Making Epoxyethane from Ethene
- Page ID
- 3888
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)This page looks at the manufacture of epoxyethane from ethene, and then at some of the products that are made from epoxyethane.
The manufacture of epoxyethane
Conditions
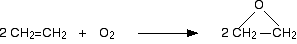
| Temperature: | about 250 - 300°C |
| Pressure: | about 15 atmospheres |
| Catalyst: | silver |
Problems and hazards during manufacture
The main problem comes in controlling the temperature. The reaction is exothermic and so the temperature will tend to rise unless it is carefully controlled. At higher temperatures the ethene burns in the oxygen to produce carbon dioxide and water which means that the temperature would increase even more - and the whole thing get completely out of hand! Two hazards during manufacture come from the nature of epoxyethane. It is
- poisonous and carcinogenic (cancer producing);
- highly flammable or explosive in contact with air.
The reactivity of epoxyethane
The reason that epoxyethane is so reactive is that bonding pairs in the ring of atoms in the molecule are forced very close together. The bond angles are about 60° rather than about 109.5° when carbon atoms normally form single bonds.
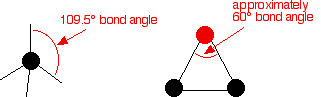
The overlap between the atomic orbitals in forming the carbon-carbon and carbon-oxygen bonds is less good than it is normally, and there is considerable repulsion between the bonding pairs. The system becomes more stable if the ring is broken. When epoxyethane reacts a carbon-oxygen bond is always broken and the ring opens up.
Uses of epoxyethane
Manufacture of ethane-1,2-diol (ethylene glycol)
Acid catalyzed hydrolysis of epoxyethane
Epoxyethane reacts with water in the presence of an acid catalyst (very dilute sulphuric acid) at a temperature of about 60°C. Ethane-1,2-diol is produced.
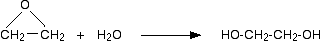
A large excess of water is used to try to prevent the product from reacting with the original epoxyethane. Ethane-1,2-diol is an alcohol (because it contains simple -OH groups), and alcohols react with epoxyethane (see below). Even in the presence of a large excess of water, this reaction happens as well:
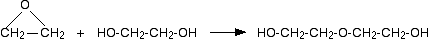
The product is still an alcohol, and similar reactions can also lead to quite long chains.
Uses of ethane-1,2-diol
Ethane-1,2-diol is used as an antifreeze in car engines. It is added to the cooling water to prevent it from freezing under very cold conditions. Ethane-1,2-diol is also used in the manufacture of polyesters such as poly(ethylene terephthalate). You may have come across this as a fibre used to make clothes (perhaps under the brand name Terylene), or as a clear material used to make plastic drinks bottles (PET).
The reaction of epoxyethane with alcohols
This is a reaction which students at this level often find difficulty remembering. It is actually probably easier to work out than remember. Think of it as an extension of the reaction with water.
Alcohols have the formula R-OH, where R is an alkyl group. Water can be thought of as H-OH. The reaction of epoxyethane with water can be color-coded like this:
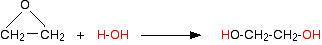
Now do the same thing with the alcohol:
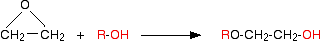
Product molecules of this type are used as solvents.
Notice that the product is still an alcohol. It has an -OH group at the right-hand end of the molecule. If the epoxyethane is in excess, the reaction can continue. (In fact, it continues to some extent even if the epoxyethane isn't in excess.)
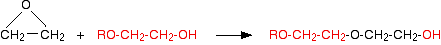
The product from this reaction is again an alcohol, and can go on to react with even more epoxyethane! What you get eventually is a chain with a structure:
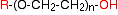
Compounds of this type are used as plasticisers (added, for example, to PVC to make it more flexible) or as non-ionic surfactants (detergents). To make the surfactant, you would start with a fairly long chain alcohol to produce a molecule such as:
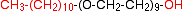
Contributors
Jim Clark (Chemguide.co.uk)


