30.7: Rearrangements of Cationic Oxygen
- Page ID
- 44006
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Protonation of the divalent oxygen atom of alcohols and ethers by strong acids produces a tricoordinate oxonium cation. Because the oxygen of an oxonium ion has a valence shell octet, it does not constitute an electron deficient site and cannot serve as a rearrangement terminus. To induce rearrangement in the same manner as a tricoordinate carbocation, oxygen must be converted to a unicoordinate oxacation, as noted in the following diagram. A 1,2-alkyl or aryl shift then transforms a relatively unstable oxacation into a more stable carbocation.
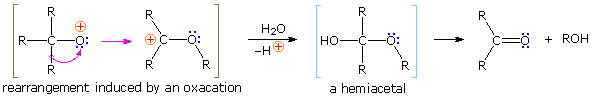
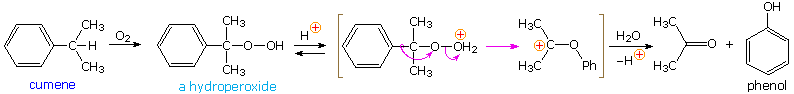
The simplest precursor of an oxa cation is a peroxide or equivalent derivative (e.g. R-O-OH or R-O-X). Removal of hydroxide anion from a hydroperoxide is energetically unfavorable, unless it is initially converted to a better leaving group in a manner similar to that used to facilitate substitution reactions of alcohols. By protonating the hydroxyl group, the leaving group becomes water, thus generating an oxacation. A useful industrial procedure for preparing phenol (and acetone) is based on this strategy.
Baeyer-Villiger Rearrangement
The acid-catalyzed reaction of ketones with hydroperoxide derivatives is known as the Baeyer-Villiger reaction. A general equation illustrating this oxidation reaction is shown below, and it may be noted that the rearrangement step is similar to that of a pinacol rearrangement. Esters or lactones are the chief products from ketone reactants. In this equation a discrete oxacation is drawn as an intermediate, but it is more likely that the rearrangement is concerted, as will be shown by clicking on the equation. Once the peracid has added to the carbonyl group, the rearrangement may be facilitated by an intramolecular hydrogen bond, in the manner depicted in brackets on the right.
The migratory aptitude of various substituent groups (e.g. 1R & 2R) is generally: 3º-alkyl > 2º-alkyl ~ benzyl ~ phenyl > 1º-alkyl > methyl. Stereoelectronic factors favor an anti-periplanar orientation of the migrating group to the leaving moiety, and will control the rearrangement in some cases. An example will be displayed below on clicking the display a second time. Peracid exchange with peracetic acid leads to an intramolecular Baeyer-Villiger reaction by way of the bicyclic acylperoxide drawn in brackets. Here stereoelectronics favor migration of the less substituted α-carbon. The lactone product was identified by esterification and ester exchange with methanol to give methyl 2-carbomethoxy-7-hydroxyheptanoate.
Aldehydes are usually oxidized to carboxylic acids under the conditions used for the Baeyer-Villiger reaction.
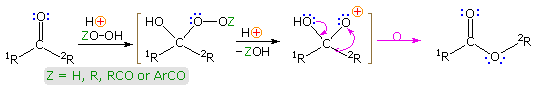
Although hydrogen peroxide itself may be used in the Bayer-Villiger reaction, it may add at both ends to reactive carbonyl groups, producing cyclic dimeric, trimeric and higher addition compounds. Consequently, derivatives such as peracids (Z = RCO & ArCO above) are the preferred reagents for this reaction. Among the most common peracids used in this respect are: peracetic acid, perbenzoic acid & meta-chloroperbenzoic acid (MCPBA). Four examples of this oxidative rearrangement are given in the following diagram. In most of these examples the migrating group retains its configuration in the course of the rearrangement, as expected for a concerted process. In example #3 it is interesting that migration of the bridgehead 3º-alkyl group is preferred over a possible phenyl shift.

