4.8: Acid-Base Extraction
- Page ID
- 93535
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)How They Work
A modification of the extractions previously discussed in this chapter is to perform a chemical reaction in the separatory funnel in order to change the polarity and therefore partitioning of a compound in the aqueous and organic layers. A common method is to perform an acid-base reaction, which can convert some compounds from neutral to ionic forms (or vice versa).
For example, imagine that a mixture of benzoic acid and cyclohexane is dissolved in an organic solvent like ethyl acetate in a separatory funnel. To separate the components, a water wash may be attempted to remove benzoic acid, but benzoic acid is not particularly water-soluble due to its nonpolar aromatic ring, and only small amounts would be extracted into the aqueous layer (Figure 4.54a).
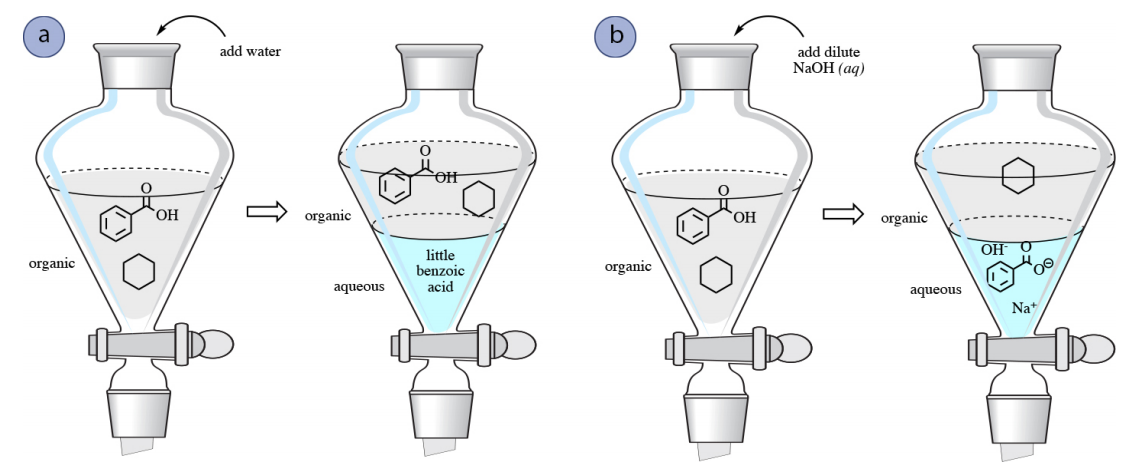
Separation of a mixture of benzoic acid and cyclohexane is however possible using a wash with a base such as \(\ce{NaOH}\). Due to its acidic nature, benzoic acid can undergo a reaction with \(\ce{NaOH}\) as follows, resulting in the carboxylate salt sodium benzoate.
\[\begin{array}{ccccccccc} \ce{PhCO_2H} \left( aq \right) & + & \ce{NaOH} \left( aq \right) & \rightarrow & \ce{H_2O} \left( l \right) & + & \ce{PhCO_2Na} \left( aq \right) & & \left( \text{or } \ce{PhCO_2^-} \ce{Na^+} \right) \\ \text{Benzoic acid} & & & & & & \text{Sodium benzoate} & & \end{array}\]
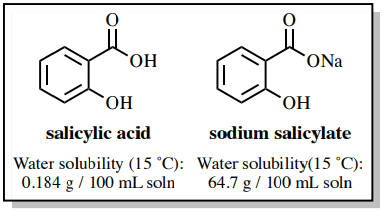
The solubility properties of carboxylic acids are substantially different than their corresponding carboxylate salts. Sodium salicylate is roughly 350 times more soluble in water than salicylic acid due to its ionic character (Figure 4.55), and it is rather insoluble in organic solvents such as diethyl ether.
Therefore, a wash with \(\ce{NaOH}\) would convert benzoic acid into its ionic carboxylate form, which would then be more soluble in the aqueous layer, allowing for the sodium benzoate to be extracted into the aqueous layer. Cyclohexane would remain in the organic layer as it has no affinity for the aqueous phase, nor can react with \(\ce{NaOH}\) in any way. In this manner, a mixture of benzoic acid and cyclohexane can be separated (Figure 4.54b). The aqueous layer may be later acidified with \(\ce{HCl} \left( aq \right)\) if desired to convert the benzoic acid back to its neutral form.
Sodium Bicarbonate Washes
An acid-base extraction can be used to extract carboxylic acids from the organic layer into the aqueous layer. As was discussed in the previous section, \(\ce{NaOH}\) can be used to convert a carboxylic acid into its more water-soluble ionic carboxylate form. However, if the mixture contains a desired compound that can react with \(\ce{NaOH}\), a milder base such as sodium bicarbonate should be used. A similar reaction occurs:
\[\begin{array}{ccccccccccc} \ce{PhCO_2H} \left( aq \right) & + & \ce{NaHCO_3} \left( aq \right) & \rightarrow & \ce{PhCO_2Na} \left( aq \right) & + & \ce{H_2CO_3} \left( aq \right) & \rightleftharpoons & \ce{H_2O} \left( l \right) & + & \ce{CO_2} \left( g \right) \\ \text{Benzoic acid} & & & & \text{Sodium benzoate} & & & & & & \end{array}\]
One difference in using the base \(\ce{NaHCO_3}\) instead of \(\ce{NaOH}\) is that the byproduct carbonic acid \(\left( \ce{H_2CO_3} \right)\) can decompose to water and carbon dioxide gas. When shaking an acidic solution with sodium bicarbonate in a separatory funnel, care should be taken to swirl gently and vent more frequently to release pressure from the gas.
An example of a reaction that often uses sodium bicarbonate wash in the work-up is a Fischer Esterification reaction. To demonstrate, benzoic acid was refluxed in ethanol along with concentrated sulfuric acid in order to form ethyl benzoate (Figure 4.56a+b). A TLC plate of the reaction mixture at 1 hour of reflux showed residual unreacted carboxylic acid (Figure 4.56c), which is not uncommon due to the energetics of the reaction.
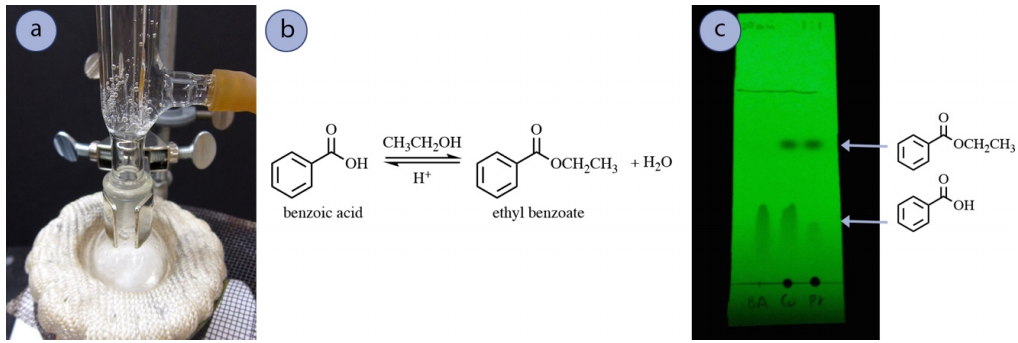
The residual carboxylic acid can be removed from the desired ester product using an acid-base extraction in a separatory funnel. A wash with sodium bicarbonate converts benzoic acid into its more water-soluble sodium benzoate form, extracting it into the aqueous layer (Figure 4.57). Additionally, the sodium bicarbonate neutralizes the catalytic acid in this reaction.
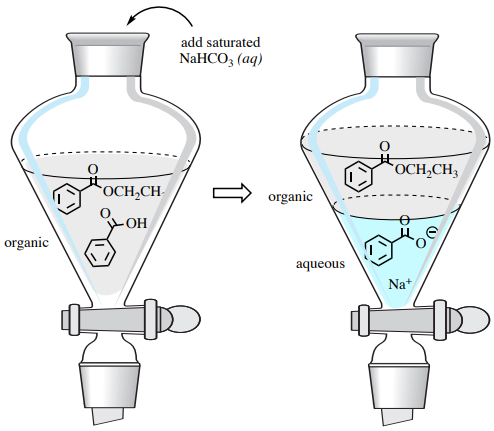
Sodium bicarbonate is preferable to \(\ce{NaOH}\) in this process, as it is a much weaker base; washing with \(\ce{NaOH}\) could cause hydrolysis of the ester product.
Mixtures of Acids and Bases
As has been discussed previously, the acid-base properties of compounds can be utilized to selectively extract certain compounds from mixtures. This strategy can be extended to other examples
Extracting Bases
Basic compounds such as amines can be extracted from organic solutions by shaking them with acidic solutions to convert them into more water-soluble salts. In this way, they can be extracted from an organic layer into an aqueous layer.
\[\begin{array}{ccccccc} \ce{PhNH_2} \left( aq \right) & + & \ce{HCl} \left( aq \right) & \rightarrow & \ce{PhNH_3Cl} \left( aq \right) & & \left( \text{or } \ce{PhNH_3^+} \ce{Cl^-} \right) \\ \text{Basic amine} & & & & \text{Ammonium salt} & & \end{array}\]
Extracting Carboxylic Acids vs. Phenols
As previously discussed, carboxylic acids can be extracted from an organic layer into an aqueous layer by shaking them with basic solutions, which converts them into their more water-soluble salts.
\[\begin{array}{ccccccccc} \ce{PhCO_2H} \left( aq \right) & + & \ce{NaOH} \left( aq \right) & \rightarrow & \ce{H_2O} \left( l \right) & + & \ce{PhCO_2Na} \left( aq \right) & & \left( \text{or } \ce{PhCO_2^-} \ce{Na^+} \right) \\ \text{Carboxylic acid} & & & & & & \text{Carboxylate salt} & & \end{array}\]
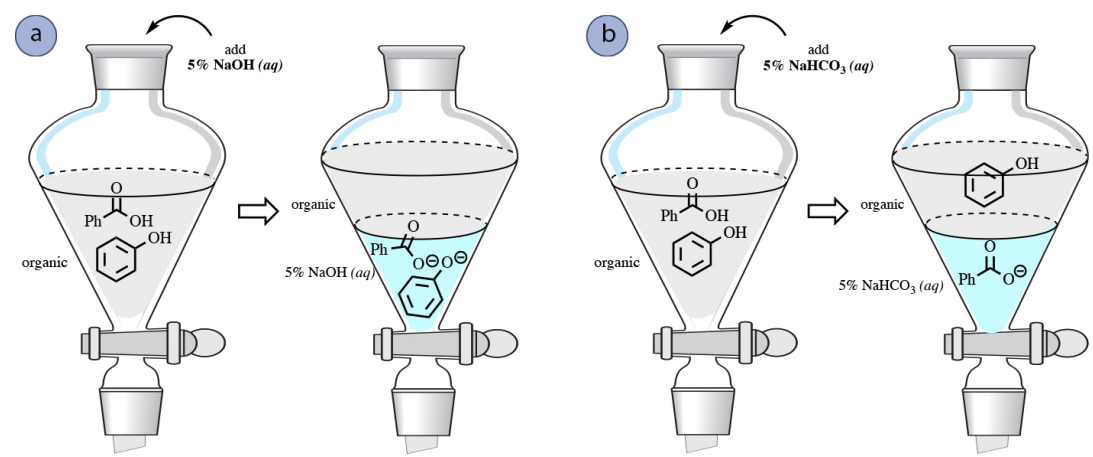
A similar reaction occurs with phenols \(\left( \ce{PhOH} \right)\), and they too can be extracted into an aqueous \(\ce{NaOH}\) layer (Figure 4.58a).
However, phenols are considerably less acidic than carboxylic acids, and are not acidic enough to react completely with \(\ce{NaHCO_3}\), a weaker base. Therefore, a solution of bicarbonate can be used to separate mixtures of phenols and carboxylic acids (Figure 4.58b).
Extracting Acid, Base, and Neutral Compounds
The acid-base properties previously discussed allow for a mixture containing acidic (e.g. \(\ce{RCO_2H}\)), basic (e.g. \(\ce{RNH_2}\)), and neutral components to be purified through a series of extractions, as summarized in Figure 4.59 (which uses an organic solvent less dense than water).
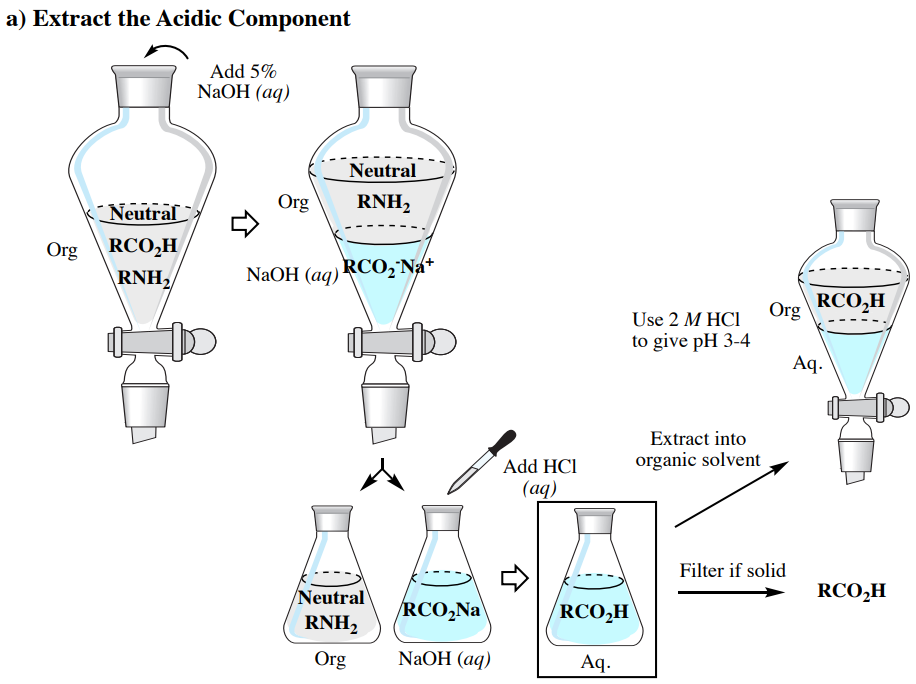
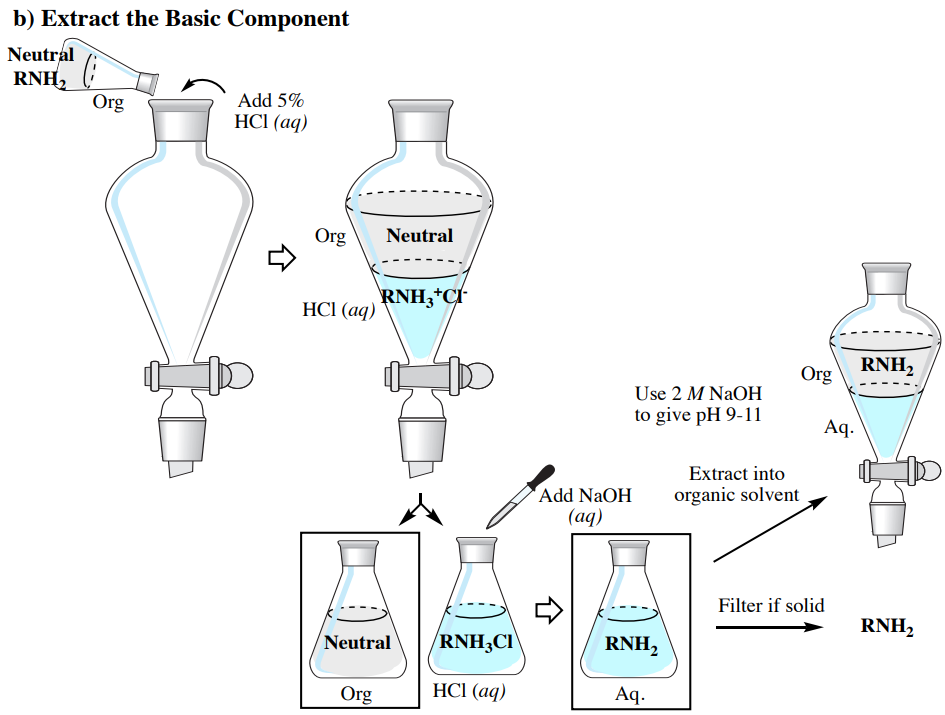
It is assumed that readers conducting this type of experiment are familiar with performing single and multiple extractions. In this section are described differences between general extraction procedures and the process as summarized in Figure 4.59.
- Isolating the Acidic component:
- When the acidic component is in the aqueous layer in an Erlenmeyer flask, it can be converted back to the neutral component through addition of \(2 \: \text{M} \: \ce{HCl} \left( aq \right)\) until the solution gives a pH of 3-4 (as determined by pH paper). If large quantities of acid are present such that acidification would require too great a volume of \(2 \: \text{M} \: \ce{HCl} \left( aq \right)\), concentrated \(\ce{HCl} \left( aq \right)\) may be instead added dropwise. Lower concentrations of \(\ce{HCl} \left( aq \right)\) are less hazardous, but increasing the volume of the aqueous layer by a large amount would affect the efficiency of subsequent extractions and filtering steps.
- After acidification, two routs may be taken, depending on if the acidic component is solid or liquid.
- If a solid forms upon acidification of the ionic salt, it can be collected through suction filtration. This method should only be used if large quantities of large-sized crystals are seen. If fine crystals form (which are quite common), they will clog the filter paper and interfere with adequate drainage. If only a small amount of solid is seen compared to the theoretical quantity, it is likely the compound is quite water-soluble, and filtration would lead to low recovery.
- If no solid forms upon acidification (or if fine crystals or low quantity of solid forms), extract the acidic component back into an organic solvent (\(\times 3\)). As a general rule of thumb, use one-third as much solvent for the extractions as the original layer (e.g. if using \(100 \: \text{mL}\) aqueous solution, extract with \(33 \: \text{mL}\) organic solvent each time). Be sure to first cool the aqueous solution in an ice bath before extraction if the acidification created noticeable heat. Follow up with a brine wash (\(\times 1\)) if using diethyl ether or ethyl acetate, dry with a drying agent, and remove the solvent via rotary evaporator to leave the pure acidic component.
- Isolating the Basic component:
- Use a similar process as the isolation of the acidic component, except basify the solution using \(2 \: \text{M} \: \ce{NaOH} \left( aq \right)\) until it gives a pH of 9-10 as determined by pH paper.
- Isolating the Neutral component:
- The neutral component will be the "leftover" compound in the organic layer. To isolate, wash with brine (\(\times 1\)) if using diethyl ether or ethyl acetate, dry with a drying agent, and remove the solvent via rotary evaporator to leave the pure neutral component.


