21.5: Reduction of Carboxyl Groups to Aldehydes Using Modified Hydride Reagents
- Page ID
- 216884
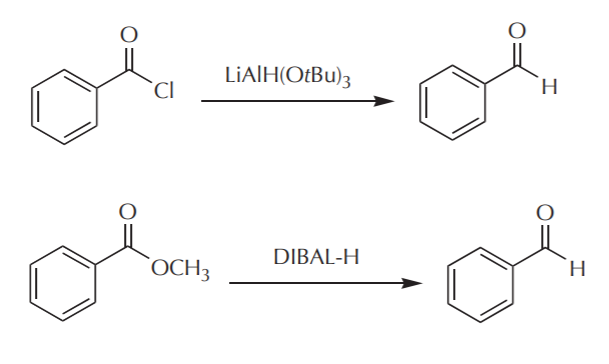
It was stated before that carboxyl groups get reduced all the way to primary alcohols when full reactivity reducing agents are used. The mechanism of reduction goes through an aldehyde stage, but it cannot stop there because the aldehyde gets further reduced to alcohol. So the question is, can we stop at the aldehyde stage by using modified hydride reagents that have bulky groups in the structure and are capable of delivering only one hydride per equivalent. The answer is yes, because those reactions are slower and we can control the number of hydride ions delivered, so that by limiting this parameter we prevent the aldehyde from undergoing further reduction. Such reductions can be accomplished using DIBAL-H or LiAlH(OtBu)3
SEE NEXT PAGE FOR A SUMMARY OF REDUCTIONS OF CARBONYL AND CARBOXYL GROUPS


