divided into several categories. The four prominent types are:
- Ion-Ion Interactions: Recall lattice energy and its relation to properties of solids. The more ionic, the higher the lattice energy. Examine the following list and see if you can explain the observed values by way of ionic attraction: \(\mathrm{LiF = 1036}\); \(\mathrm{LiI = 737}\) ; \(\mathrm{KF = 821}\); \(\mathrm{MgF_2 = 2957\: kJ/mol}\)
- Dipole-dipole Interactions: Substances whose molecules have dipole moment have a higher melting point or boiling point than those of similar molecular mass, but whose molecules have no dipole moment.
- London dispersion forces or van der Waal's force: These forces always operate in any substance. The force arisen from induced dipole and the interaction is weaker than the dipole-dipole interaction. In general, the heavier the molecule, the stronger the van der Waal's force of interaction. For example, the boiling points of inert gases increase as their atomic masses increase due to stronger London dispersion interactions.
- Hydrogen bonds: Certain substances such as \(\ce{H2O}\), \(\ce{HF}\), and \(\ce{NH3}\) form hydrogen bonds, which affects properties (mp, bp, solubility) of the substance. Other compounds containing \(\ce{OH}\) and \(\ce{NH2}\) groups also form hydrogen bonds. Molecules of many organic compounds such as alcohols, acids, amines, and amino acids contain these groups, and thus hydrogen bonding plays a important role in biological science.
- Covalent bonding: Covalent bonding is really intramolecular force rather than intermolecular force. It is mentioned here, because some solids are formed due to covalent bonding. For example, in diamond, silicon, quartz etc., all the atoms in the entire crystal are linked together by covalent bonding. These solids are hard, brittle, and have high melting points. Covalent bonding holds atoms tighter than ionic attraction.
- Metallic bonding: Forces between atoms in metallic solids belong to another category. Valence electrons in metals are rampant. They are not restricted to certain atoms or bonds. Rather they run freely in the entire solid, providing good conductivity for heat and electric energy. This behavior of electrons gives special properties such as ductility and mechanical strength to metals.
The division into types is for convenience in their discussion. Of course all types can be present simultaneously for many substances. Usually, intermolecular forces are discussed together with The States of Matter. Intermolecular forces also play important roles in solutions, a discussion of which is given in Hydration, solvation in water. A summary of the interactions is illustrated in the following diagram:
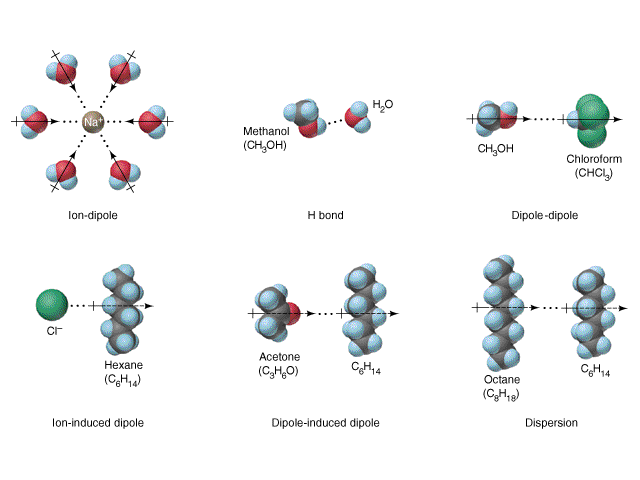
See if you can answer the following questions.
- What are dipoles?
- What are dipole moments?
- How do dipoles interact?
- Why do molecules attract one another?
- How do London dispersion forces come about?
- What parameters cause an increase of the London dispersion forces?
- What is a hydrogen bond?
- What type of hydrogen bonds are strong?
- What chemical groups are hydrogen acceptors for hydrogen bonds?
If you are looking for specific information, your study will be efficient. Some answers can be found in the Confidence Building Questions. Consider carefully the purpose of each question, and figure out what there is to be learned in it.
Confidence Building Questions
- Which of the following molecules have a permanent dipole moment?
- \(\ce{H2O}\)
- \(\ce{CO2}\)
- \(\ce{CH4}\)
- \(\ce{N2}\)
- \(\ce{CO}\)
- \(\ce{NH3}\)
Discussion -
\(\ce{CO2}\), \(\ce{CH4}\), and \(\ce{N2}\) are symmetric, and hence they have no permanent dipole moments. A molecule with polar bonds unsymmetrically arranged will possess a permanent dipole. - Which has the higher boiling point, \(\ce{Br2}\) or \(\ce{ICl}\)?
Discussion -
They have similar molecular weights: \(\mathrm{Br_2 = 160}\); \(\mathrm{ICl = 162}\). Their boiling points are 332 K and 370 K respectively. - An atom or molecule can be temporarily polarized by a nearby species. Polarization separates centers of charge giving
- permanent dipole
- temporary charges
- hydrogen bonding
- induced dipole
- induced ions
- radicals
Discussion -
Induced dipoles are responsible for the London dispersion forces. The heavier the molecule, the larger the induced dipole will be. Thus, London dispersion forces are strong for heavy molecules. - Which has a higher boiling point, \(\ce{I2}\) or \(\ce{Br2}\)?
Discussion -
Atomic weights for \(\ce{Br}\) and \(\ce{I}\) are 80 and 127 respectively. The higher the molecular weight, the stronger the London dispersion forces. - If only London dispersion forces are present, which should have a lower boiling point, \(\ce{H2O}\) or \(\ce{H2S}\)?
Discussion -
The b.p. for \(\ce{H2O}\) is 100 deg C, and that of \(\ce{H2S}\) is -70 deg C. Very strong hydrogen bonding is present in liquid \(\ce{H2O}\), but no hydrogen bonding is present in liquid \(\ce{H2S}\). - Contrary to most other substances, the density of water decreases as temperature decreases between 4 and 0 deg C. This is due to
- dipole-dipole interaction
- London dispersion
- decreasing number of hydrogen bonds
- increasing number of hydrogen bonds formed
Discussion -
As more hydrogen bonds form when the temperature decreases, the volume expands, causing a decrease in density. Above 4 deg C, the thermal expansion is more prominent than the effect of hydrogen bonds. - Ethanol (\(\ce{C2H5OH}\), molar mass 46) boils at 351 K, but water (\(\ce{H2O}\), molar mass 18) boils at higher temperature, 373 K. This is because:
- water is denser
- water has stronger London dispersion forces
- water has stronger hydrogen bonds
- water molecules contain no carbon
Discussion -
A hydrogen atom between two small, electronegative atoms (such as \(\ce{F}\), \(\ce{O}\), \(\ce{N}\)) causes a strong intermolecular interaction known as the hydrogen bond. The strength of a hydrogen bond depends upon the electronegativities and sizes of the two atoms. - Ethanol (\(\ce{C2H5OH}\)) and methyl ether (\(\ce{CH3OCH3}\)) have the same molar mass. Which has a higher boiling point?
Hint: Ethanol has a higher boiling point.
Discussion -
\(\ce{R-OH}\) group is both proton donor and acceptor for hydrogen bonding. Methyl groups have very weak hydrogen bonding, if any.
Contributors and Attributions
Chung (Peter) Chieh (Professor Emeritus, Chemistry @ University of Waterloo)

