11.2: Using Resonance Structures to Predict Relative Reactivities of Basic Sites
- Page ID
- 215880
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
Sometimes a molecule can have several atoms which could be potentially basic. The question then is, is one more basic than the other, or are they of equal basicity? Sometimes resonance structures can reveal relative reactivities that are not apparent from the main, or most stable contributor. An example is acetic acid, which contains two oxygen atoms that could in principle become protonated by acid. The problem can be approached in two ways. One is to look at the relative stabilities of the conjugate bases. It is shown below that the conjugate base that results from protonation of oxygen (a) is resonance stabilized, whereas the one that results from protonation of oxygen (b) is not. Based on this criterion, one predicts that oxygen (a) is more basic than oxygen (b), and that protonation will occur predominantly at oxygen (a).
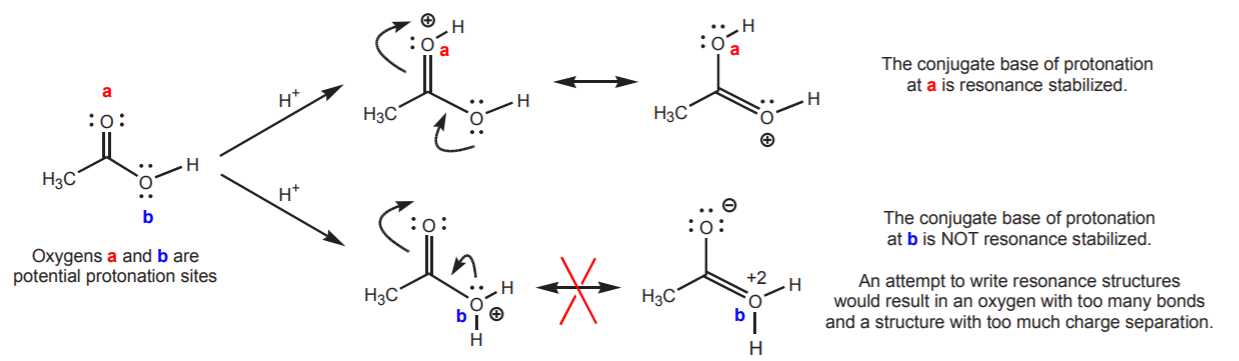
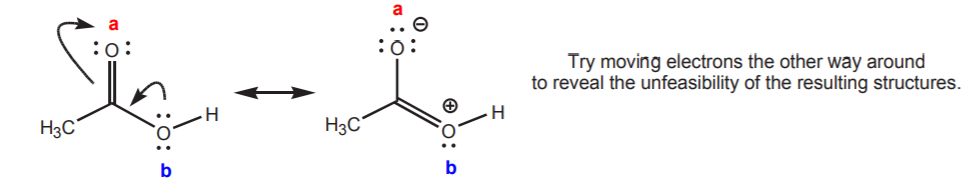
One can arrive at the same conclusion by examining the resonance structures of acetic acid, which reveal that oxygen (a) carries a partial negative charge and that oxygen (b) carries a partial positive charge. The more negative character of (a) makes it the more basic of the two.
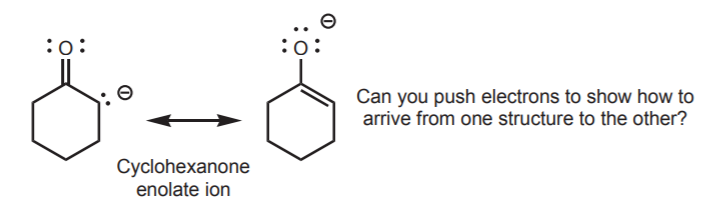
Resonance structures can be used in conjunction with other information, such as periodic trends, to make a prediction. In the example below, the two resonance structures of the cyclohexanone enolate ion show carbon and oxygen sharing the negative charge. However, basicity trends in the periodic table predict that in comparable situations, carbon is more basic than oxygen. Remember that we’re not looking at electronegativity trends here, but basicity, which runs opposite to electronegativity across the same row. The molecule will react with acids to favor protonation of the carbon over the oxygen atom.
Writing resonance structures involving σ-bonds is not as common as doing it with π-bonds. However it’s perfectly acceptable to do it in the case of highly polarized σ-bonds, such as might be the case in H-Cl.
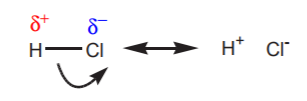
Although fairly obvious to a chemistry student, the structure on the right makes the acidic nature of the hydrogen atom apparent. The same can be done with bases. One way to have a highly polarized σ-bond involving carbon is by bonding it to a metal. The higher electronegativity of carbon biases the electron density distribution in its favor. In this case, resonance structures reveal the negative character of such carbon, and therefore its basic character.



