9.6: Molecules With 2 or More Chiral Centers- Diastereomers and Meso Forms
- Page ID
- 215743
As was stated before, molecules containing 2 or more chiral centers may or may not be chiral themselves. Let’s consider the case of achiral molecules first. Molecules that contain two or more chiral centers and at least one plane of symmetry are called meso forms. Cis-disubstituted cyclohexanes are examples of meso forms. Since these molecules have symmetry, they cannot have enantiomers. They are one and the same with their mirror images.
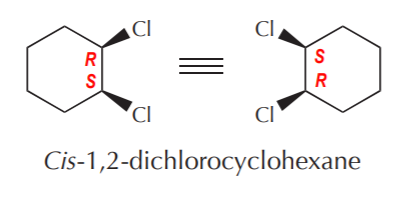
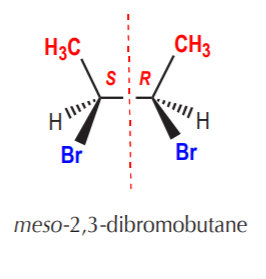
Meso forms can also be open chain, as illustrated below.
Now let’s consider the case of chiral molecules that contain two or more stereocenters. Such molecules can have enantiomers because they are not the same as their mirror images.
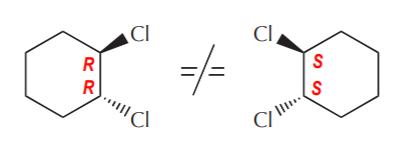
Trans-1,2-dichlorocyclohexane. The mirror images are different compounds.
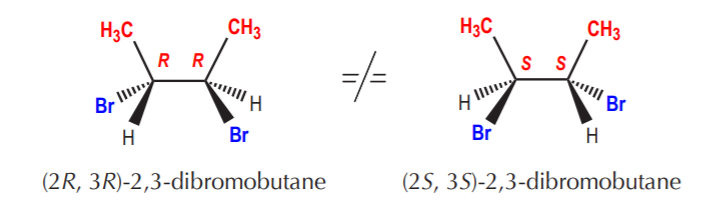
Chiral molecules with two or more chiral centers can also have stereoisomers which are not their mirror images. Such sets of stereoisomers are called diastereomers. In the following example the two molecules shown are stereoisomers (same connectivity but different spacial arrangement of the atoms) but they are not mirror images. Their relationship is one of diastereomers.
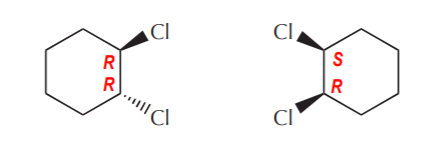
Cis- and trans-1,2-dichlorocyclohexane. are examples of diasteromers
As a corollary, we can state that cis/trans pairs of disubstituted cyclohexanes (or any other rings for this matter) are always diastereomers. Notice that we have referred to such sets before as geometric isomers. Geometric isomers are in fact a subcategory of diastereomers.
The following example is an illustration of open chain molecules with a diastereomeric relationship.
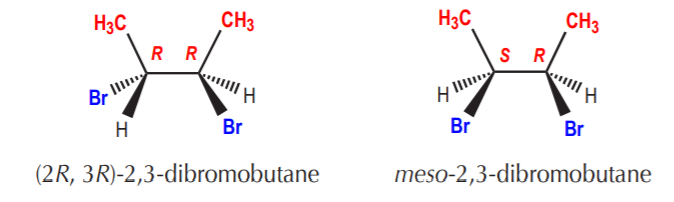
Also notice that in the above examples one of the members in each pair is chiral and the other is not. Diastereomeric sets are frequently made up of molecules where one of the molecules is chiral and the other is not. Also notice that although they are not mirror images, part of their structures do mirror each other. It is frequently the case that one half of one molecule mirrors one half of the other one, but the other halves are identical.
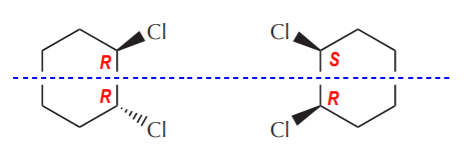
In this pair of cis/trans isomers, the top half of one molecule mirrors the top half of the other one (the chiral centers have opposite configurations), while the bottom halves are the same (the chiral centers have the same configuration).
SUMMARY OF RELATIONSHIPS BETWEEN MOLECULES WITH TWO OR MORE CHIRAL CENTERS
If n = number of chiral centers, the maximum possible number of stereoisomers is 2n
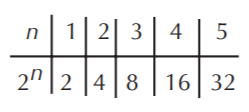
EXAMPLE 1: Possible combinations for 2,3-dibromobutane.
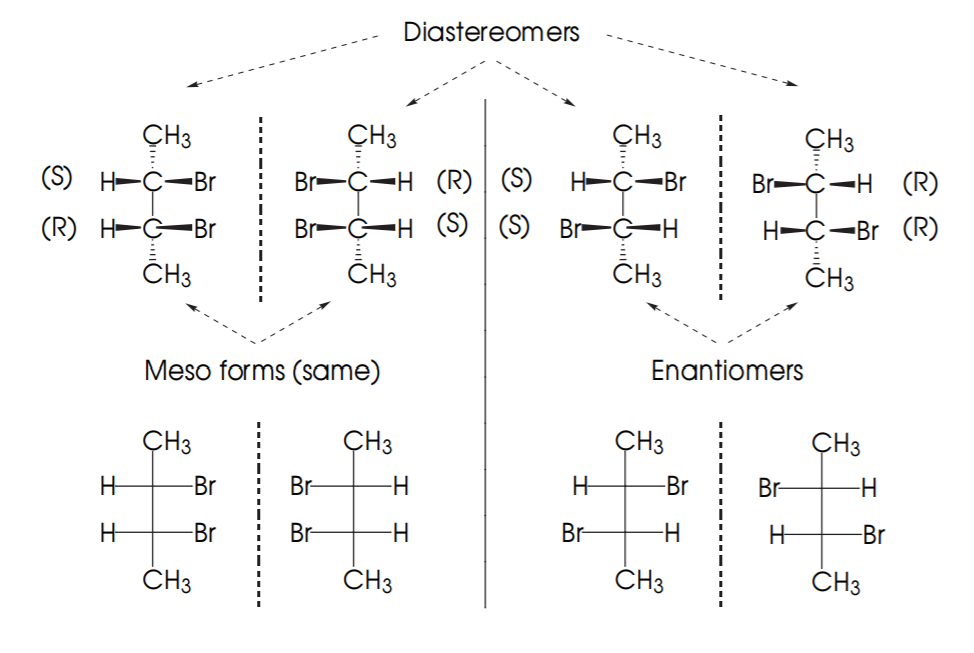
EXAMPLE 2: Possible combinations for 2-bromo-3-chlorobutane.
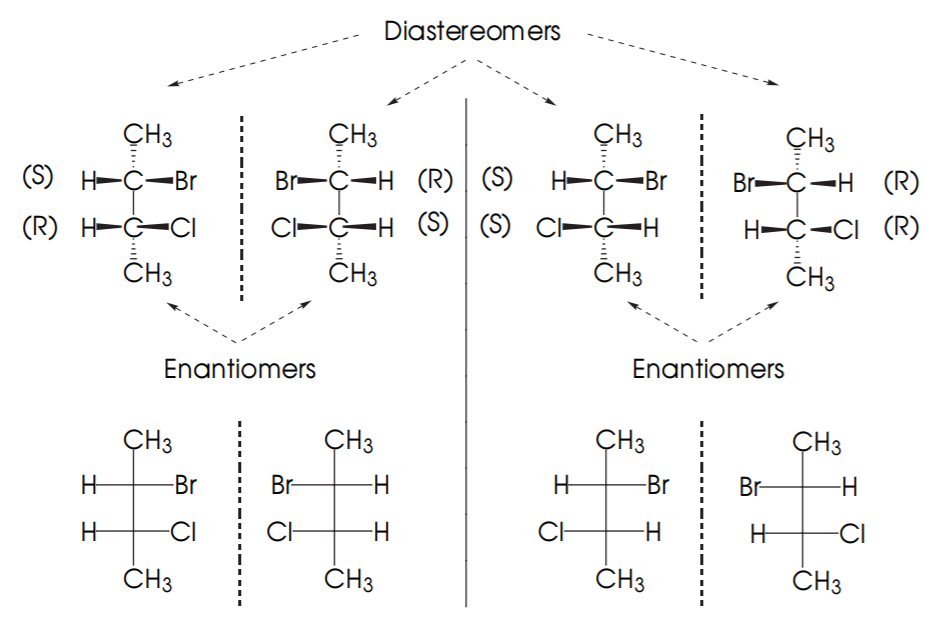
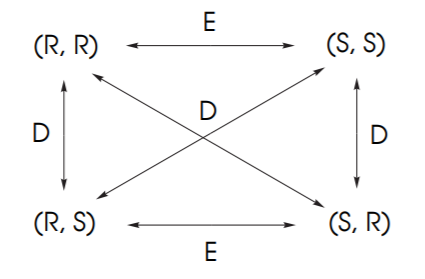
The diagram below shows the possible combinations of configurations for molecules with 2 chiral centers. Since each chiral center can be (R) or (S), the possible combinations are (R,R), (S,S), (R,S), (S,R).
If two molecules are mirror images, then their configurations are exactly opposite and they are enantiomers (E). If they are not mirror images but still they are stereoisomers then they are diastereomers (D).